��Ŀ����
14������ͼת����ϵ�У��������ɫ��Ӧ�ʻ�ɫ��MΪ������Һ�����ʣ���G����Ҫ�Ļ�����Ʒ�ͻ���ԭ�ϣ�����H���ܽ���A��Һ����G�У���HΪ���õ��ͻ���ϣ�ͼ�в��ֲ���û���г�����
��1��д��M�����Ӧ�Ļ�ѧ����ʽ��2Na2O2+2H2O=4NaOH+O2��
��2����ͼ��ʾ����������G�Ĺ�ҵ���̣�

���豸�ҵ�����Ϊ�Ӵ��ң�
������X����Ҫ�ɷ�ΪSO2��O2��N2��
��д���豸���г����Ļ�ѧ��Ӧ4FeS2+11O2$\frac{\underline{\;����\;}}{\;}$2Fe2O3+8SO2��
��3����������M��Ӧ�Ļ�ѧ����ʽΪAl2S3+6H2O$\frac{\underline{\;\;��\;\;}}{\;}$2Al��OH��3��+3H2S����
���� �������ɫ��Ӧ�ʻ�ɫ�����к�����Ԫ�أ�MΪ������Һ�����ʣ����ܹ���M��Ӧ����A��Һ������B�����ΪNa2O2��MΪH2O��AΪNaOH��BΪO2������H���ܽ���A��Һ����G�У���HΪ���õ��ͻ���ϣ���HΪAl2O3����ɫ����DΪAl��OH��3������C����������������ˮ��Ӧ������G����G����Ҫ�Ļ�����Ʒ�ͻ���ԭ�ϣ�����������Ϣ���ɵ�98%����G����GΪH2SO4������EΪSO2��FΪSO3������B��C��Ӧ���ɶ��������ˮ����CΪH2S����������ˮ��Ӧ�����������������������ΪAl2S3����ҵ�ϳ����ᣬ����¯�в���SO2���ӷ���¯�г�����������������������������������Ӵ��ң��õ�����SO3���ӽӴ��ҳ�����ѭ�����õ�����X����SO2��O2��N2�����������������ˮ��98%��H2SO4���Դ˽����⣮
��� �⣺�������ɫ��Ӧ�ʻ�ɫ�����к�����Ԫ�أ�MΪ������Һ�����ʣ����ܹ���M��Ӧ����A��Һ������B�����ΪNa2O2��MΪH2O��AΪNaOH��BΪO2������H���ܽ���A��Һ����G�У���HΪ���õ��ͻ���ϣ���HΪAl2O3����ɫ����DΪAl��OH��3������C����������������ˮ��Ӧ������G����G����Ҫ�Ļ�����Ʒ�ͻ���ԭ�ϣ�����������Ϣ���ɵ�98%����G����GΪH2SO4������EΪSO2��FΪSO3������B��C��Ӧ���ɶ��������ˮ����CΪH2S����������ˮ��Ӧ�����������������������ΪAl2S3����ҵ�ϳ����ᣬ����¯�в���SO2���ӷ���¯�г�����������������������������������Ӵ��ң��õ�����SO3���ӽӴ��ҳ�����ѭ�����õ�����X����SO2��O2��N2�����������������ˮ��98%��H2SO4��
��1��MΪH2O����ΪNa2O2������M�����Ĺ����Ӧ�Ļ�ѧ����ʽΪ2Na2O2+2H2O=4NaOH+O2�����ʴ�Ϊ��2Na2O2+2H2O=4NaOH+O2����
��2�����ٸ�������ķ�����֪���豸�ҵ�����Ϊ�Ӵ��ң�
�ʴ�Ϊ���Ӵ��ң�
�ڸ�������ķ�����֪������X����Ҫ�ɷ�ΪSO2��O2��N2��
�ʴ�Ϊ��SO2��O2��N2��
�۷���¯�г����Ļ�ѧ��ӦΪ4FeS2+11O2 $\frac{\underline{\;����\;}}{\;}$2Fe2O3+8SO2��
�ʴ�Ϊ��4FeS2+11O2 $\frac{\underline{\;����\;}}{\;}$2Fe2O3+8SO2��
��3����ΪAl2S3������ˮ��Ӧ�Ļ�ѧ����ʽΪAl2S3+6H2O$\frac{\underline{\;\;��\;\;}}{\;}$2Al��OH��3��+3H2S����
�ʴ�Ϊ��Al2S3+6H2O$\frac{\underline{\;\;��\;\;}}{\;}$2Al��OH��3��+3H2S����
���� ���⿼��������ƶϣ�Ϊ�߿��������ͣ�������ѧ���ķ��������Ŀ��飬ע�����ʱ������Ҫ����Ŀ���ڳ�һЩ���Ի�������������ץסͻ�ƿڣ�ͻ�ƿ�������������������Ӧ�������ṹ���������ó����ۣ��������˰ѽ��۴���ԭ������֤������·���ߵ�ͨ����ȷ���ۺϿ���Ԫ�ػ��������ʼ���ѧ�����Ŀ�Ѷ��еȣ�

 ��ʦ�㾦�ִʾ��ƪϵ�д�
��ʦ�㾦�ִʾ��ƪϵ�д�| A�� | ������CuSO4���� | B�� | ��п�۴���п�� | C�� | ��ˮϡ��H2SO4 | D�� | �ʵ����� |
| A�� | ������FeCl3��Һ�ķ�Ӧ��Fe+2Fe3+�T3Fe2+ | |
| B�� | ̼���������ķ�Ӧ��CO32-+2H+�TH2O+CO2�� | |
| C�� | ������������ͭ�ķ�Ӧ��2H++Cu��OH��2�TCu2++2H2O | |
| D�� | ����NaͶ��ˮ�еķ�Ӧ��2Na+2H2O�T2Na++2OH-+H2�� |
| A�� | ��ϡHNO3�еμ� Na2SO3��Һ��SO32-+2H+=SO2��+H2O | |
| B�� | ��Na2SiO3��Һ��ͨ�����SO2��SiO32-�TSO2+H2O | |
| C�� | ��CuSO4��Һ�м���Na2O2��2Na2O2+2Cu2++2H2O=4Na++2Cu ��OH��2��+O2�� | |
| D�� | ��A12��SO4��3��Һ�м��������NO3•H2O��Al3++4 NH3•H2O=[A1��OH��4]-+4NH4+ |



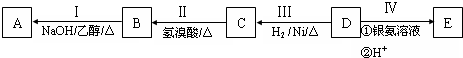
 ��
��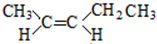 ��
��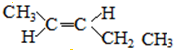 ��
��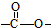 �ṹ����12�֣�E����һ��ͬ���칹���ܷ���������Ӧ�����������������������������ܷ�����ȥ��Ӧ����ṹ��ʽΪ
�ṹ����12�֣�E����һ��ͬ���칹���ܷ���������Ӧ�����������������������������ܷ�����ȥ��Ӧ����ṹ��ʽΪ ��
��