��Ŀ����
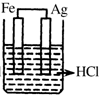
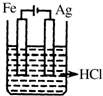
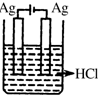
20����ͼ��ʵ�����Ʊ�����������һϵ�����ʵ���װ�ã��г��豸���ԣ���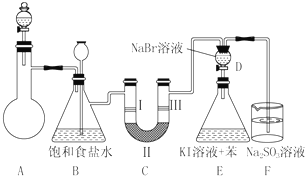
��1���Ʊ�����ѡ�õ�ҩƷΪƯ�۾������Ũ���ᣬ��ط�Ӧ�Ļ�ѧ����ʽΪCa��ClO��2+4HCl��Ũ��=CaCl2+2Cl2��+2H2O��
��2��װ��C��ʵ��Ŀ������֤�����Ƿ����Ư���ԣ�Ϊ��C�Т����η������ʵ������d�����ţ���
| ��� | a | b | c | d |
| �� | �������ɫ���� | �������ɫ���� | ʪ�����ɫ���� | ʪ�����ɫ���� |
| �� | ��ʯ�� | �轺 | Ũ���� | ��ˮ�Ȼ��� |
| �� | ʪ�����ɫ���� | ʪ�����ɫ���� | �������ɫ���� | �������ɫ���� |
��3�����װ��D��E��Ŀ���DZȽ��ȡ��塢�ⵥ�ʵ������ԣ���Ӧһ��ʱ���������װ��D��������Һ����װ��E�У����۲쵽��������E����Һ��Ϊ���㣬�ϲ㣨���㣩Ϊ�Ϻ�ɫ���������ܣ���ܡ����ܡ���˵��Br2��������ǿ��I2��
��4�����������װ��F�пɸ��ù�����NaHSO3��Һ�������ȣ���д����Ӧ��Ӧ�����ӷ���ʽ4HSO3-+Cl2=SO42-+2Cl-+3SO2��+2H2O������Ϊ���ܣ���ܡ����ܡ�������NaHSO3��Һ��
���� ��1�����������Ũ���ᷴӦ�����Ȼ��ơ�������ˮ��
��2��װ��C��ʵ��Ŀ������֤�����Ƿ����Ư���ԣ�ͨ�������ɫ�����Ա���֤����Ϊ�������ֻ��Ϊ���������Ҳ�����������Ӧ��װ��C��D֮��Ӧ����ʢ��Ũ�����װ�ã���ֹD��ˮ��������C�У�
װ��B�ǰ�ȫƿ��C�ж���ʱ��A��Bװ����ѹǿ����B�б���ʳ��ˮ�ᱻѹ�볤��©���У�
��3��������KBr��Ӧ�õ�Br2��������Br2��KI��Ӧ�õ�I2������ȡ��Һ��I2���л������Ϸ����Ϻ�ɫ��D��Һ�к�����������������������ȣ�
��4����������ǿ�����ԣ���������������õ����������ᣬ���ᡢ�����������������Ӧ���ɶ���������Ⱦ������
��� �⣺��1�����������Ũ���ᷴӦ�����Ȼ��ơ�������ˮ����Ӧ����ʽΪ��Ca��ClO��2+4HCl��Ũ��=CaCl2+2Cl2��+2H2O��
�ʴ�Ϊ��Ca��ClO��2+4HCl��Ũ��=CaCl2+2Cl2��+2H2O��
��2��װ��C��ʵ��Ŀ������֤�����Ƿ����Ư���ԣ�ͨ�������ɫ�����Ա���֤����Ϊ�������ֻ��Ϊ���������Ҳ�����������Ӧ��װ��C�Т����η���ʪ�����ɫ�������Ȼ��ƻ�轺���������ɫ��������ѡ��d��
C�ж���ʱ��A��Bװ����ѹǿ������ʳ��ˮ�ᱻѹ�볤��©���У�B������Ϊ����ƿ��Һ���½�������©����Һ��������
�ʴ�Ϊ��d����ƿ��Һ���½�������©����Һ��������
