��Ŀ����
3������ʯ��������õ�����[KAl��SO4��2•12H2O]���������Ʊ�Al��K2SO4��H2SO4�Ĺ��չ�����ͼ��ʾ��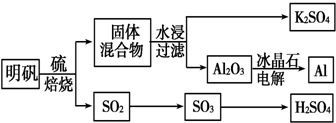
���������Ļ�ѧ����ʽΪ��4KAl��SO4��2•12H2O+3S2K2SO4+2Al2O3+9SO2��+48H2O
��ش��������⣺
��1���ڱ��������ķ�Ӧ�У���ԭ����S����
��2����ˮ�������Һ�еõ�K2SO4����ķ����������ᾧ��
��3����Al��NiO��OH��Ϊ�缫��NaOH��ҺΪ���Һ���һ�����͵�أ��ŵ�ʱNiO��OH��ת��ΪNi��OH��2���õ�ط�Ӧ�Ļ�ѧ����ʽ��Al+3NiO��OH��+NaOH+H2O�TNaAlO2+3Ni��OH��2��
���� ���������շ���4KAl��SO4��2•12H2O+3S=2K2SO4+2Al2O3+9SO2��+48H2O�������������������������������������������������������������������98.3%��Ũ�������գ�SO3+H2O=H2SO4���Ƶ�����������ù��������ˮ����Ϊ��߽����ʣ��ɲ�ȡ�������������Ͻ��裬��ˮ�������Һ�������ᾧ�õ�K2SO4���壬����۵��Al2O3���Ƶ�Al���Դ˽����⣮
��� �⣺���������շ���4KAl��SO4��2•12H2O+3S=2K2SO4+2Al2O3+9SO2��+48H2O�������������������������������������������������������������������98.3%��Ũ�������գ�SO3+H2O=H2SO4���Ƶ�����������ù��������ˮ����Ϊ��߽����ʣ��ɲ�ȡ�������������Ͻ��裬��ˮ�������Һ�������ᾧ�õ�K2SO4���壬����۵��Al2O3���Ƶ�Al��
��1�����������շ���4KAl��SO4��2•12H2O+3S=2K2SO4+2Al2O3+9SO2��+48H2O����Ӧ��SS���������ɶ������ʻ�ԭ��ΪS���ʴ�Ϊ��S����
��2������Һ�л�þ��壬��Ҫ��������Ũ������ȴ�ᾧ�����˵Ȳ������ʴ�Ϊ�������ᾧ��
��3���ŵ�ʱNiO��OH��ת��ΪNi��OH��2��ͬʱ��������������Һ�ʼ��ԣ�������ƫ�����ƣ���Ӧ���ܷ���ʽΪAl+3NiO��OH��+NaOH+H2O�TNaAlO2+3Ni��OH��2��
�ʴ�Ϊ��Al+3NiO��OH��+NaOH+H2O�TNaAlO2+3Ni��OH��2��
���� ���⿼�����ʵ��Ʊ���Ϊ�߿��������ͣ���Ŀ�Ѷ��еȣ���������������ѧ�������������������ѧ��������û���֪ʶ���ʵ�������������ʵ��������

| A�� | ��3��Ԫ��������� | |
| B�� | ÿһ�ж��зǽ���Ԫ�� | |
| C�� | ���ϵ��µ�1��Ԫ�صĵ����۵������ߣ�����17��Ԫ�صĵ����۵����� | |
| D�� | ֻ�е�2��Ԫ�ص�ԭ���������2������ |
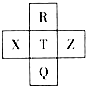 Ԫ��R��X��T��Z��Q��Ԫ�����ڱ��е����λ�����±���ʾ������R�����ڰ�����H2���һ��ϲ�������ը���������ж���ȷ���ǣ�������
Ԫ��R��X��T��Z��Q��Ԫ�����ڱ��е����λ�����±���ʾ������R�����ڰ�����H2���һ��ϲ�������ը���������ж���ȷ���ǣ�������| A�� | �ǽ����ԣ�Z��T��X | B�� | R��Q�ĵ��������26 | ||
| C�� | ��̬�⻯���ȶ��ԣ�R��T��Q | D�� | ����������ˮ��������ԣ�R��Q |
��1���������ͷۡ��ӵ��Ρ�ζ����ijЩʳƷ����������Ҫ�ɷֶ����н���Ԫ��A�����ӣ�A���ӵĽṹʾ��ͼΪ
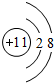 ��
����2��д��E2J4��ˮ��Ӧ�Ļ�ѧ����ʽCH2=CH2+H2O$\stackrel{һ������}{��}$CH3CH2OH���䷴Ӧ����Ϊ�ӳɷ�Ӧ��
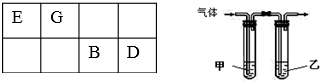
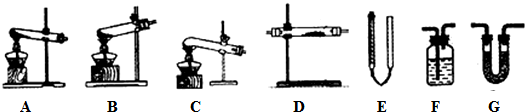
��3��������ͼװ�����һ��ʵ��֤��Ԫ�طǽ�����D��B������Ӧʢ��ij��������ʵ���Һ�������ʵĻ�ѧʽΪH2S����ͨ������������ijǿ�������Һ��Ӧ�����ӷ���ʽΪCl2+2OH-=Cl-+ClO-+H2O��
��4��Ԫ��D�����������Ϊ��ɫҺ�壬0.20mol��������һ����ˮ��ϵõ�һ��ϡ��Һ�����ų�QkJ���������÷�Ӧ���Ȼ�ѧ����ʽΪCl2O7��l��+H2O��l��=HClO4��aq����H=-QkJ/mol��
��5��G������⻯������ˮ����ҺX��D���⻯������ˮ����ҺY�������£���X��Y�������ϣ�������Һ��Ũ�Ⱥͻ�Ϻ�������Һ��pH�����±���
| ʵ���� | X���ʵ���Ũ�ȣ�mol•L-1�� | Y���ʵ���Ũ�ȣ�mol•L-1�� | �����Һ��pH |
| �� | 0.1 | 0.1 | pH=5 |
| �� | 0.2 | 0.1 | pH��7 |
��������Һ�и�����Ũ���ɴ�С��˳����c��NH4+����c��Cl-����c��OH-����c��H+����
| A�� | X��������Y������ | |
| B�� | ����һ��ʱ���KOH�����ʵ������� | |
| C�� | ��ع���ʱ����һ��������Һ��pH��С | |
| D�� | ���ڱ�״����ͨ��5.6 L O2��ȫ��Ӧ������1.0mol���ӷ���ת�� |
 ��=4����
��=4����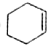 ��=2����
��=2���� ��=4��
��=4��