��Ŀ����
17�� ��2016��11��2�տƼ��ձ��������칬���Ű�������ͨ��7�ι�����ƣ����칬�������Ϸ���Խ���������˶����Ƭ��������Ƭ�����������ͷ��CCD��ͼ���������� �֣����о�ͷ�IJ���Ϊ������CCDΪ�뵼����ϣ�����������ȷ���ǣ�������
��2016��11��2�տƼ��ձ��������칬���Ű�������ͨ��7�ι�����ƣ����칬�������Ϸ���Խ���������˶����Ƭ��������Ƭ�����������ͷ��CCD��ͼ���������� �֣����о�ͷ�IJ���Ϊ������CCDΪ�뵼����ϣ�����������ȷ���ǣ�������| A�� | �����Ͱ뵼����Ͼ�����SiO2�Ʊ� | |
| B�� | ��ͨ�������Ʊ�ԭ���Ǵ�������ʯӢ | |
| C�� | ͨѶ����Ĺ��ά�ǵ����� | |
| D�� | ������������������ʹSiO2ת��ΪSi |
���� A����������Ҫ�ɷ��Ƕ������裬����������̿��Ӧ���ɰ뵼����Ϲ���һ����̼��
B������������ԭ���Ǵ��ʯ��ʯ��ʯӢ��
C�����ά�ijɷ��Ƕ������裻
D�����������еĹ���+4�۱��0�ۣ�Ӧ�ӻ�ԭ����
��� �⣺A����������Ҫ�ɷ��Ƕ������裬����������̿��Ӧ���ɹ���һ����̼�����Բ����Ͱ뵼����Ͼ�����SiO2�Ʊ�����A��ȷ��
B��������ͨ������ԭ���Ǵ��ʯ��ʯ��ʯӢ����B����
C�����ά�ijɷ��Ƕ������裬�����ǵ����裬��C����
D�����������еĹ���+4�۱��0�ۣ�Ӧ�ӻ�ԭ��������������������D����
��ѡA��
���� ���⿼���˹輰�仯��������ʺ���;����ȷ�������衢��������ǽ���ؼ����Ƚ����ף�

��ϰ��ϵ�д�
�����Ŀ
7�����з�Ӧ�����ӷ���ʽ��д����ȷ���ǣ�������
| A�� | ��Ba��OH��2��Һ����μ���NH4HSO4��Һ���պó�����ȫ��Ba2++2OH-+H++SO42-+NH4+=BaSO4��+NH3•H2O+H2O | |
| B�� | ������SO2����ͨ��NaClO��Һ�У�SO2+H2O+ClO-=SO42-+Cl-+2H+ | |
| C�� | Na2CO3��Һ�еμӼ���ϡ���CO32+H+=HCO3- | |
| D�� | NaHCO3��Һ�еμ���������ʯ��ˮ��2HCO3-+Ca2++2OH-=CaCO3��+2H2O+CO32- |
8��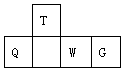 ������Ԫ��T��Q��W��G��Ԫ�����ڱ��е����λ����ͼ��ʾ������Q�����ǽ������ϵ����ǣ������ж���ȷ���ǣ�������
������Ԫ��T��Q��W��G��Ԫ�����ڱ��е����λ����ͼ��ʾ������Q�����ǽ������ϵ����ǣ������ж���ȷ���ǣ�������
 ������Ԫ��T��Q��W��G��Ԫ�����ڱ��е����λ����ͼ��ʾ������Q�����ǽ������ϵ����ǣ������ж���ȷ���ǣ�������
������Ԫ��T��Q��W��G��Ԫ�����ڱ��е����λ����ͼ��ʾ������Q�����ǽ������ϵ����ǣ������ж���ȷ���ǣ�������| A�� | ԭ�Ӱ뾶��W��T | |
| B�� | �����̬�⻯������ȶ��ԣ�Q��T | |
| C�� | ����������Ӧˮ��������ԣ�W��Q | |
| D�� | �����ӵĻ�ԭ�ԣ�W��G |
5���������[Fe2��SO4��3•��NH4��2SO4•6H2O]�㷺����������ˮ�ľ����������Ʊ�������淋�ʵ���У����˳���Ʒ��������ȡĸҺ��pH��1���������м���ָ�����ʣ���Ӧ����Һ����Ҫ���ڵ�һ��������ȷ���ǣ�������
