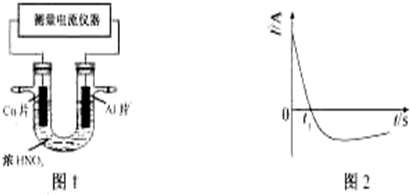
��Ŀ����
8��C��SiԪ���ڻ�ѧ��ռ�м�����Ҫ�ĵ�λ����1��д��Si�Ļ�̬ԭ�Ӻ�������Ų�ʽ1s22s22p63s23p2���ӵ縺�ԽǶȷ�����C��Si��OԪ�صķǽ�����������ǿ������˳��ΪO��C��Si��
��2��̼�����ɵ������ʯī��������ɣ���ṹ������ʯī���壬ÿ��̼ԭ��ͨ��SP2 �ӻ�����Χ̼ԭ�ӳɼ������̼���ܵIJ����֮�俿���Ӽ����������»����������һ��
��3��SiC�ľ���ṹ�뾧�������ƣ�����Cԭ�ӵ��ӻ���ʽΪsp3��������ڵ��������ǹ��ۼ���
��4��������MO�ĵ���������SiC����ȣ���MΪMg����Ԫ�ط��ţ���MO�����������²��ϣ��侧��ṹ��NaCl�������ƣ�MO���۵��CaO�ĸߣ���ԭ����Mg2+�뾶��Ca2+С��MgO�����ܴ�
���� ��1��Siԭ�Ӻ��14�����ӣ������������ԭ����д�����Ų�ʽ��Ԫ�����ڱ�������Ԫ��Խ���ҡ�Խ���ϵ縺��Խ��F�ĵ縺�����Ϊ4.0����
��2��ʯī�Dz�״�ṹ�ģ�ÿ��ÿ��Cԭ������Χ������̼ԭ�ӳɦҼ�������Ϊ120�㣨��Щ����ʵ�����ݵó��ģ������ݼ۲���ӶԻ������۷��������֮����ڷ��Ӽ���������
��3���������һ����ԭ����Χ��4����ԭ������������������ṹ�������ӻ���ʽ��sp3���ǽ���ԭ��֮���γɵĻ�ѧ��Ϊ���ۼ���
��4�����ݵ��������ж�M��ԭ������������ȷ��Ԫ�ط��ţ���Ӱ�����Ӿ����۷е�ߵ͵����ط��������۷е�ĸߵͣ�
��� �⣺��1��Si��14��Ԫ�أ�Siԭ�Ӻ��14�����ӣ������������ԭ�����������������͵�1s��������������������������ϸߵĹ����������Ų�ʽΪ��1s22s22p63s23p2���ӵ縺�ԵĽǶȷ�����O��Cλ��ͬһ���ڣ��ǽ�����Oǿ��C��C��SiΪ��ͬһ���壬C�ķǽ�����ǿ��Si������ǿ����ΪO��C��Si��
�ʴ�Ϊ��1s22s22p63s23p2�� O��C��Si��
��2��ʯī�Dz�״�ṹ��ÿ����ÿ��Cԭ������Χ��3��Cԭ�ӳɦҼ����Һ���һ���м�������Cԭ���ӻ���ʽΪsp2������Ϊ120�㣬���֮����ڷ��Ӽ����������ʴ�Ϊ��sp2�����Ӽ����������»�������
��3����Ϊ�������һ����ԭ����Χ��4����ԭ������������������ṹ������SiC��Si��Cԭ�Ӿ��γ��ĸ�����������Ϊsp3�ӻ����ǽ���ԭ��֮���γɵĻ�ѧ��ȫ���ǹ��ۼ���
�ʴ�Ϊ��sp3�����ۼ���
��4��MO��SiC�ĵ���������ȣ��ʺ��еĵ�����Ϊ20����M����12�����ӣ���Mg�������������������������ɳ����ȣ������Ӱ뾶�ɷ��ȣ�MgO��CaO�����ӵ������ͬ��Mg2+�뾶��Ca2+С��MgO�����ܴ��۵�ߣ�
�ʴ�Ϊ��Mg��Mg2+�뾶��Ca2+С��MgO�����ܴ�
���� ���⿼�����ʽṹ�����ʣ��漰�����Ų�ʽ����д���縺�ԵĴ�С�Ƚϡ��ӻ�������͵��ж��Լ������۵�ıȽϣ���Ŀ��Ϊ�ۺϣ���ֿ�����ѧ���ķ������������������Ӧ��������

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ������̼�Ľṹʽ��O=C=O | B�� | �����ӵĽṹʾ��ͼ��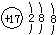 | ||
| C�� | �����ӵĵ���ʽ��Ca2+ | D�� | �廯����ӵĵ���ʽ��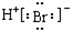 |
| A�� | ��״���£�1mol H2O�����Ϊ22.4L | |
| B�� | 1mol•L-1���Ȼ�����Һ��ָ����Һ�к���1mol NaCl | |
| C�� | ��1L 1mol•L-1��NaCl��Һ��ȡ��100mL��Һ�������ʵ���Ũ�ȱ�Ϊ0.1mol•L-1 | |
| D�� | ��g•mol-1Ϊ��λʱ������ֵ��Ħ����������Է������������ԭ��������� |
| A�� | 4 �� | B�� | 3 �� | C�� | 2 �� | D�� | 1 �� |
| A�� | 11.2L | B�� | 5.6L | C�� | 4.48L | D�� | 2.24L |
| A�� | Cu��OH��2��H2SO4��Cu��OH��2��CH3COOH | B�� | Ba��OH��2��Na2SO4��Ba��OH��2��CuSO4 | ||
| C�� | NaOH��H2SO4��NaOH��NaHSO4 | D�� | Na2CO3�����NaHCO3������ |