��Ŀ����
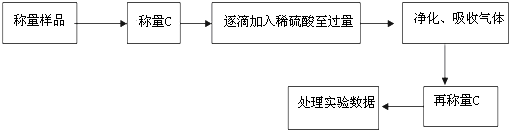
20��ijУ����С��Ϊ�ⶨij̼���ƺ�̼�����ƻ������̼���Ƶ������������ס�������ͬѧ�ֱ�������������ʵ�飮��������ͬѧ����������������ͼ��ʾ��ʵ�����̽���ʵ�飺

��1����ͬѧ��Ϊ�������������ᡱ���òٿأ�Ӧ��Ϊ���������ᡱ�����ڲ����Ҳ�Ӱ��ⶨ��ȷ�ԣ�����Ϊ�Ի�����������������ӷ��������������ڼ��ȹ����лӷ�����������Ӱ������
��2��ʵ�����ʱ�����Ĺ���ɷ����Ȼ��ƣ��ѧʽ��������ÿ�ʼ��Ʒ����Ϊ19g������ʱ��������Ϊ17.55g���������к��е�̼���Ƶ�����Ϊ10.6g��
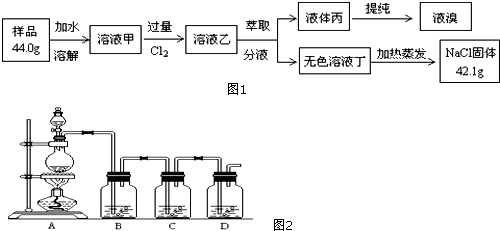
����������ͬѧ����Ҫʵ������ͼ���£�
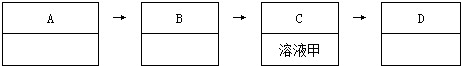
˵��������ͼ�С�����C��ָ���dz���װ��C������ͼ��������������ͼ��ʾװ�ý���ʵ�飺
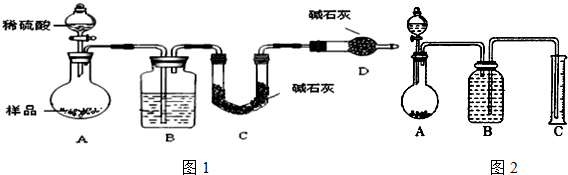
��3��ͼB����װ�Լ�ΪŨ���ᣮ��C��װ��ʯ�������վ���������壮Dװ�õ����������տ����е�ˮ�����Ͷ�����̼����ȷ��ǰһ���������������������ȷ�ԣ�
��4���е�ͬѧ��ΪΪ�˼���ʵ�����ڷ�Ӧǰ��Ӧͨ��N2����Ӧ��ͨ��N2��Ŀ���ǽ�B��Cװ���в���CO2ȫ������Dװ�õļ�ʯ���У���Сʵ����
���������������
��5����һ������Ʒ������ϡ���ᷴӦ������ͼװ�ò�������CO2����������B��Һ��ò���b������ţ�ʹ��������С��
a������̼������Һ b������̼��������Һ
c����������������Һ d����������ͭ��Һ��
���� ������1�����ݲⶨ�������������õ�����Ʒ�����ͷ�Ӧ�������Ȼ��Ƶ��������㣬��������������Ũ��ʱ�ӷ����Բⶨ�����Ӱ�죻
��2��̼���ƺ�̼�����������ᷴӦ�����Ȼ��ƣ���Һ��������Ȼ��ƹ��壬������Ʒ�������������Ȼ��Ƶ������з��������û�����к��е�̼���Ƶ�������
������̼���ƺ�̼��������ϡ���ᷴӦ���ɶ�����̼���ѳ�ȥˮ�����Ķ�����̼����ʯ�����գ�ͨ����ʯ�ҵ�������ö�����̼�����������ݶ�����̼����������Ʒ�������̼���Ƶ����ʵ����������������������
��3��װ��B�������ǰ������е�ˮ������ȥ����C��װ��ʯ�������վ���������壬�ⶨ���������������÷����ؼ���Ҫ��ò�����CO2����������Ӧ��֤�����Ķ�����̼��ȫ�����գ��������ж�����̼���ܱ����գ�װ��D�Ƿ�ֹ�����е��������װ��C��
��4�����Է�Ӧǰ��Ҫͨ��N2����Ӧ��ͨ��N2��Ŀ���ǣ��ž�װ���ڵĿ����������ɵĶ�����̼���������ų�����Cװ���м�ʯ�����գ�
���������������
��5����ѡҺ�岻�ܽ������̼��
��� �⣺������1�����õ�����Ʒ�����ͷ�Ӧ�������Ȼ��Ƶ��������㣬��������������Ũ��ʱ�ӷ����Բⶨ�����Ӱ�죬��Ϊ���������ᡱ�����ڲ����Ҳ�Ӱ��ⶨ��ȷ�ԣ��������ӷ��������������ڼ��ȹ����лӷ�����������Ӱ������
�ʴ�Ϊ���������ӷ��������������ڼ��ȹ����лӷ�����������Ӱ������
��2��̼���ƺ�̼�����������ᷴӦ�����Ȼ��ƣ���Һ��������Ȼ��ƹ��壬����Ʒ��̼���Ƶ����ʵ���Ϊxmol��̼�����Ƶ����ʵ���Ϊymol����$\left\{\begin{array}{l}{106x+84y=19}\\{��2x+y����58.5=17.55}\end{array}\right.$�����x=0.1mol�����Ի�����к��е�̼���Ƶ�����Ϊ0.1mol��106g/mol=10.6g��
�ʴ�Ϊ���Ȼ��ƣ�10.6��
