��Ŀ����
�±���Ԫ�����ڱ���һ���֣��������е���ĸ�ֱ����һ�ֻ�ѧԪ�أ�
�Իش��������⣺
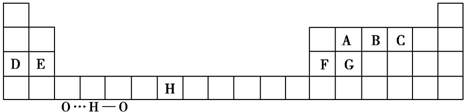
��1��B��D��F��G�ĵ�һ��������С�����˳��Ϊ ����Ԫ�ط��ű�ʾ����
��2����д��Ԫ��H�Ļ�̬ԭ�ӵ����Ų�ʽ ��
Ԫ��B�Ļ�̬ԭ����Χ�����Ų�ͼ ��
��3��A������⻯�����������ԭ�Ӳ�ȡ ��ʽ�ӻ���A�������������ӵĿռ乹�������� ��
��4��Ԫ��C��Ԫ��G�γɵľ��������ľ�������Ϊ ���壬�ڸþ�����ԭ�Ӽ��γɵĹ��ۼ����� ��������ѡ����ѡ����ȷ���ͣ���
A���Ҽ�����B���м�����C�����ЦҼ����֦м� D�����Լ� E���Ǽ��Լ�
��5��A��B��C����Ԫ���⻯����ȶ�����ǿ������˳���û�ѧʽ��ʾ�� ��
B���⻯���VSEPRģ���� ����ʾC��Һ̬�⻯���д��ڵ���� ��

�Իش��������⣺
��1��B��D��F��G�ĵ�һ��������С�����˳��Ϊ
��2����д��Ԫ��H�Ļ�̬ԭ�ӵ����Ų�ʽ
Ԫ��B�Ļ�̬ԭ����Χ�����Ų�ͼ
��3��A������⻯�����������ԭ�Ӳ�ȡ
��4��Ԫ��C��Ԫ��G�γɵľ��������ľ�������Ϊ
A���Ҽ�����B���м�����C�����ЦҼ����֦м� D�����Լ� E���Ǽ��Լ�
��5��A��B��C����Ԫ���⻯����ȶ�����ǿ������˳���û�ѧʽ��ʾ��
B���⻯���VSEPRģ����
���㣺Ԫ�������ɺ�Ԫ�����ڱ����ۺ�Ӧ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
��������Ԫ�������ڱ��е�λ�ÿ�֪��AΪCԪ�أ�BΪNԪ�أ�CΪOԪ�أ�DΪNaԪ�أ�EΪMgԪ�أ�FΪAlԪ�أ�GΪSiԪ�أ�HΪMnԪ�أ�
��1��ͬ�����������Ԫ�صĵ�һ�����ܳ��������ƣ�����A�������Ϊns�ܼ�����2�����ӣ�Ϊȫ��ȷ��״̬�������ϵͣ���һ�����ܸ���ͬ��������Ԫ�أ���A��np�ܼ�����3�ĵ��ӣ�Ϊ�����ȶ�״̬�������ϵͣ���һ������Ҳ����ͬ��������Ԫ�أ�ͬ�������϶��µ����ܽ��ͣ�
��2��Mn�ĺ�����25�����ӣ����ݵ����Ų�������д��N�Ļ�̬ԭ����Χ������Ϊ2s22p3����Ϻ�������Ų����ɻ���������ʾʽ��
��3����������ԭ��Cԭ�Ӽ۲���Ӷ������µ��Ӷ�����ȷ���ӻ���ʽ��A��������������Ϊ������̼������ֱ���η��ӣ�
��4��Ԫ��C��Ԫ��G�γɵĻ�����Ϊ�������裬���ݾ����еĹ������жϣ�����������ֻ���ڹ��۵�����
��5��Ԫ�صķǽ�����Խǿ�����⻯��Խ�ȶ�������NH3�����м۲���Ӷ��жϣ�ˮ�з��Ӽ���������
��1��ͬ�����������Ԫ�صĵ�һ�����ܳ��������ƣ�����A�������Ϊns�ܼ�����2�����ӣ�Ϊȫ��ȷ��״̬�������ϵͣ���һ�����ܸ���ͬ��������Ԫ�أ���A��np�ܼ�����3�ĵ��ӣ�Ϊ�����ȶ�״̬�������ϵͣ���һ������Ҳ����ͬ��������Ԫ�أ�ͬ�������϶��µ����ܽ��ͣ�
