��Ŀ����
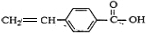
�����廯����Aֻ��������λȡ��������һ������������ͼ��ʾת����ϵ�����У�1molC��Na��ȫ��Ӧ����1mol H2����1molC��NaHCO3��ȫ��Ӧ��Ҳ����1mol���壬E����ʹ������Ȼ�̼��Һ��ɫ��
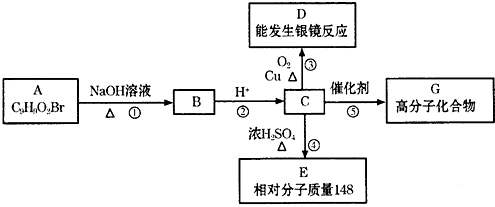
��1��D�����еĺ���������������______��A�Ľṹ��ʽ��______��
��2����Ӧ�ܵĻ���������______��Ӧ����Ӧ�ݵĻ�ѧ����ʽ��______��
��3����Cȡ����λ����ͬ��������FeCl3��Һ��ɫ�����ܷ���ˮ�ⷴӦ��ͬ���칹��ṹ��ʽ�У�
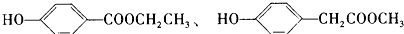
����д��������������������ͬ���칹��Ľṹ��ʽ______��
��4������C��E�Ļ����n mol���ڿ�������ȫȼ������O2______ L����״������������CO2aL����״������H2Obg����C��E������к�E�����ʵ����ļ���ʽΪ��______��

��1��D�����еĺ���������������______��A�Ľṹ��ʽ��______��
��2����Ӧ�ܵĻ���������______��Ӧ����Ӧ�ݵĻ�ѧ����ʽ��______��
��3����Cȡ����λ����ͬ��������FeCl3��Һ��ɫ�����ܷ���ˮ�ⷴӦ��ͬ���칹��ṹ��ʽ�У�
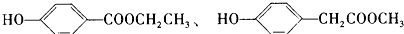
����д��������������������ͬ���칹��Ľṹ��ʽ______��
��4������C��E�Ļ����n mol���ڿ�������ȫȼ������O2______ L����״������������CO2aL����״������H2Obg����C��E������к�E�����ʵ����ļ���ʽΪ��______��
1molC��Na��ȫ��Ӧ����1molH2����1molC��NaHCO3��ȫ��Ӧ��Ҳ����1mol���壬˵��C�к���1��-COOH��1��-OH����λ�ڶ�λλ�ã���C���������ܷ���������Ӧ�����A�Ľṹ�ص�ͷ���ʽ��֪CӦΪ
����AΪ
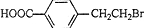
��BΪ
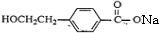
��DΪ
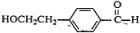
��C��Ũ���������·�����ȥ��Ӧ����E��ӦΪ
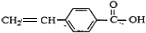
��
��1�������Ϸ��ӿ�֪��DΪ

������ȩ�����Ȼ���A�Ľṹ��ʽ��

��
�ʴ�Ϊ��ȩ�����Ȼ���

��
��2����Ӧ��ΪC����ȥ��Ӧ��CΪ

������-OH��-COOH���ɷ������۷�Ӧ���ɸ߷��ӻ������Ӧ�ķ���ʽΪ
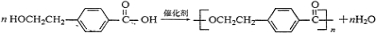
��
�ʴ�Ϊ����ȥ��
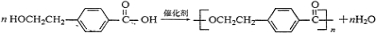
��
��3����Cȡ����λ����ͬ��������FeCl3��Һ��ɫ��˵�����з��ǻ������ܷ���ˮ�ⷴӦ��˵��������������Ӧ��ͬ���칹����

��
�ʴ�Ϊ��

��
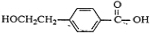
��4��CΪ

������ʽΪC9H10O3��EΪ

��
����ʽΪC9H8O2�������ʵ���ʱ���������������ʵ�����ͬ�������nmol���ڿ�������ȫȼ������O2����9+
-
��
nmol=10nmol��9+
-
��nmol=10nmol�������Ϊ10nmol��22.4L/mol=224n��
���������xmolE����
��x=5n-
��
-
��
�ʴ�Ϊ��224n��5n-
��
-
��

����AΪ

��BΪ

��DΪ

��C��Ũ���������·�����ȥ��Ӧ����E��ӦΪ

��
��1�������Ϸ��ӿ�֪��DΪ

������ȩ�����Ȼ���A�Ľṹ��ʽ��

��
�ʴ�Ϊ��ȩ�����Ȼ���

��
��2����Ӧ��ΪC����ȥ��Ӧ��CΪ

������-OH��-COOH���ɷ������۷�Ӧ���ɸ߷��ӻ������Ӧ�ķ���ʽΪ
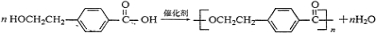
��
�ʴ�Ϊ����ȥ��
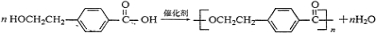
��
��3����Cȡ����λ����ͬ��������FeCl3��Һ��ɫ��˵�����з��ǻ������ܷ���ˮ�ⷴӦ��˵��������������Ӧ��ͬ���칹����

��
�ʴ�Ϊ��

��
��4��CΪ

������ʽΪC9H10O3��EΪ

��
����ʽΪC9H8O2�������ʵ���ʱ���������������ʵ�����ͬ�������nmol���ڿ�������ȫȼ������O2����9+
| 8 |
| 4 |
| 2 |
| 2 |
nmol=10nmol��9+
| 10 |
| 4 |
| 3 |
| 2 |
���������xmolE����
|
| b |
| 18 |
| 5n |
| 22.4��9 |
| b |
| 18 |
�ʴ�Ϊ��224n��5n-
| b |
| 18 |
| 5n |
| 22.4��9 |
| b |
| 18 |

��ϰ��ϵ�д�
 ��Ч���ܿ�ʱ��ҵϵ�д�
��Ч���ܿ�ʱ��ҵϵ�д�
�����Ŀ



 +H2O
+H2O


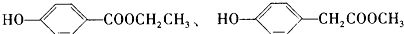 ����д��������������������ͬ���칹��Ľṹ��ʽ
����д��������������������ͬ���칹��Ľṹ��ʽ

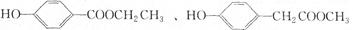 ��___________��___________��(д���ּ���)
��___________��___________��(д���ּ���)
 ��___________��___________��(д���ּ���)
��___________��___________��(д���ּ���)