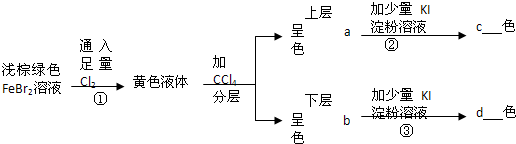
��Ŀ����
�л���A��B��C��D���������ʣ�
��1������֧���Ļ�����A�ķ���ʽΪC4H6O2��A����ʹBr2�����Ȼ�̼��Һ��ɫ��1mol A��1mol NaHCO3����ȫ��Ӧ����A�Ľṹ��ʽ�� ��
д����A������ͬ�����ŵ�A������ͬ���칹��Ľṹ��ʽ ��
��2��������B����C��H��O����Ԫ�أ�������Ϊ60������̼����������Ϊ60%�������������Ϊ13.33%��B�ڴ���Cu�������±�������C��C�ܷ���������Ӧ����B�Ľṹ��ʽ�� ��
��3��D��NaOHˮ��Һ�м��ȷ�Ӧ��������A�����κ�B����Ӧ��Ӧ�Ļ�ѧ����ʽ�� ��
��1������֧���Ļ�����A�ķ���ʽΪC4H6O2��A����ʹBr2�����Ȼ�̼��Һ��ɫ��1mol A��1mol NaHCO3����ȫ��Ӧ����A�Ľṹ��ʽ��
д����A������ͬ�����ŵ�A������ͬ���칹��Ľṹ��ʽ
��2��������B����C��H��O����Ԫ�أ�������Ϊ60������̼����������Ϊ60%�������������Ϊ13.33%��B�ڴ���Cu�������±�������C��C�ܷ���������Ӧ����B�Ľṹ��ʽ��
��3��D��NaOHˮ��Һ�м��ȷ�Ӧ��������A�����κ�B����Ӧ��Ӧ�Ļ�ѧ����ʽ��
���㣺�л�����ƶ�
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
��������1��A�ķ���ʽΪC4H6O2��1mol A��1mol NaHCO3����ȫ��Ӧ��A���Ӻ���1��-COOH��A����ʹBr2�����Ȼ�̼��Һ��ɫ��A�IJ����Ͷ�Ϊ
=2���ʻ�����1��̼̼˫������A����֧������AΪ ��
��
��2��������B����C��H��O����Ԫ�أ�������Ϊ60��̼����������Ϊ60%�������������Ϊ13.33%���������N��C��=
=3��N��H��=
=8��N��O��=
=1����B�ķ���ʽΪC3H8O��B�ڴ���Cu�������±�������C��C�ܷ���������Ӧ����BΪCH3CH2OH��CΪCH3CH2CHO��
��3��D��NaOHˮ��Һ�м��ȷ�Ӧ��������A�����κ�B����DΪCH2=C��CH3��COOCH2CH2CH3��
| 2��4+2-6 |
| 2 |
 ��
����2��������B����C��H��O����Ԫ�أ�������Ϊ60��̼����������Ϊ60%�������������Ϊ13.33%���������N��C��=
| 60��60% |
| 12 |
| 60��13.33% |
| 1 |
| 60-12��3-8 |
| 16 |
��3��D��NaOHˮ��Һ�м��ȷ�Ӧ��������A�����κ�B����DΪCH2=C��CH3��COOCH2CH2CH3��
���
�⣺��1��A�ķ���ʽΪC4H6O2��1mol A��1mol NaHCO3����ȫ��Ӧ��A���Ӻ���1��-COOH��A����ʹBr2�����Ȼ�̼��Һ��ɫ��A�IJ����Ͷ�Ϊ
=2���ʻ�����1��̼̼˫������A����֧������AΪ ����A������ͬ�����ŵ�A������ͬ���칹��Ľṹ��ʽΪ��CH2=CH-CH2-COOH CH3-CH=CH-COOH���ʴ�Ϊ����CH2=CH-CH2-COOH CH3-CH=CH-COOH��
