��Ŀ����
11��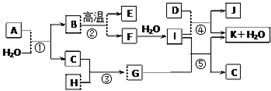 ��ͼ��ʾ����������1��20��Ԫ���в���Ԫ����ɵĵ��ʻ��仯���ͼ�в��ַ�Ӧ����δ�г�����֪C��H����ɫ�д̼�����ζ�����壬D��һ�ֻ���ɫ�����嵥�ʣ�����J������Ӿ����������Ӧ�ں͢��ǻ��������е���Ҫ��Ӧ����Ӧ����ʵ�����Ʊ�����C����Ҫ������
��ͼ��ʾ����������1��20��Ԫ���в���Ԫ����ɵĵ��ʻ��仯���ͼ�в��ַ�Ӧ����δ�г�����֪C��H����ɫ�д̼�����ζ�����壬D��һ�ֻ���ɫ�����嵥�ʣ�����J������Ӿ����������Ӧ�ں͢��ǻ��������е���Ҫ��Ӧ����Ӧ����ʵ�����Ʊ�����C����Ҫ��������ش��������⣺
��1������E�ĵ���ʽ
 ��I��������ѧ���������Ӽ������ۼ���
��I��������ѧ���������Ӽ������ۼ�����2��G��ˮ��Һ�����ԣ������ӷ���ʽ��ʾ��ԭ��NH4++H2O?NH3•H2O+H+��
��3����֪��7.4gI��ϡ��Һ��200mL1mol/L��H��Һ��Ӧ�ų�11.56kJ��������д���÷�Ӧ���Ȼ�ѧ����ʽCa��OH��2��aq��+2NH4Cl��aq��=CaCl2��aq��+2NH3����g��+2H2O��l����H=-115.6kJ/mol��
��4������A������Ԫ����ɣ�1mol A��ˮ��Ӧֻ����1mol B��2mol C��A��ѧʽΪCaCN2��
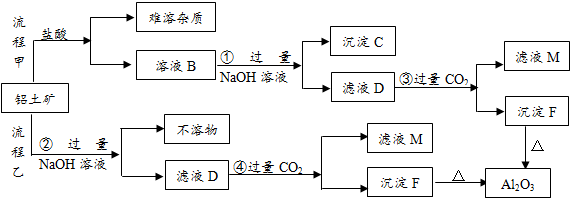
���� ͼ�и���������1��20��Ԫ���в���Ԫ����ɵĵ��ʻ��仯���D��һ�ֻ���ɫ�����嵥�ʣ���DΪCl2������J��������ˮ��������Ӧ���ǻ��������е���Ҫ��Ӧ��Ӧ�ǹ�ҵ�Ʊ�Ư��ԭ������IΪCa��OH��2��JΪCa��ClO��2��KΪCaCl2��C����ɫ�д̼�����ζ�����壬��Ӧ����ʵ�����Ʊ�����C����Ϸ�Ӧ�������ɿ�֪��Ϊ�Ʊ������ķ�Ӧ����CΪNH3��GΪNH4Cl�����ת����ϵ��֪HΪHCl��F��ˮ��Ӧ����Ca��OH��2����FΪCaO����Ӧ���ǻ��������е���Ҫ��Ӧ��Ϊ̼��Ƹ��·ֽⷴӦ����BΪCaCO3��EΪCO2������A������Ԫ����ɣ�1molA��ˮ��Ӧ������1molB��CaCO3����2molC��NH3��������Ԫ���غ��֪A����Ca��C��N����Ԫ�أ���ԭ���غ��֪��A�Ļ�ѧʽΪCaCN2���ݴ˽��
��� �⣺ͼ�и���������1��20��Ԫ���в���Ԫ����ɵĵ��ʻ��仯���D��һ�ֻ���ɫ�����嵥�ʣ���DΪCl2������J��������ˮ��������Ӧ���ǻ��������е���Ҫ��Ӧ��Ӧ�ǹ�ҵ�Ʊ�Ư��ԭ������IΪCa��OH��2��JΪCa��ClO��2��KΪCaCl2��C����ɫ�д̼�����ζ�����壬��Ӧ����ʵ�����Ʊ�����C����Ϸ�Ӧ�������ɿ�֪��Ϊ�Ʊ������ķ�Ӧ����CΪNH3��GΪNH4Cl�����ת����ϵ��֪HΪHCl��F��ˮ��Ӧ����Ca��OH��2����FΪCaO����Ӧ���ǻ��������е���Ҫ��Ӧ��Ϊ̼��Ƹ��·ֽⷴӦ����BΪCaCO3��EΪCO2������A������Ԫ����ɣ�1molA��ˮ��Ӧ������1molB��CaCO3����2molC��NH3��������Ԫ���غ��֪A����Ca��C��N����Ԫ�أ���ԭ���غ��֪��A�Ļ�ѧʽΪCaCN2��
