��Ŀ����
��1��ȡ����FeCl3��Һ�����Թ��У�����NaOH��Һ���к��ɫ�������ɣ���Ӧ�Ļ�ѧ����ʽΪ ���˷�Ӧ���� ���Ӧ���ͣ���
��2����С�ձ��м���20mL����ˮ�����������ں����ˮ�е��뼸��FeCl3��Һ���������ֱ����Һ�� ɫ�������Ƶ�Fe��OH��3���壬���ӷ���ʽΪ ��
��3����֪�����ܾ�ˮ������Ϊ ���÷���ʽ�ͱ�Ҫ������˵����
��4�������θҺ��һ�����Ժ�ǿ�Ļ��������е���������Ϊ���ᣬθҺ�е�����ά����һ��Ũ��������ʵģ����θ���������Ҫ����ҩ���Щҩ������� ��
A��NaOH B��Al��OH��3 C��̼���� D��̼������
��5��ѪҺ���ڷ�ɢϵ�е� ��
��2����С�ձ��м���20mL����ˮ�����������ں����ˮ�е��뼸��FeCl3��Һ���������ֱ����Һ��
��3����֪�����ܾ�ˮ������Ϊ
��4�������θҺ��һ�����Ժ�ǿ�Ļ��������е���������Ϊ���ᣬθҺ�е�����ά����һ��Ũ��������ʵģ����θ���������Ҫ����ҩ���Щҩ�������
A��NaOH B��Al��OH��3 C��̼���� D��̼������
��5��ѪҺ���ڷ�ɢϵ�е�
���㣺����ˮ���Ӧ��,��ѧ����ʽ����д,�������Ҫ����,��ѧ������Ӧ����
ר�⣺
��������1����ѧ����ʽ�ɸ����κͼӦ�����¼�����Σ�ֱ��д�������ڸ��ֽⷴӦ��
��2��Fe��OH��3������ɫΪ���ɫ�������������������Ʊ���д���ӷ���ʽ��
��3��������ˮ��������ˮ��������������������������������ʵ����ã����Ծ�ˮ��
��4��̼���ơ��������Ƽ��Ժ�ǿ����θ�Ĥ��
��5��ѪҺ���ڽ��壻
��2��Fe��OH��3������ɫΪ���ɫ�������������������Ʊ���д���ӷ���ʽ��
��3��������ˮ��������ˮ��������������������������������ʵ����ã����Ծ�ˮ��
��4��̼���ơ��������Ƽ��Ժ�ǿ����θ�Ĥ��
��5��ѪҺ���ڽ��壻
���
�⣺��1���κͼӦ�����¼�����Σ�FeC13��Һ��NaOH��Һ��Ӧ�����ɺ��ɫ����Fe��OH��3��H2O��
��Ӧ�Ļ�ѧ����ʽΪFeC13+3NaOH=Fe��OH��3��+3NaC1���������ںͼ�ķ�Ӧ���ֽⷴӦ��
�ʴ�Ϊ��FeC13+3NaOH=Fe��OH��3��+3NaC1�����ֽⷴӦ��
��2��Fe��OH��3������ɫΪ���ɫ���Ʊ������ӷ���ʽΪ��Fe3++3H2O
Fe��OH��3�����壩+3H+��
�ʴ�Ϊ����֣�Fe3++3H2O
Fe��OH��3�����壩+3H+��
��3��������ˮ��������ˮ�����������������壬Al3++3H2O=Al��OH��3�����壩+3H+�����������������ʵ����ã����Ծ�ˮ��
�ʴ�Ϊ��������ˮ�����������������壬Al3++3H2O=Al��OH��3�����壩+3H+�����������������ʵ����ã����Ծ�ˮ��
��4��̼���ơ��������Ƽ��Ժ�ǿ����θ�Ĥ�����θ���������Ҫ����ҩ��Ϊ����������̼�����ƣ�
�ʴ�Ϊ��BD��
��5��ѪҺ���ڽ����ɢϵ��
�ʴ�Ϊ�����壻
��Ӧ�Ļ�ѧ����ʽΪFeC13+3NaOH=Fe��OH��3��+3NaC1���������ںͼ�ķ�Ӧ���ֽⷴӦ��
�ʴ�Ϊ��FeC13+3NaOH=Fe��OH��3��+3NaC1�����ֽⷴӦ��
��2��Fe��OH��3������ɫΪ���ɫ���Ʊ������ӷ���ʽΪ��Fe3++3H2O
| ||
�ʴ�Ϊ����֣�Fe3++3H2O
| ||
��3��������ˮ��������ˮ�����������������壬Al3++3H2O=Al��OH��3�����壩+3H+�����������������ʵ����ã����Ծ�ˮ��
