��Ŀ����
3�� �١�����Ԫ�����ڱ���ǰ�����ڵ�8��Ԫ�أ������λ����ͼ��ʾ��
�١�����Ԫ�����ڱ���ǰ�����ڵ�8��Ԫ�أ������λ����ͼ��ʾ����1����8��Ԫ���У��ǽ�������ǿ��Ԫ��λ�����ڱ��ڶ����ڢ�A�壻����������ˮ����������ǿ����HClO4���ѧʽ����
��2���ݡ��ޡ�������Ԫ�صļ��⻯�����ȶ���������PH3���ѧʽ�����������ӽṹʾ��ͼΪ
 ��
����3��������γɵĻ������dz����Ĵ�����Ⱦ��֮һ�����âڵļ��⻯���ڴ��������������»�ԭ
Ϊ�����壬�÷�Ӧ�Ļ�ѧ����ʽΪ4NH3+6NO$\frac{\underline{\;����\;}}{��}$5N2+6H2O��8NH3+6NO2$\frac{\underline{\;����\;}}{��}$7N2+12H2O��
��4����˵����ķǽ����ԱȢ�ǿ�Ļ�ѧ����ʽΪH2S+Br2=2HBr+S������Na2S+Br2=2NaBr+S��������1������
��5���ɢ١��ڡ�������Ԫ�ؿ��γ�һ��Ħ������Ϊ84g•mol-1�ķ���X����X������Ԫ�ص�ԭ����Ŀ֮��Ϊ1��1��1����֪X�����и�ԭ�Ӿ��ﵽ8�����ȶ��ṹ���ҷ�������˫��������X���ӵĽṹʽ��N��C-O-O-C��N��
���� ��ͼ��֪�١���Ԫ�طֱ��ǣ���C����N����O����F����P����S����Cl����Br���ɴ˷������
��� �⣺��1����8��Ԫ���У��ǽ�������ǿ��Ԫ����F��λ�����ڱ��ڶ����ڣ��ڢ�A �壻����������ˮ����������ǿ���Ǹ����ᣬ��ѧʽΪ��HClO4���ʴ�Ϊ��������A��HClO4��
��2����P����S����Cl�ǽ������������ף�����������ȶ����������ӵĽṹʾ��ͼΪ ���ʴ�Ϊ��PH3��
���ʴ�Ϊ��PH3�� ��
��
��3���������γɳ�����Ⱦ������һ�������Ͷ����������백����Ӧ�Ļ�ѧ����ʽ�ֱ�Ϊ��4NH3+6NO$\frac{\underline{\;����\;}}{��}$5N2+6H2O��8NH3+6NO2$\frac{\underline{\;����\;}}{��}$7N2+12H2O���ʴ�Ϊ��4NH3+6NO$\frac{\underline{\;����\;}}{��}$5N2+6H2O��8NH3+6NO2$\frac{\underline{\;����\;}}{��}$7N2+12H2O��
��4�����ʼ���û���Ӧ��ȷ��������ǿ����������H2S+Br2=2HBr+S������Na2S+Br2=2NaBr+S������˵����ķǽ�����ǿ���ʴ�Ϊ��H2S+Br2=2HBr+S������Na2S+Br2=2NaBr+S������
��5��X������Ԫ�ص�ԭ����Ŀ֮��Ϊ1��1��1�������ʽΪ����CON��x����x=$\frac{84}{12+16+14}$=2�����Է���ʽΪ����CON��2����ԭ�Ӿ��ﵽ8�����ȶ��ṹ���ҷ�������˫�������ԽṹʽΪ��N��C-O-O-C��N���ʴ�Ϊ��N��C-O-O-C��N��
���� ���⿼����Ԫ�����ڱ���Ԫ�������ɵ��ۺ�Ӧ�ã��漰���ӽṹʾ��ͼ��д����ѧ��Ӧ����ʽ��Ԫ�������ɵ�֪ʶ�㣬ע��O��FԪ��û����ۺ����ᣬΪ�״��㣮