| A�� | �������NaOH��Һ��Na+��NH4+��SO42-��OH- | |
| B�� | �������KI��Һ��K+��Fe2+��NH4+��SO42-��I- | |
| C�� | �������BaCl2��Һ��Ba2+��NH4+��Fe3+��SO42-��Cl- | |
| D�� | �������KSCN��Һ��K+��NH4+��Fe3+��SO42-��SCN- |
12���������ӷ���ʽ����д����ȷ���ǣ�������
| A�� | ����ʯ�����ᷴӦ CaCO3+2H+�TCa2++H2O+CO2�� | |
| B�� | �Ȼ�����Һ�в���ͭƬ��2Fe3++Cu�T2Fe2++Cu2+ | |
| C�� | ̼��������Һ���������Ʒ�Ӧ��OH-+HCO3-�TCO32-+H2O | |
| D�� | ����Ƭ������������Һ�У�Fe+Fe3+�T2Fe2+ |
2����NAΪ�����ӵ�������ֵ�������й�������ȷ���ǣ�������
| A�� | ���³�ѹ�£�5.6L��������ϩ�Ļ�������к���ԭ����ΪNA | |
| B�� | 72gCaO2�����к���������Ϊ3NA | |
| C�� | ��1LO��lmol/LKMnO4������Һ��ͨ������SO2��ת�Ƶ���ЧΪ0.5NA | |
| D�� | 0��lmol������ȫ����ˮ��1L��ˮ����Һ��ClO-��Ŀ��0.1NA |
9������Ũ����ʱ�����ù���Ľ���������Ҫ�ǣ�������


| A�� | þ | B�� | �� | C�� | �� | D�� | �� |
7��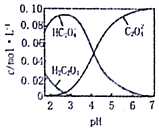 ��֪H2C2O4Ϊ��Ԫ���ᣮ20��ʱ������һ��c��H2C2O4��+c��HC2O4-��+c��C2O42-��=0.100mol•L-1��H2C2O4��NaOH�����Һ����Һ�в����������ʵ���Ũ����PH�ı仯������ͼ��ʾ������ָ����Һ���������ʵ���Ũ�ȹ�ϵһ����ȷ���ǣ�������
��֪H2C2O4Ϊ��Ԫ���ᣮ20��ʱ������һ��c��H2C2O4��+c��HC2O4-��+c��C2O42-��=0.100mol•L-1��H2C2O4��NaOH�����Һ����Һ�в����������ʵ���Ũ����PH�ı仯������ͼ��ʾ������ָ����Һ���������ʵ���Ũ�ȹ�ϵһ����ȷ���ǣ�������
 ��֪H2C2O4Ϊ��Ԫ���ᣮ20��ʱ������һ��c��H2C2O4��+c��HC2O4-��+c��C2O42-��=0.100mol•L-1��H2C2O4��NaOH�����Һ����Һ�в����������ʵ���Ũ����PH�ı仯������ͼ��ʾ������ָ����Һ���������ʵ���Ũ�ȹ�ϵһ����ȷ���ǣ�������
��֪H2C2O4Ϊ��Ԫ���ᣮ20��ʱ������һ��c��H2C2O4��+c��HC2O4-��+c��C2O42-��=0.100mol•L-1��H2C2O4��NaOH�����Һ����Һ�в����������ʵ���Ũ����PH�ı仯������ͼ��ʾ������ָ����Һ���������ʵ���Ũ�ȹ�ϵһ����ȷ���ǣ�������| A�� | PH=2.5����Һ�У�c��H2C2O4��+c��C2O42-����c��HC2O4-�� | |
| B�� | c��HC2O4-��=c��C2O42-������Һ�У�c��Na+����0.100 mol•L-1+c��HC2O4-�� | |
| C�� | c��Na+��=0.100 mol•L-1����Һ�У�c��H+��+c��H2C2O4��=c��OH-��+c��C2O42-�� | |
| D�� | PH=7����Һ�У�c��Na+����2 c��C2O42-�� |