�ʴ�Ϊ��������ˮ�����������������壬Al3++3H2O=Al��OH��3�����壩+3H+�����������������ʵ����ã����Ծ�ˮ��
��4��̼���ơ��������Ƽ��Ժ�ǿ����θ�Ĥ�����θ���������Ҫ����ҩ��Ϊ����������̼�����ƣ�
�ʴ�Ϊ��BD��
��5��ѪҺ���ڽ����ɢϵ��
�ʴ�Ϊ�����壻
���������⿼���˽����Ʊ���Ӧ�ã��������ʷ����жϣ�����ˮ��ԭ���ǽ���ؼ�����Ŀ�ϼ�

��ϰ��ϵ�д�
�����Ŀ
9.2g������Ͷ�뵽��������ˮ��2H216O���У�������������к��У�������
| A��0.2 mol���� |
| B��0.4 mol���� |
| C��0.2 mol���� |
| D��0.4 mol���� |
�����£����и���������ָ����Һ��һ���ܴ���������ǣ�������
| A��pH=1����Һ�У�Fe2+��NO3-��SO42-��Na+ | ||
| B����ɫ��Һ�У�K+��Fe3+��SO42-��SCN- | ||
C��
| ||
| D����ˮ�����c ��H+��=1��10-14 mol/L����Һ�У�Ca2+��K+��Cl-��HCO3- |
�������������ʵ�Ӧ�û�������ʹ�����ǣ�������
| A��ʳ�ο�����ζ����Ҳ����ʳƷ������ |
| B������ȼ�ջ�ʯȼ�������������������Ҫ����֮һ |
| C��װ�β����ͷŵļ�ȩ����ɿ�����Ⱦ |
| D��ĥ�����Ĵ��������ʣ�������к��ʱ���˰����� |
������������Ȫʵ�飬�������ð�����������
| A������Һ�� | B���ȿ����� |
| C����������ˮ | D���ܸ�ˮ��Ӧ |
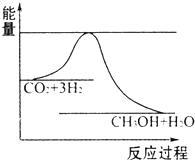
��CH4����ԭNOx���������������������Ⱦ�����磺����˵���д�����ǣ�������
��CH4��g��+4NO2��g��=4NO��g��+CO2��g��+2H2O��g������H=-574kJ/mol
��CH4��g��+4NO��g��=2N2��g��+CO2��g��+2H2O��g������H=-1160kJ/mol��
��CH4��g��+4NO2��g��=4NO��g��+CO2��g��+2H2O��g������H=-574kJ/mol
��CH4��g��+4NO��g��=2N2��g��+CO2��g��+2H2O��g������H=-1160kJ/mol��
| A�������ʵ�����CH4�ڷ�Ӧ�١�����ת�Ƶ�������ͬ |
| B���ɷ�Ӧ�ٿ���֪��CH4��g��+4NO2��g��=4NO��g��+CO2��g��+2H2O��l������H��-574kJ/mol |
| C��4NO2��g��+2N2��g��=8NO��g������H=+586kJ/mol |
| D�����ñ�״����4.48L CH4��NO2��ԭΪN2������������ת�Ƶĵ�������Ϊ1.6NA |
�����Ȼ�����Һ������������������ķ����ǣ�������
| A����ȡ | B������ |
| C������ | D�������ЧӦ |
��NAΪ�����ӵ���������ֵ������˵����ȷ���ǣ�������
| A��1 mol-CH3��1 mol OH-������10 NA������ |
| B�����³�ѹ�£�8gCH4����2NA�����ۼ� |
| C��1L 0.1 mol?L-1�Ĵ�����Һ�к���0.1NA��H+ |
| D����״����22.4 L �Ҵ���̼ԭ����ΪNA |
 ����Ժ�����¼ұ��ڸ籾���������������ϱ�ʾ���й������ڹ�ҵ���������ٷ�չ�Ĺؼ��Σ���Դ�ṹ��úΪ���������ŷŴ����������ѣ�����ʼ�հ�Ӧ������仯��Ϊ��Ҫս���������ڡ���̼ѭ�����Ѿ������˹�������ӣ��Իش��������⣺
����Ժ�����¼ұ��ڸ籾���������������ϱ�ʾ���й������ڹ�ҵ���������ٷ�չ�Ĺؼ��Σ���Դ�ṹ��úΪ���������ŷŴ����������ѣ�����ʼ�հ�Ӧ������仯��Ϊ��Ҫս���������ڡ���̼ѭ�����Ѿ������˹�������ӣ��Իش��������⣺