��Ŀ����
�������˻�Ľ��Ʊ�����Ƕ�Ű�����Ļ�ѧʽ����CaxMgySipO22��OH��2��ʾ��Ҳ����Ca��Mg��Si��H���������ʾ����
��1��ȡ8.10g�����ĩ���������أ����������0.18g��������Ħ������Ϊ ��
��2����ȡ4.05g�����ĩ����1mol/L������100mL�г���ܽ⣬�ò�����2.40g�����ˣ�����Һ��ϴ��Һ�ϲ��������м�����������м���õ�����336mL����״������ͨ������ȷ���������ɻ�ѧʽΪ�������������ʽ��ʾ�� ��
��1��ȡ8.10g�����ĩ���������أ����������0.18g��������Ħ������Ϊ
��2����ȡ4.05g�����ĩ����1mol/L������100mL�г���ܽ⣬�ò�����2.40g�����ˣ�����Һ��ϴ��Һ�ϲ��������м�����������м���õ�����336mL����״������ͨ������ȷ���������ɻ�ѧʽΪ�������������ʽ��ʾ��
���㣺��ѧ����ʽ���йؼ���
ר�⣺������
��������1�����հ����ĩ�������ٵ���ˮ��������ԭ���غ�ð����ˮ�Ĺ�ϵʽ��CaxMgySipO22��OH��2--H2O�����ݶ��ȹ�ϵ��������Ħ��������
��2���������Ƕ������裬���ݹ�ԭ���غ�ð������������Ĺ�ϵʽ��CaxMgySipO22��OH��2--p SiO2�����ݰ�����������趨�ȹ�ϵ����p��
������Һ����CaCl2��MgCl2��FeCl2������Ca��Mg ԭ���غ�ֱ���x��y��ʾ��CaCl2��MgCl2�����ʵ���������H2��������FeCl2�����ʵ���������Clԭ���غ��г����̣������ð�������ԭ�������г���һ���̣�����������⣬ȷ������Ļ�ѧʽ��
��2���������Ƕ������裬���ݹ�ԭ���غ�ð������������Ĺ�ϵʽ��CaxMgySipO22��OH��2--p SiO2�����ݰ�����������趨�ȹ�ϵ����p��
������Һ����CaCl2��MgCl2��FeCl2������Ca��Mg ԭ���غ�ֱ���x��y��ʾ��CaCl2��MgCl2�����ʵ���������H2��������FeCl2�����ʵ���������Clԭ���غ��г����̣������ð�������ԭ�������г���һ���̣�����������⣬ȷ������Ļ�ѧʽ��
���
�⣺��1�������ĩ���պ��������ٵ���ˮ��������ԭ���غ�ð����ˮ�Ĺ�ϵʽ��������Ħ������ΪM����
CaxMgySipO22��OH��2--H2O
1mol 1mol
����1mol��1mol=
��
���M=810g/mol��
�ʴ�Ϊ��810g/mol��
��2��ͨ������֪������������Բ������Ƕ������裬���ݹ�ԭ���غ�ð������������Ĺ�ϵʽ��
CaxMgySipO22��OH��2--PSiO2��
1mol pmol
1mol��pmol=
��
�����p=8
4.05g��������ʵ���=
=0.005mol��
�� FeCl2��H2��ϵʽ����n�� FeCl2��=n��H2��=
=0.015mol��
��Clԭ���غ��У�2��0.005xmol+2��0.005ymol+2��0.015mol=0.1L��lmol/L��
�ɰ�������ԭ������Ϊ810����40x+24y+28��8+16��22+17��2=810��
�������̣���ã�x=2��y=5
�ʰ���Ļ�ѧʽΪ��Ca2Mg2Si8O22��OH��2��
�ʴ�Ϊ��Ca2Mg2Si8O22��OH��2��
CaxMgySipO22��OH��2--H2O
1mol 1mol
| 8.10g |
| M |
| 0.18g |
| 18g/mol |
����1mol��1mol=
| 8.10g |
| M |
| 0.18g |
| 18g/mol |
���M=810g/mol��
�ʴ�Ϊ��810g/mol��
��2��ͨ������֪������������Բ������Ƕ������裬���ݹ�ԭ���غ�ð������������Ĺ�ϵʽ��
CaxMgySipO22��OH��2--PSiO2��
1mol pmol
| 4.05g |
| 810g/mol |
| 2.40g |
| 60g/mol |
1mol��pmol=
| 4.05g |
| 810g/mol |
| 2.40g |
| 60g/mol |
4.05g��������ʵ���=
| 4.05g |
| 810g/mol |
�� FeCl2��H2��ϵʽ����n�� FeCl2��=n��H2��=
| 0.336L |
| 22.4L/mol |
��Clԭ���غ��У�2��0.005xmol+2��0.005ymol+2��0.015mol=0.1L��lmol/L��
�ɰ�������ԭ������Ϊ810����40x+24y+28��8+16��22+17��2=810��
�������̣���ã�x=2��y=5
�ʰ���Ļ�ѧʽΪ��Ca2Mg2Si8O22��OH��2��
�ʴ�Ϊ��Ca2Mg2Si8O22��OH��2��
�����������Թ�����Ϊ���忼��������㣬���巴Ӧ�����DZ���Ĺؼ���ע�������غ㷨����ϵʽ���㣬��ѧϰ��Ҫ���ע�⣮

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
����˵����ȷ���ǣ�������
| A��������к͵ζ�ʵ���п��ô��м��������к͵ζ����� |
| B�����Թ��м���4mL 0.1mol?L-1K2Cr2O7��Һ���ٵμ�����1 mol?L-1NaOH��Һ����Һ��ɫ�ɻ�ɫ��Ϊ��ɫ |
| C�����Ʊ�����ؾ����ʵ���У����ȹ���ʱ���н���Һ��С�ձ����ȼ���2mL��ˮ���Է�����ʱ�ձ��е�����ؾ���������� |
| D������ʱ�¶ȼ�ˮ�������������ƿ֧�ܿڵ��·�λ�ã����ռ����߷е���� |
 ����Һ�������壩X�����루��ͨ�룩��һ������Y��Һ�У������������������X���ʵ����Ĺ�ϵ��ͼ������ͼ�������һ�������ǣ�������
����Һ�������壩X�����루��ͨ�룩��һ������Y��Һ�У������������������X���ʵ����Ĺ�ϵ��ͼ������ͼ�������һ�������ǣ�������
 ��O2��O3�� ��CH3CH��CH3��CH3��CH��CH3��3 ����ʯ��ʯī��
��O2��O3�� ��CH3CH��CH3��CH3��CH��CH3��3 ����ʯ��ʯī��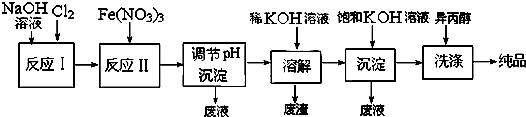
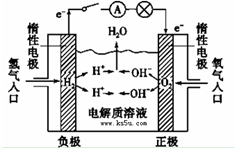 ��֪��1molH2��Ҫ����436kJ��������1molO2��Ҫ����496kJ�������γ�ˮ�����е�1molH-O�ܹ��ͷ�463kJ�����������������������ݼ��㷴Ӧ��
��֪��1molH2��Ҫ����436kJ��������1molO2��Ҫ����496kJ�������γ�ˮ�����е�1molH-O�ܹ��ͷ�463kJ�����������������������ݼ��㷴Ӧ��