��Ŀ����
6��ij���᳦��ҩ����Ч�ɷֵĺϳ�·�����£����ַ�Ӧ��ȥ�Լ�����������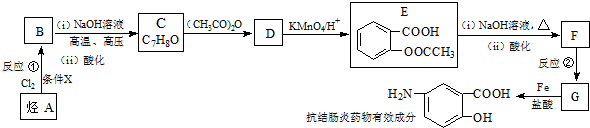
��֪����a��
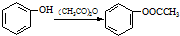
��b��
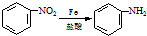
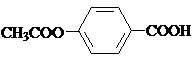
��ش��������⣺
��1����A�Ľṹ��ʽ��
 ����Ӧ�ٵķ�Ӧ������FeCl3����������Ӧ�ڵķ�Ӧ������ȡ����Ӧ��
����Ӧ�ٵķ�Ӧ������FeCl3����������Ӧ�ڵķ�Ӧ������ȡ����Ӧ����2�������жԸÿ��᳦��ҩ����Ч�ɷֿ��ܾ��е������Ʋ���ȷ����AD��
A��ˮ���Աȱ��Ӻ� B���ܷ�����ȥ��ӦҲ�ܷ����ۺϷ�Ӧ
C��1mol������������3mol�巢����Ӧ D�������������м���
��E������NaOH��Һ��Ӧ�Ļ�ѧ����ʽ��
 ��
����3��д����������������E������ͬ���칹��Ľṹ��ʽ��
 ��
�� ��
��a����E������ͬ�Ĺ�����
b�������ϵ�һ����ȡ������������
c���˴Ź��������Ϲ���4����
��4����֪������
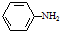 ���ױ��������������������ʱ������һ��ȡ��������ȡ����������ڶ�λ�����������������Ȼ�ʱ��ȡ���ڼ�λ���ݴ˰��Ⱥ�˳��д����AΪԭ�Ϻϳ��ڰ��������ᣨ
���ױ��������������������ʱ������һ��ȡ��������ȡ����������ڶ�λ�����������������Ȼ�ʱ��ȡ���ڼ�λ���ݴ˰��Ⱥ�˳��д����AΪԭ�Ϻϳ��ڰ��������ᣨ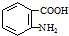 ���ϳ�·���������м����Ľṹ��ʽ�����ַ�Ӧ��������ȥ��
���ϳ�·���������м����Ľṹ��ʽ�����ַ�Ӧ��������ȥ��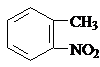 ��
��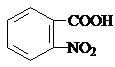 ��
��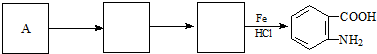
���� C������Ϣ��a���еķ�Ӧ����D��D����������Ӧ����E�����C�ķ���ʽ��E�Ľṹ��ʽ����֪C�Ľṹ��ʽΪ ����DΪ
����DΪ ����A��������FeCl3���������·�Ӧ�õ�B��B����ˮ�ⷴӦ���ữ�õ�C��AΪ
����A��������FeCl3���������·�Ӧ�õ�B��B����ˮ�ⷴӦ���ữ�õ�C��AΪ ��BΪ
��BΪ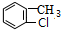 ��E���������Ʒ�Ӧ���ữ�õ�FΪ
��E���������Ʒ�Ӧ���ữ�õ�FΪ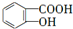 ������Ϣ��b������Ͽ��᳦��ҩ����Ч�ɷֵĽṹ��֪��F��Ũ���ᡢŨ������������·�����Ӧ����GΪ
������Ϣ��b������Ͽ��᳦��ҩ����Ч�ɷֵĽṹ��֪��F��Ũ���ᡢŨ������������·�����Ӧ����GΪ ��G������ԭ��Ӧ�õ����᳦��ҩ����Ч�ɷ֣��ݴ˽��
��G������ԭ��Ӧ�õ����᳦��ҩ����Ч�ɷ֣��ݴ˽��
��� �⣺C������Ϣ��a���еķ�Ӧ����D��D����������Ӧ����E�����C�ķ���ʽ��E�Ľṹ��ʽ����֪C�Ľṹ��ʽΪ ����DΪ
����DΪ ����A��������FeCl3���������·�Ӧ�õ�B��B����ˮ�ⷴӦ���ữ�õ�C��AΪ
����A��������FeCl3���������·�Ӧ�õ�B��B����ˮ�ⷴӦ���ữ�õ�C��AΪ ��BΪ
��BΪ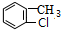 ��E���������Ʒ�Ӧ���ữ�õ�FΪ
��E���������Ʒ�Ӧ���ữ�õ�FΪ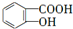 ������Ϣ��b������Ͽ��᳦��ҩ����Ч�ɷֵĽṹ��֪��F��Ũ���ᡢŨ������������·�����Ӧ����GΪ