ClO3-��Cl-��MnO4-��Mn2+��Cl2��2Cl-��Fe3+��Fe2+��
����ֱ�����Щ���������� KI���ɵ����� I2�������������ʵ������ٵ��ǣ�������
| A�� | ClO3- | B�� | MnO4- | C�� | Cl2 | D�� | Fe3+ |
| ��� | ʵ������ | ʵ��ԭ�� | ���巢��װ�� |
| �� | �Ʊ����� | H2O2��O2 | �� |
| �� | �Ʊ��������� | Na2SO3��SO2 | �� |
| �� | �Ʊ����� | HCl��Cl2 | ���� |
��2�����ݱ�������ʵ��ԭ��������ͼ1װ����ѡ����ʵ����巢��װ�ã������������ϱ��еĿո��У�

��3��ijѧϰС���������ͼ2ʵ�飬����������ͨ������װ������֤���������ʣ�
��ͨ��������A�е���������Һ����ɫ������ʵ��װ�ô��ڵ�����ȱ������β������װ�ã�
��Cװ���з�����Ӧ�����ӷ���ʽΪ2Fe2++Cl2�T2Fe3++2Cl-��
�۸�С��ͬѧ�����һ��ʵ�飬֤��ϴ��ƿB�е�Na2SO3�����������������Ϊ��ȡһ������B����Һ��һ�ྻ���Թ��У������м��������ữ�����ᱵ��Һ����������ɫ������֤���Ѿ�������������Ϊ�� ����ǡ���������
| �� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 2 | �� | |||||||
| 3 | �� | �� | �� | �� | �� | |||
| 4 | �� | �� |
��1���ڡ��ۡ�������Ԫ�ص�����������Ӧ��ˮ������м�����ǿ����NaOH����д��ѧʽ����
��2���ڡ��ۡ�������Ԫ�ذ�ԭ�Ӱ뾶�ɴ�С��˳������ΪK��Na��Mg����Ԫ�ط��ű�ʾ����
��3��Ԫ�آٵ���̬�⻯��ĵ���ʽ��
 ��д��ʵ��������ȡ������Ļ�ѧ����ʽΪCa��OH��2+2NH4Cl$\frac{\underline{\;\;��\;\;}}{\;}$CaCl2+2NH3��+2H2O��
��д��ʵ��������ȡ������Ļ�ѧ����ʽΪCa��OH��2+2NH4Cl$\frac{\underline{\;\;��\;\;}}{\;}$CaCl2+2NH3��+2H2O����4��Ԫ�آ��ԭ�ӽṹʾ��ͼ��
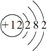 ��
��Ԫ�آݵĵ��ʵĵ����Խ��ڵ���;�Ե��֮�䣬�����һ�����õİ뵼����ϣ�
��5����Ԫ�آڵĵ�����ˮ��Ӧ�����Һ�м���Ԫ�آܵĵ��ʣ�������Ӧ�Ļ�ѧ����ʽΪ2Al+2NaOH+2H2O�T2NaAlO2+3H2����
��6�������ӷ���ʽ֤���ߢ���Ԫ�طǽ����Ե�ǿ��Cl2+2Br���TBr2+2Cl����
| A�� | ԭ�� | B�� | ���� | C�� | ������ | D�� | ������ |

 ��
�� ��
�� ��
�� ��
�� ��д�ṹ��ʽ����
��д�ṹ��ʽ����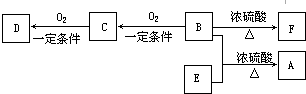
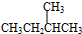 ��
�� G��
G��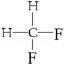 ��
�� 
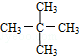
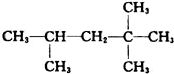 ����������ȼ��Ʒ�ʿ������ܵIJ��������A��ͬ���칹���к���Ч��ԭ���������ٵ�һ�ֽṹ��ʽΪ��CH3C��CH3��2C��CH3��2CH3����A����ϩ����H2ͨ���ӳɷ�Ӧ�õ������ϩ�������п��ܵĽṹ��ʽΪ
����������ȼ��Ʒ�ʿ������ܵIJ��������A��ͬ���칹���к���Ч��ԭ���������ٵ�һ�ֽṹ��ʽΪ��CH3C��CH3��2C��CH3��2CH3����A����ϩ����H2ͨ���ӳɷ�Ӧ�õ������ϩ�������п��ܵĽṹ��ʽΪ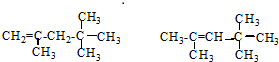 ��
��
 SO3(g)��ƽ�ⳣ��K1=20����ͬһ�¶��£���Ӧ2SO3(g)
SO3(g)��ƽ�ⳣ��K1=20����ͬһ�¶��£���Ӧ2SO3(g)