��3��װ��B�������ǰ������е�ˮ������ȥ������Ũ��������ȥˮ�����������е�ˮ�����Ͷ�����̼�ᱻ��ʯ�����գ���D�����������տ����е�ˮ�����Ͷ�����̼����ȷ��ǰһ���������������������ȷ�ԣ�
�ʴ�Ϊ��Ũ������տ����е�ˮ�����Ͷ�����̼����ȷ��ǰһ���������������������ȷ�ԣ�
��4���÷����ؼ���Ҫ��ò�����CO2��������ʵ��ǰ�����ں��п����������к��ж�����̼����Ӱ�����ɵĶ�����̼��������Ӧ��װ���������ں��ж�����̼�����ܱ�C�м�ʯ����ȫ���գ����²ⶨ����нϴ������Է�Ӧǰ��Ҫͨ��N2����Ӧ��ͨ��N2��Ŀ���ǣ������ɵĶ�����̼���������ų�����Cװ���м�ʯ�����գ�
�ʴ�Ϊ����B��Cװ���в���CO2ȫ������Dװ�õļ�ʯ���У���Сʵ����
���������������
��5�����òⶨ��Ӧ���ɵĶ�����̼��������ķ����ⶨ����̼��������Һ�ж�����̼����Ӧ���ܽ�ȼ�С������װ��ͼ�ű���̼��������Һ�ⶨ������̼����������������̼��̼���ơ��������ƶ���Ӧ��������ͭ��Һ��Ҳ�ܽ⣬
�ʴ�Ϊ��b��
���� ���⿼�����������ʵ�̽������ɷ����жϣ���Ҫ��ʵ����̵ķ��������ջ��������Ͳⶨԭ���ǽ���ؼ�����Ŀ�Ѷ��еȣ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | NaHCO3 ��ˮ��Һ��NaHCO3�TNa++H++CO32- | |
| B�� | ����״̬��NaHSO4��NaHSO4�TNa++HSO4- | |
| C�� | HF��ˮ��Һ��HF�TH++F- | |
| D�� | H2S ��ˮ��Һ��H2S?2H++S2- |
| A�� | �����ﺬ��˫�����л���һ����ϩ�� | |
| B�� | �����ﺬ�б������ǻ��Ļ�����һ�����ڷ��� | |
| C�� | ȩ���ͨʽ��CnH2nO��n��1�� | |
| D�� | ��Է�������Ϊ46�����ĺ��������ﲻһ�����Ҵ� |
| ʵ��Ŀ�� | ʵ����� | |
| A | ��ȡ2.0gNaOH���� | ���������ϸ���һ����ֽ��Ȼ��������������2g���룬����������NaOH���� |
| B | �Ʊ�Fe��OH��3���� | ���Ȼ���������Һ����μ�������NaOH��Һ�����������Һ���Ϊ���ɫ |
| C | ֤��̼�������ǿ�ڹ��� | CO2ͨ��Na2SiO3��Һ�У��������ὺ�� |
| D | ��ȡ��ˮ�еĵ� | ����ˮ�����Һ©����Ȼ����ע��ƾ��������÷ֲ���²�Һ����¿ڷų����ϲ�Һ����Ͽڵ��� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ���������c��Na+����c��OH-��=c��CH3COO-����c��H+�� | |
| B�� | �����������c��OH-��-c��H+��=c��Na+��-c��CH3COO-�� | |
| C�� | ������10.0 mLNaOH��Һʱ�У�c��CH3COOH��-c��CH3COO-���T2[c��OH-��-c��H+��] | |
| D�� | ������10.0 mLNaOH��Һʱ�����Һ��pH=4.75����c��CH3COO-����c��CH3COOH����c��Na+����c��H+����c��OH-�� |
| A�� | 22.4LO2���е�O2���Ӹ���ΪNA | |
| B�� | �ڱ�״���£�4.48L H2O��������ԭ������Ϊ0.6NA | |
| C�� | 40 g NaOH�ܽ���1 Lˮ�У����Ƶõ���Һ�����ʵ���Ũ��Ϊ1mol•L-1 | |
| D�� | ���³�ѹ�£�22g CO2���е�ԭ�Ӹ���Ϊ1.5NA |
| A�� | pH=1����Һ�У�K+��Fe2+��Cl-��NO3- | |
| B�� | ����Al�ܷų�H2����Һ�У�NH+4��Cl-��SO2-4��HCO-3 | |
| C�� | �ں��д���Fe3+����Һ�У�NH4+��Cl-��Na+��SCN- | |
| D�� | ��ˮ�����c��OH-��=10-13mol•L-1����Һ�У�Ba2+��Cl-��Na+��Br- |