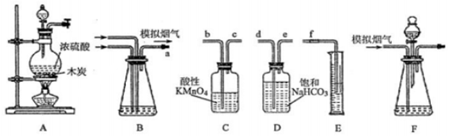
��Ŀ����
14����ͼ��ʾ������ƿ�зֱ�װ�뺬��̪��0.01mol•L-1CH3COONa��Һ�����ֱ������ʢ��ˮ���ձ��У�Ȼ�����ձ����м�����ʯ�ң����ձ����м���NH4NO3���壬�ձ����в����κ����ʣ�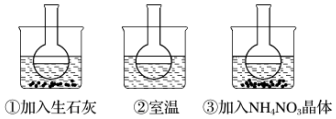
��1������̪��0.01mol•L-1CH3COONa��Һ��dz��ɫ��ԭ��ΪCH3COO-+H2O?CH3COOH+OH-��ʹ��Һ�Լ��ԣ������ӷ���ʽ�ͱ�Ҫ���ֽ��ͣ���
��2����0.01mol•L-1CH3COONa��Һ�зֱ����NaOH���塢Na2CO3���塢FeSO4���壬ʹCH3COO-ˮ��ƽ���ƶ��ķ���ֱ�Ϊ�����ң���������ҡ����ƶ�����
���� ��1�����������ˮ����Һ�ʼ��ԣ���Һ�ʺ�ɫ��
��2�������ˮ���Լ��ԣ�CH3COO-+H2O?CH3COOH+OH-��NaOH������CH3COO-��ˮ�⣬Na2CO3ˮ���Լ��ԣ���CH3COO-��ˮ������ƣ�FeSO4��ˮ�������ԣ���CH3COO-��ˮ����ٽ����ݴ˷�����
��� �⣺��1�����������ˮ����Һ�ʼ��ԣ���Һ�ʺ�ɫ��CH3COO-+H2O?CH3COOH+OH- ʹ��Һ�Լ��ԣ��ʴ�Ϊ��CH3COO-+H2O?CH3COOH+OH-��
��2����0.01mol•L-1CH3COONa��Һ�д���ˮ��ƽ�⣬CH3COO-+H2O?CH3COOH+OH- ʹ��Һ�Լ��ԣ�����NaOH����ƽ�����ơ�����Na2CO3�����ܽ��̼�������ˮ���Լ���ʹˮ��ƽ�����ƣ�FeSO4����Һ�У��������ӻ������������ӣ�ƽ��������У�
�ʴ�Ϊ�������ң�
���� ���⿼��������ˮ���Ӧ�ã�ˮ��ƽ���Ӱ���������ط����жϣ������ܽ���̵������仯����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
18����ľ���к��п����Լ��Σ���Ҫ�ɷ���K2SO4��K2CO3��KCl����ijѧ�������в�����ȡ��ľ���еļ��Σ���ȡ��ľ�Ҽ�ˮ�ܽ⣻�ڹ��ˡ�ȡ��Һ����������Һ������ȴ�ᾧ��
��1���ڢ٢ڢۢܵIJ�������Ҫ�õ����������Ǣ٢ڢۣ�����ţ���
��2��Ϊ�����ľ���е����ӣ�ȡ������������ˮ��������Һ�ֳ��ķݣ��������ʵ�鱨�棨�ں�������д��ص����ݣ���
��3����������õĽ��ۣ�����Ϊ�Ƿ���ȷ������ǡ����������Ӧ����θĽ������Ȼ�����Ϊ���ᱵ��
��1���ڢ٢ڢۢܵIJ�������Ҫ�õ����������Ǣ٢ڢۣ�����ţ���
��2��Ϊ�����ľ���е����ӣ�ȡ������������ˮ��������Һ�ֳ��ķݣ��������ʵ�鱨�棨�ں�������д��ص����ݣ���
| ʵ�鲽�� | ʵ������ | ʵ����� |
��ȡ��һ����Һ������ϡ�����������װ�õ��Թ��У��ѳ����ʯ��ˮ�����ձ��� | ���ܿ������ݣ� | ֤������CO32- |
| ��ȡ�ڶ�����Һ���ȵμ�ϡ������ | ֤������SO42- | |
| ��ȡ��������Һ�������BaCl2��Һ�����ˣ���ȥSO42-��CO32-���ٵμ�AgNO3��Һ��ϡ���� | �а�ɫ�������� | ֤������Cl- |
| ����ȡ���ķ���Һ������ɫ��Ӧ | ����ɫ�ܲ����������ɫ | ֤������K+ |
2�����з�Ӧ�У��������뻹ԭ���ĸ���֮��Ϊ 1��1 ���ǣ�������
| A�� | Cu+2 H2SO4��Ũ�� $\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+SO2��+2H2O | |
| B�� | 2Na+H2SO4�TNa2 SO4+H2�� | |
| C�� | 3S+6NaOH $\frac{\underline{\;\;��\;\;}}{\;}$ 2Na2S+Na2SO3+3H2O | |
| D�� | 2Na+Cl2$\frac{\underline{\;��ȼ\;}}{\;}$ 2NaCl |
3����������ʵ�����������ó��Ľ��۲��������ǣ�������
| ѡ�� | ʵ����� | ���� | ���� |
| A | ���Ҵ����������ظ���أ�K2Cr2O7����Һ�� | ��ɫ��Һ��Ϊ��ɫ | �Ҵ����л�ԭ�� |
| B | ��ͭ�ۼ���1.0mol•L-1Fe2��SO4��3��Һ�� | ��Һ��Ϊ��ɫ | ��������ͭ���� |
| C | ��������þ������NH4Cl��Һ�� | �д������ݲ��� | ��Ӧ����H2��NH3 |
| D | ��NaAlO2��Һ����NaHCO3��Һ�� | �а�ɫ�������� | AlO2-���H+��������HCO3-ǿ |
| A�� | A | B�� | B | C�� | C | D�� | D |
11�����л��������;������ȷ���ǣ�������
| A�� | ����ͨ��ʹ�õĹ��µ���Ҫ�ɷ��Ǿ���Si��̫���ܵ��ʹ�õIJĖ���Ҫ��SiO2 | |
| B�� | ˮ���������������������� | |
| C�� | ���������Һ��SO2��˫��ˮ����ɱ������������ԭ����ͬ����������ǿ������ | |
| D�� | SO2������Ư��ֽ����ë��˿����ñ�衢����ʳƷ�� |
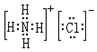 �����д��ڵĻ�ѧ������Ϊ���Ӽ����ۼ���
�����д��ڵĻ�ѧ������Ϊ���Ӽ����ۼ���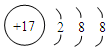 ��Ԫ��D����������ˮ����Ļ�ѧʽΪH3PO4��HPO3��
��Ԫ��D����������ˮ����Ļ�ѧʽΪH3PO4��HPO3��