��3��������KBr��Ӧ�õ�Br2��������Br2��KI��Ӧ�õ�I2������ȡ��Һ��I2���л������Ϸ����Ϻ�ɫ��D��Һ�к�����������������������ȣ���������˵��Br2��������ǿ��I2��
�ʴ�Ϊ��E����Һ��Ϊ���㣬�ϲ㣨���㣩Ϊ�Ϻ�ɫ�����䣻
��4����������ǿ�����ԣ���������������õ����������ᣬ���ᡢ�����������������Ӧ���ɶ���������Ⱦ��������Ӧ���ӷ���ʽΪ��4HSO3-+Cl2=SO42-+2Cl-+3SO2��+2H2O��
�ʴ�Ϊ�����ܣ� 4HSO3-+Cl2=SO42-+2Cl-+3SO2��+2H2O��
���� ���⿼��������ʵ�����Ʊ�������ʵ�飬���ؿ���ԭ����װ�õķ��������۵ȣ��ؼ�����ȷʵ��ԭ����ע��ʵ�鷽����ȱ�ݣ�Ӧ��װ��C��D֮��Ӧ����ʢ��Ũ�����װ�ã���ֹD��ˮ��������C�У��Ѷ��еȣ�

 ��ʱ�ƿ�������ϰϵ�д�
��ʱ�ƿ�������ϰϵ�д� һ��һ��һ��ͨϵ�д�
һ��һ��һ��ͨϵ�д� �㽭֮��ѧҵˮƽ����ϵ�д�
�㽭֮��ѧҵˮƽ����ϵ�д�| A�� | ������ԭ��Ӧ�У��ǽ�������һ������ԭ | |
| B�� | ��H2O2��O2�Ĺ���һ�����������������ʵ�� | |
| C�� | �û���Ӧ������������ԭ��Ӧ����Ӧ���еĵ���һ������ԭ�� | |
| D�� | ���������ӱ���ԭ��һ���õ��������� |
| A�� | Na+��H+��Cl-��SO42- | B�� | Na+��H+��Cl-��HCO3- | ||
| C�� | Al3+��K+��OH-��NO3- | D�� | NH4+��OH-��NO3-��Na+ |
| A�� | NH4+ Fe3+ SO42- NO3- | B�� | K+ Na+ CO32- NO3- | ||
| C�� | K+ NH4+ OH- SO42- | D�� | Na+ K+ AlO2- Cl- |

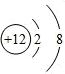 ��A�ĵ���ʽΪ
��A�ĵ���ʽΪ ��
��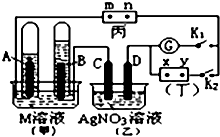 ��ͼ��ʾ��ʵ��װ�ã���Ϊ�õ��۵⻯�غͷ�̪�����Һ��ʪ����ֽ��m��nΪ������ֽ���˵IJ��У���Ϊֱ����Դ��x��yΪ��Դ��������GΪ�����ƣ��缫��Ϊʯī�缫���պ�K2���Ͽ�K1��һ��ʱ���A��B�����������������֮��Ϊ2��1���ش��������⣺
��ͼ��ʾ��ʵ��װ�ã���Ϊ�õ��۵⻯�غͷ�̪�����Һ��ʪ����ֽ��m��nΪ������ֽ���˵IJ��У���Ϊֱ����Դ��x��yΪ��Դ��������GΪ�����ƣ��缫��Ϊʯī�缫���պ�K2���Ͽ�K1��һ��ʱ���A��B�����������������֮��Ϊ2��1���ش��������⣺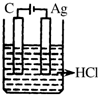
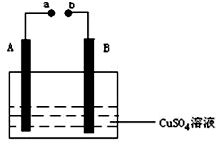 ��ͼ��ʾװ��Ϊ��ֱ����������µ��CuSO4��Һͼ������A��BΪʯī�缫��a��bΪ��Դ������������ͨ��Դ��ͨ��һ��ʱ���B�缫ȡ��ϴ�ɾ�����������������������3.2g����
��ͼ��ʾװ��Ϊ��ֱ����������µ��CuSO4��Һͼ������A��BΪʯī�缫��a��bΪ��Դ������������ͨ��Դ��ͨ��һ��ʱ���B�缫ȡ��ϴ�ɾ�����������������������3.2g����