��2��Mn�ĺ�����25�����ӣ����ݵ����Ų�������д��N�Ļ�̬ԭ����Χ������Ϊ2s22p3����Ϻ�������Ų����ɻ���������ʾʽ��
��3����������ԭ��Cԭ�Ӽ۲���Ӷ������µ��Ӷ�����ȷ���ӻ���ʽ��A��������������Ϊ������̼������ֱ���η��ӣ�
��4��Ԫ��C��Ԫ��G�γɵĻ�����Ϊ�������裬���ݾ����еĹ������жϣ�����������ֻ���ڹ��۵�����
��5��Ԫ�صķǽ�����Խǿ�����⻯��Խ�ȶ�������NH3�����м۲���Ӷ��жϣ�ˮ�з��Ӽ���������
���
�⣺��Ԫ�������ڱ��е�λ�ÿ�֪��AΪCԪ�أ�BΪNԪ�أ�CΪOԪ�أ�DΪNaԪ�أ�EΪMgԪ�أ�FΪAlԪ�أ�GΪSiԪ�أ�HΪMnԪ�أ�
��1��ͬ�����������Ԫ�صĵ�һ�����ܳ��������ƣ�����A�������Ϊns�ܼ�����2�����ӣ�Ϊȫ��ȷ��״̬�������ϵͣ���һ�����ܸ���ͬ��������Ԫ�أ���A��np�ܼ�����3�ĵ��ӣ�Ϊ�����ȶ�״̬�������ϵͣ���һ������Ҳ����ͬ��������Ԫ�أ�ͬ�������϶��µ����ܽ��ͣ���N��Na��Al��Si�ĵ�һ��������С�����˳��ΪNa��Al��Si��N��
�ʴ�Ϊ��Na��Al��Si��N��
��2��Mn�ĺ�����25�����ӣ�������Ų�ʽΪ��1s22s22p63s23p63d54s2��N�Ļ�̬ԭ����Χ������Ϊ2s22p3���������Χ�����Ų�ͼΪ��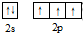 ��
��
�ʴ�Ϊ��1s22s22p63s23p63d54s2��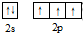 ��
��
��3��A������⻯�����ΪCH4������ԭ��Cԭ�Ӽ۲���Ӷ���Ϊ4��û�йµ��Ӷԣ����ӻ���ʽΪsp3��A��������������Ϊ������̼������ֱ���η��ӣ�
�ʴ�Ϊ��sp3��ֱ���Σ�
��4��Ԫ��C��Ԫ��G�γɵĻ�����Ϊ�������裬�������辧��������ԭ�Ӻ�ԭ���γɵĿռ���״�ṹ�ľ�������ԭ�Ӿ��壻����������ֻ�����ڹ��۵��������ڦҼ�����ͬ�ǽ���ԭ�Ӽ��γɵļ��Լ�����Si-O��Ϊ���Լ���
�ʴ�Ϊ��ԭ�Ӿ��壻AE��
��5���ǽ����ԣ�O��N��C�����⻯����ȶ��ԣ�H2O��NH3��CH4��NH3�����м۲���Ӷ�=3+1=4��������VSEPRģ��Ϊ�������壻ˮ�з��Ӽ�������O-H��O��
�ʴ�Ϊ��H2O��NH3��CH4���������壻O-H��O��
��1��ͬ�����������Ԫ�صĵ�һ�����ܳ��������ƣ�����A�������Ϊns�ܼ�����2�����ӣ�Ϊȫ��ȷ��״̬�������ϵͣ���һ�����ܸ���ͬ��������Ԫ�أ���A��np�ܼ�����3�ĵ��ӣ�Ϊ�����ȶ�״̬�������ϵͣ���һ������Ҳ����ͬ��������Ԫ�أ�ͬ�������϶��µ����ܽ��ͣ���N��Na��Al��Si�ĵ�һ��������С�����˳��ΪNa��Al��Si��N��
�ʴ�Ϊ��Na��Al��Si��N��
��2��Mn�ĺ�����25�����ӣ�������Ų�ʽΪ��1s22s22p63s23p63d54s2��N�Ļ�̬ԭ����Χ������Ϊ2s22p3���������Χ�����Ų�ͼΪ��
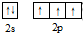 ��
���ʴ�Ϊ��1s22s22p63s23p63d54s2��
