��Ŀ����
11���������ڱ�ǰ�����ڵ�H��C��N��O��F��Mg��Cl��K��Ca��Ni��Br��ʮһ��Ԫ�أ���1��C��N��O����Ԫ�صĵ�һ�����ܴ�С�����˳��Ϊ��C��O��N
��2��N��O��F����Ԫ�صĵ縺�Դ�С�����˳��Ϊ��N��O��F
��3��MgO��CaO��KCl���۵��ɵ͵��ߵ�˳��Ϊ��
��4��HF��HCl��HBr����ͬѹǿ�·е��ɵ͵��ߵ�˳��Ϊ��HCl��HBr��HF
��5��Ni��CO��4���ʻ������������Ƹߴ����ۼ��������ȣ�
����λ�����ڱ���������VIII�壮
��Ni��CO��4��Ni��CO��Cԭ���γ���λ���������ǿռ乹�ͣ�Ni��CO��4�Ľṹ����ʾ��ͼ��ʾΪ
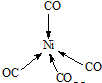 ��
��
���� ��1��ͬһ����Ԫ�صĵ�һ����������ԭ���������������С��ͬһ����Ԫ�صĵ�һ����������ԭ�����������������ע��ͬһ���ڵĵڢ�AԪ�صĵ�һ�����ܴ��ڵڢ�A��ģ��ڢ�A��Ĵ��ڵڢ�A��ģ�
��2��ͬ���ڴ�����Ԫ�صĵ縺������
��3�����Ӿ����۷е��뾧���ܳ����ȣ������������Ӱ뾶�ɷ��ȡ����ɳ����ȣ�
��4��HF���Ӽ��������е�ϸߣ���Է�������Խ���Ӽ�������Խ�е�Խ�ߣ�
��5����Ni��ԭ������Ϊ28��
��Ni��CO��4��Ni��CO��Cԭ���γ���λ�����γ�������ṹ��
��� �⣺��1��C��N��O����ͬһ����Ԫ����ԭ���������μ�С��ͬһ����Ԫ�صĵ�һ����������ԭ������������������ڢ�A��Ĵ��ڵڢ�A��ģ��������һ�����ܴ�С˳����C��O��N��
�ʴ�Ϊ��C��O��N��
��2��ͬ���ڴ�����Ԫ�صĵ縺��������縺�Դ�С�����˳��Ϊ��N��O��F��
�ʴ�Ϊ��N��O��F��
��3�����Ӿ����۷е��뾧���ܳ����ȣ������������Ӱ뾶�ɷ��ȡ����ɳ����ȣ�þ���ӡ������ӡ������ӵ�ɴ��ڼ����Ӻ������ӣ������Ӱ뾶���ڸ����ӣ������Ӵ���þ���ӣ������Ӱ뾶���������ӣ����Ծ�������С�����˳����KCl��CaO��MgO�����۵��ɵ͵��ߵ�˳��ΪKCl��CaO��MgO��
�ʴ�Ϊ��KCl��CaO��MgO��
��4��HF���Ӽ��������е�ϸߣ���Է�������Խ���Ӽ�������Խ�е�Խ�ߣ���е��ɵ͵��ߵ�˳��ΪHCl��HBr��HF��
�ʴ�Ϊ��HCl��HBr��HF��
��5����Ni��ԭ������Ϊ28�������ڱ���λ�ڵ������ڡ���VIII�壻
�ʴ�Ϊ����VIII��
��Ni��CO��4��Ni��CO��Cԭ���γ���λ�����γ�������ṹ����ṹͼΪ ��
��
�ʴ�Ϊ�� ��
��
���� �����Ƕ����ʽṹ�Ŀ��飬�漰��һ�����ܡ��縺�ԡ��۷е�Ƚϡ�����������ȣ��������֪ʶ�ǽ��ؼ�����Ŀ�Ѷ��еȣ������ڿ���ѧ���Ի���֪ʶ��Ӧ��������

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | ��ȡ8gNaOH���壬����100mL��Ͳ�У��߽����������������ˮ����������ȫ�ܽ��������ˮϡ����100mL | |
| B�� | ��ȡ8gNaOH���壬����100mL����ƿ�У�������������ˮ��������ƿʹ�����ܽ⣬�ټ���ˮ���̶ȣ��Ǻ�ƿ��������ҡ�� | |
| C�� | ��ȡ8gNaOH���壬����100mL�ձ��У����ձ��м�������ˮ��ͬʱ���Ͻ����������ܽ� | |
| D�� | ��100mL��Ͳ��ȡ40ml 5mol/L NaOH��Һ��ϡ�ͺ�ת����100ml����ƿ�У�ϴ�Ӳ����� |
| A�� | MA=MB | B�� | nA��nB | C�� | nA��nB | D�� | MA��MB |
| A�� | CH2�TCH-CH�TCH21��3-����ϩ | B�� | CH2BrCH2Br �������� | ||
| C�� | CH3CH2CH��OH��CH33-���� | D�� | 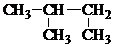 2-������ 2-������ |
| A�� | ��������춡�� | B�� | ���ۺ���ά�� | ||
| C�� | 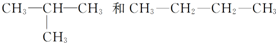 | D�� | CH3CH2COOH��CH3COOCH3 |
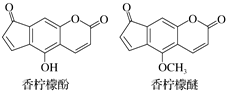 ���������Ǵ����������Ĺ�Ƥ������������ζ�����ͣ�33%��Ů����ˮ�õ�������ԭ�ϣ��������ͺ����������ʷӺ��������ѣ���ṹ��ͼ������˵����ȷ���ǣ�������
���������Ǵ����������Ĺ�Ƥ������������ζ�����ͣ�33%��Ů����ˮ�õ�������ԭ�ϣ��������ͺ����������ʷӺ��������ѣ���ṹ��ͼ������˵����ȷ���ǣ�������| A�� | �������ѵķ���ʽΪC13H6O4 | |
| B�� | �����ʷӿ�����ᷴӦת������������ | |
| C�� | 1 mol�����ʷ�������2 mol Br2������Ӧ | |
| D�� | 1 mol�����ʷ���������3 mol NaOH��Һ��Ӧ |
| A�� | v��NH3��=0.002mol/��L•s�� | B�� | v��O2��=0.01mol/��L•s�� | ||
| C�� | v��NO��=0.01mol/��L•s�� | D�� | v��H2O��=0.001mol/��L•s�� |

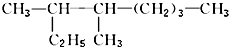 ������3��4-�������飮
������3��4-�������飮 ��
��