��1��EΪCO2�������ʽΪ ��IΪCa��OH��2���������Ӽ������ۼ���
��IΪCa��OH��2���������Ӽ������ۼ���
�ʴ�Ϊ�� �����Ӽ������ۼ���
�����Ӽ������ۼ���
��2��GΪNH4Cl��G��ˮ��Һ�����ԣ���Ӧ�����ӷ���ʽΪNH4++H2O?NH3•H2O+H+��
�ʴ�Ϊ��NH4++H2O?NH3•H2O+H+��
��3��7.4gCa��OH��2Ϊ0.1mol��200mL1mol/L��HCl��Һ�к���0.2molHCl�����߷�Ӧ�ų�11.56kJ����������1molCa��OH��2��2molHCl��Ӧ����NH4Cl�ų�115.6kJ�����������Է�Ӧ���Ȼ�ѧ����ʽΪCa��OH��2��aq��+2NH4Cl��aq��=CaCl2��aq��+2NH3����g��+2H2O��l����H=-115.6kJ/mol��
�ʴ�Ϊ��Ca��OH��2��aq��+2NH4Cl��aq��=CaCl2��aq��+2NH3����g��+2H2O��l����H=-115.6kJ/mol��
��4������A������Ԫ����ɣ�1molA��ˮ��Ӧ������1molCaCO3��2molNH3������Ԫ���غ��֪A����Ca��C��N����Ԫ�أ���ԭ���غ��֪��A�Ļ�ѧʽΪCaCN2��
�ʴ�Ϊ��CaCN2��
���� ���⿼��������ƶϣ���D�ǻ���ɫ���嵥�ʡ���ͻ�ƿڣ����J����;����ѧ��������Ҫ������Ӧ�����ƶϣ�����ѧ����Ԫ�ػ�����֪ʶ���������տ��飬�Ѷ��еȣ�

| A�� | ͨ������SO2����ҺƯ������ǿ | |
| B�� | ��������NaHCO3���壬c��ClO����С | |
| C�� | ��������ˮ��ˮ�ĵ���ƽ�����������ƶ� | |
| D�� | ����NaOH������ǡ����ȫ��Ӧ��һ����c��Na+��=c��Cl-��+c��ClO-��+c��HClO�� |
| A�� | 1��2��3 | B�� | 3��4��3 | C�� | 6��3��2 | D�� | 4��3��4 |
 ���ж���������Ԫ��X��Y��Z��R��T��Rԭ�������������ǵ��Ӳ�����2����Y��Z���γ�Z2Y��Z2Y2�����ӻ����Z��T�γɵ�Z2T ���������ƻ�ˮ�ĵ���ƽ�⣮����Ԫ�ص�ԭ�Ӱ뾶��ԭ�������Ĺ�ϵ��ͼ��ʾ�������ƶ���ȷ����
���ж���������Ԫ��X��Y��Z��R��T��Rԭ�������������ǵ��Ӳ�����2����Y��Z���γ�Z2Y��Z2Y2�����ӻ����Z��T�γɵ�Z2T ���������ƻ�ˮ�ĵ���ƽ�⣮����Ԫ�ص�ԭ�Ӱ뾶��ԭ�������Ĺ�ϵ��ͼ��ʾ�������ƶ���ȷ����A��B����������
| A�� | ԭ�Ӱ뾶�����Ӱ뾶�����㣺Y��Z | |
| B�� | �⻯��ķе�����Y��T��R | |
| C�� | ����������Ӧ��ˮ��������ԣ�T��R | |
| D�� | ���������ԣ�Y��R��T |
| A�� | 1.2 mol | B�� | 1.5mol | C�� | 3.0 mol | D�� | 2.0 mol |
| A�� | ����ʹ������Ȼ�̼��Һ��ɫ | B�� | ��ʹ���Ը��������Һ��ɫ | ||
| C�� | ���ܷ����ӳɷ�Ӧ | D�� | ���ڸ߷��ӻ����� |