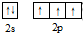 ��
����3��A������⻯�����ΪCH4������ԭ��Cԭ�Ӽ۲���Ӷ���Ϊ4��û�йµ��Ӷԣ����ӻ���ʽΪsp3��A��������������Ϊ������̼������ֱ���η��ӣ�
�ʴ�Ϊ��sp3��ֱ���Σ�
��4��Ԫ��C��Ԫ��G�γɵĻ�����Ϊ�������裬�������辧��������ԭ�Ӻ�ԭ���γɵĿռ���״�ṹ�ľ�������ԭ�Ӿ��壻����������ֻ�����ڹ��۵��������ڦҼ�����ͬ�ǽ���ԭ�Ӽ��γɵļ��Լ�����Si-O��Ϊ���Լ���
�ʴ�Ϊ��ԭ�Ӿ��壻AE��
��5���ǽ����ԣ�O��N��C�����⻯����ȶ��ԣ�H2O��NH3��CH4��NH3�����м۲���Ӷ�=3+1=4��������VSEPRģ��Ϊ�������壻ˮ�з��Ӽ�������O-H��O��
�ʴ�Ϊ��H2O��NH3��CH4���������壻O-H��O��
�����������Ƕ����ʽṹ�Ŀ��飬�漰Ԫ�����ڱ�����������Ų����ӻ���������ӽṹ���縺�ԡ��������͡��⻯���ȶ��ԱȽϵȣ��Ѷ��еȣ�ע�����Ԫ�������ڱ��е�λ�á�Ԫ�ص����ʵȣ�������Ԫ�����ڱ���������֪ʶ���ۺ�Ӧ�õĿ��飮

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
25��ij��̬����������ϳ����ܱ������У����ը���ָֻ���25�棬��ʱ������ѹǿΪ��ʼʱ��һ�룬�پ�����������Һ�����������ڼ���Ϊ��գ������ķ���ʽΪ��������
| A��C2H4 |
| B��C3H6 |
| C��C2H6 |
| D��C5H12 |
�����л�ѧ��Ӧ�У��������Ӽ������Լ����ѣ��������Ӽ������Լ��γɵ��ǣ�������
| A��Na2O+H2O�T2NaOH |
| B��SO2+H2O�TH2SO3 |
| C��Cl2+H2O�THCl+HClO |
| D��4Li+O2�T2Li2O |
���ܱ������з������·�Ӧ��mA��g��+nB��g��?pC��g���ﵽƽ������¶Ȳ��䣬�����������С��ԭ����
�����ﵽ��ƽ��ʱ��C��Ũ��Ϊԭ����1.9��������˵������ȷ���ǣ�������
| 1 |
| 2 |
| A��m+n��p |
| B��ƽ�����淴Ӧ�����ƶ� |
| C��A��ת�������� |
| D��C������������� |
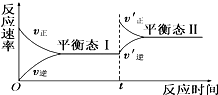 ��ͼ�Ǻ����»�ѧ��Ӧ��A��g��+B��g��?C��g�����ķ�Ӧ�����淴Ӧʱ��仯��ʾ��ͼ������������ʾ��ͼ���ϵ��ǣ�������
��ͼ�Ǻ����»�ѧ��Ӧ��A��g��+B��g��?C��g�����ķ�Ӧ�����淴Ӧʱ��仯��ʾ��ͼ������������ʾ��ͼ���ϵ��ǣ�������| A��ƽ��̬����ƽ��̬�������Ӧ������� |
| B��ƽ���ƶ���ԭ������������˷�Ӧ��Ũ�� |
| C��ƽ���ƶ���ԭ�������������ѹǿ |
| D��ƽ��״̬���ƽ��״̬���ƽ�ⳣ���Ĵ�С��ϵΪK1��K2 |
������������ȷ���ǣ�������
| A����ѧ��Ӧһ�������ʱ仯����һ���������仯 |
| B�����Ϸ�Ӧһ���Ƿ��ȷ�Ӧ |
| C�����ȷ�Ӧ����Ҫ���Ⱦ�һ���ܷ��� |
| D�����ȷ�Ӧ������Ҳ���ܷ��� |