��Ŀ����
��8�֣���0.050mol SO2��0.030mol O2�����ݻ�Ϊ1L���ܱ�����A�У���0.100mol SO2��0.060mol O2�����ݻ�Ϊ1L���ܱ�����B�У���Ӧ��2SO2(g)��O2(g) ![]() 2SO3(g) ����H����196.6kJ?mol��1�����º����´ﵽƽ��ʱ���������A�зų�3.932 kJ����������
2SO3(g) ����H����196.6kJ?mol��1�����º����´ﵽƽ��ʱ���������A�зų�3.932 kJ����������
��1������A��SO2��ƽ��ת����Ϊ ������B��SO2��ƽ��ת���� ������ڡ�����С�ڡ����ڡ�������A��SO2��ƽ��ת���ʣ�
��2���ڴ��¶��·�Ӧ2SO2(g)��O2(g) ![]() 2SO3(g)��ƽ�ⳣ��K= ��
2SO3(g)��ƽ�ⳣ��K= ��
��3���ﵽƽ���ʱ�� ���A����B���������Ƚ϶̡�
��1��80%�����ڣ���2��1600����3��B��

��ϰ��ϵ�д�
�����Ŀ
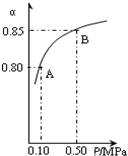 ��������Ҫ�ɷ�ΪFeS2���ǹ�ҵ��ȡ�������Ҫԭ�ϣ������ղ���ΪSO2��Fe2O3��
��������Ҫ�ɷ�ΪFeS2���ǹ�ҵ��ȡ�������Ҫԭ�ϣ������ղ���ΪSO2��Fe2O3��