����A������ͬ�����ŵ�A������ͬ���칹��Ľṹ��ʽΪ��CH2=CH-CH2-COOH CH3-CH=CH-COOH���ʴ�Ϊ����CH2=CH-CH2-COOH CH3-CH=CH-COOH��
��2��������B����C��H��O����Ԫ�أ�������Ϊ60��̼����������Ϊ60%�������������Ϊ13.33%���������N��C��=
=3��N��H��=
=8��N��O��=
=1����B�ķ���ʽΪC3H8O��B�ڴ���Cu�������±�������C��C�ܷ���������Ӧ����BΪCH3CH2CH2OH��CΪCH3CH2CHO���ʴ�Ϊ��CH3CH2CH2OH��
��3��D��NaOHˮ��Һ�м��ȷ�Ӧ��������A�����κ�B����DΪCH2=C��CH3��COOCH2CH2CH3���÷�Ӧ����ʽΪ�� ��
��
�ʴ�Ϊ�� ��
��
| 2��4+2-6 |
| 2 |
 ����A������ͬ�����ŵ�A������ͬ���칹��Ľṹ��ʽΪ��CH2=CH-CH2-COOH CH3-CH=CH-COOH���ʴ�Ϊ����CH2=CH-CH2-COOH CH3-CH=CH-COOH��
����A������ͬ�����ŵ�A������ͬ���칹��Ľṹ��ʽΪ��CH2=CH-CH2-COOH CH3-CH=CH-COOH���ʴ�Ϊ����CH2=CH-CH2-COOH CH3-CH=CH-COOH����2��������B����C��H��O����Ԫ�أ�������Ϊ60��̼����������Ϊ60%�������������Ϊ13.33%���������N��C��=
| 60��60% |
| 12 |
| 60��13.33% |
| 1 |
| 60-12��3-8 |
| 16 |
��3��D��NaOHˮ��Һ�м��ȷ�Ӧ��������A�����κ�B����DΪCH2=C��CH3��COOCH2CH2CH3���÷�Ӧ����ʽΪ��
 ��
���ʴ�Ϊ��
 ��
��
���������⿼���л����ƶϣ��ѶȲ�����ȷ��B�ķ���ʽ�����ݷ����ķ�Ӧ�жϺ��еĹ����ţ�ȷ���л���A��B�Ľṹ����Ҫѧ���������չ����ŵ�������ת����

��ϰ��ϵ�д�
�����Ŀ
����UIC��ѧ��λ���Ҽ���Ⱦ��ѧר����Ϊ����ǰ���Ƶİ����������Ѿ��ܹ�ͨ�����ܽ��������ݴˣ�����˵����ȷ���ǣ�������
| A��������������ͨ���������� |
| B��������ֿ���ЧԤ��������������Ⱦ |
| C��Ŀǰ���ҵ���Ч���ư����������ķ��� |
| D����������������ֱ����Լ80nm |
����˵����ȷ���ǣ�������
| A��CH4��Ħ������Ϊ16 |
| B��1 mol H2O������Ϊ18 g/mol |
| C��ͬ��ͬѹ�£�������ܶ����������Է������������� |
| D������ͬ�¶������£�������Ӽ��ƽ�����뼸����� |
����˵���У���ȷ���ǣ�������
A��ijԭ�ӣ�6�����ӡ�7�����ӣ��Ļ�ѧ������
| ||
| B��H2O��CH4�����о�����ͬ����������ԭ���� | ||
| C��D��T����Ԫ�ص����ֲ�ͬ�ĺ��� | ||
| D��ϡ�������ԭ�������ﵽ�ȶ��ṹ���ʶ������������ʷ�����Ӧ |
���й���MgO��Al2O3���ʺ���;��˵����������ʵ���ǣ�������
| A�����������ᷴӦ |
| B����������ˮ |
| C�����������ͻ���� |
| D������������������Һ��Ӧ |