������Ϣ��b������Ͽ��᳦��ҩ����Ч�ɷֵĽṹ��֪��F��Ũ���ᡢŨ������������·�����Ӧ����GΪ ��G������ԭ��Ӧ�õ����᳦��ҩ����Ч�ɷ֣�
��G������ԭ��Ӧ�õ����᳦��ҩ����Ч�ɷ֣�
��1��������������֪����A�Ľṹ��ʽ�� ���ٵķ�Ӧ������FeCl3���������ڵķ�Ӧ������ȡ����Ӧ��
���ٵķ�Ӧ������FeCl3���������ڵķ�Ӧ������ȡ����Ӧ��
�ʴ�Ϊ�� ��FeCl3��������ȡ����Ӧ��
��FeCl3��������ȡ����Ӧ��
��2����A���Ȼ���������ˮ����֮������γ��������ˮ���Աȱ��Ӻã���A��ȷ��
B�����ܷ�����ȥ��Ӧ����B����
C�����ǻ��Ķ�Ϊ��������λֻ��1��Hԭ�ӣ���1mol������������1mol�巢����Ӧ����C����
D�������Ȼ��������������������м��ԣ���D��ȷ��
�ʴ�Ϊ��AD��
��E������NaOH��Һ��Ӧ�Ļ�ѧ����ʽ�ǣ� ��
��
�ʴ�Ϊ�� ��
��
��3����������������E������ͬ���칹�壺a����E������ͬ�Ĺ����ţ������Ȼ���������b�������ϵ�һ����ȡ�����������֣�����2����ͬ��ȡ�����Ҵ��ڶ�λ��c���˴Ź��������Ϲ���4���壬��������4��Hԭ�ӣ����������Ľṹ��ʽΪ��

�ʴ�Ϊ��

��4���ױ���Ũ���ᷢ��ȡ����Ӧ�����������ױ����������ױ������Ը��������Һ�������������������ᣬ�������������Fe��HCl��Ӧ�����ڰ��������ᣬ��ϳ�·��Ϊ �������м����Ľṹ��ʽΪ
�������м����Ľṹ��ʽΪ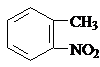
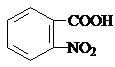
�ʴ�Ϊ��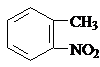
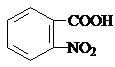
���� ���⿼���л����ƶ���ϳɣ��������C�ķ���ʽ��E�뿹�᳦��ҩ����Ч�ɷֵĽṹ�����ƶϣ���Ҫѧ���Ը������Ϣ�������ã��ϺõĿ���ѧ����ѧ�����������������Ѷ��еȣ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | 1��10-5mol/L | B�� | 1��10-12mol/L | C�� | 1��10-6mol/L | D�� | 1��10-7mol/L |
| A�� | KCl��aq����HCl��aq�� | B�� | HNO3��aq����KNO3��aq�� | C�� | HCl��aq����BaCl2��aq�� | D�� | NaNO3��aq����HNO3��aq�� |
| �����ᣨHCN�� | ̼�ᣨH2C03�� | ����ᣨHF�� |
| K=4.9��10-10 | K1=4.4��10-7 K2=4.7��10-11 | K=6.61��10-4 |
| A�� | �軯����Һ��ͨ������CO2��2CN-+H2O+CO2�T2HCN+CO32- | |
| B�� | NaCN��HCN�Ļ����Һ��һ���У�c��Na+����c��CN-����c��HCN����c��H+����c��OH+�� | |
| C�� | 0.2 mol•L-1 HCN��Һ��0.1mol•L-1NaOH��Һ�������Ϻ���Һ�и������ӵ�Ũ�ȹ�ϵ�У�2[c��H+��-c��OH+��]=c��CN+��-c��HCN�� | |
| D�� | ���������Ũ�ȵ�NaCN��NaF��Һ��������������ǰ�ߴ��ں��� |
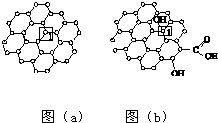 ʯīϩ[��ͼ��a����ʾ]��һ���ɵ���̼ԭ�ӹ��ɵ�ƽ��ṹ����̼���ϣ�ʯīϩ�в���̼ԭ�ӱ���������ƽ��ṹ�ᷢ���ı䣬ת��Ϊ����ʯīϩ[��ͼ��b����ʾ]��
ʯīϩ[��ͼ��a����ʾ]��һ���ɵ���̼ԭ�ӹ��ɵ�ƽ��ṹ����̼���ϣ�ʯīϩ�в���̼ԭ�ӱ���������ƽ��ṹ�ᷢ���ı䣬ת��Ϊ����ʯīϩ[��ͼ��b����ʾ]��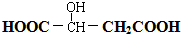 �� ��ش�
�� ��ش� ��
��