��Ŀ����
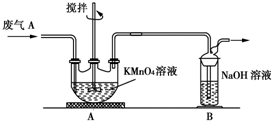 ���Ṥҵ�����ķ���A����Ҫ�ɷ֣�SO2��O2��N2��CO2�ȣ��ŷŵ������л���Ⱦ������ij��ѧ��ȤС��Է���A����ɽ���̽������ش��������⣮
���Ṥҵ�����ķ���A����Ҫ�ɷ֣�SO2��O2��N2��CO2�ȣ��ŷŵ������л���Ⱦ������ij��ѧ��ȤС��Է���A����ɽ���̽������ش��������⣮��1��ͬѧ�����ʵ��������A�к��е�CO2��Ӧѡ����Լ���
A��NaOH��Һ B������KMnO4��Һ C������ʯ��ˮ D������
��2��ͬѧ�����ⶨ����A��SO2�ĺ�����ȡa L����A�����������װ�ý���ʵ�飮
��Ϊ�˱�֤ʵ��ɹ���װ��AӦ���е�������
��ͬѧ����ΪAװ��ǰ������������a L�����к��е�SO2��������Ȼ����к������㣮��С�����ۺ�ͬѧ��������ʣ�����ͬѧ����ʵ�飬��������SO2��ȫ�����գ������ⶨ�Ľ��Ӧ��ƫ��ͬѧ���ƶϵ�������
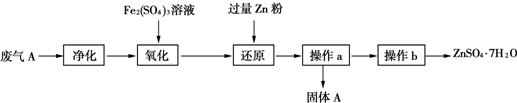
��3����ȤС���ͬѧ�������Ϸ��ֿ��������Ṥҵ����A�Ʊ�𩷯��ZnSO4?7H2O����ʵ��ģ���Ʊ�𩷯��ҵ�������£�
�١��������м���������������Ӧ�����ӷ���ʽ��
�ڼ���Zn�۵�Ŀ����
�۹���A��Ҫ�ɷ���
�ܲ���b�õ�����𩷯���壬��Ҫ�IJ�����
���㣺̽�����ʵ���ɻ�������ʵĺ���,�Ʊ�ʵ�鷽�������
ר�⣺
��������1�����ʵ��������A�к��е�CO2����Ҫ��ȥ������������Ŷ�����̼�ļ��飬�����������������ͨ������ʯ��ˮ���������̼�Ĵ��ڣ�
��2���ٲⶨ����A��SO2�ĺ�����Ϊ�˱�֤ʵ��ɹ���Ҫ��Aװ�����ø��������Һȫ�����գ����������Һ����ɫ֤��������ȫ���������Ҫ�����dz�����ն���������������ʣ�
�ڲⶨ�Ľ��Ӧ��ƫ���������ɵ������ᱻ����������������������������
��3���������������������������������Ϊ���ᣬ����������ԭΪ�����������������п�ۻ�ԭ�������Ӻ������ӣ��õ�����AΪп��������ҺΪ����п��Һ��ͨ������Ũ������ȴ�ᾧ������ϴ�Ӹ���Ȳ���õ�����п���壻
�١��������м���������������Ӧ�Ǻ������ж��������������з�Ӧ�����������������
�ڼ������п�ۻ�ԭ�������Ӻ������ӣ�
�۷�����֪����AΪп������
�ܲ���b����Һ�еõ�����𩷯���壬��ȡ��ȴ�ᾧ������ϴ�Ӹ���Ȳ���õ���
��2���ٲⶨ����A��SO2�ĺ�����Ϊ�˱�֤ʵ��ɹ���Ҫ��Aװ�����ø��������Һȫ�����գ����������Һ����ɫ֤��������ȫ���������Ҫ�����dz�����ն���������������ʣ�
�ڲⶨ�Ľ��Ӧ��ƫ���������ɵ������ᱻ����������������������������
��3���������������������������������Ϊ���ᣬ����������ԭΪ�����������������п�ۻ�ԭ�������Ӻ������ӣ��õ�����AΪп��������ҺΪ����п��Һ��ͨ������Ũ������ȴ�ᾧ������ϴ�Ӹ���Ȳ���õ�����п���壻
�١��������м���������������Ӧ�Ǻ������ж��������������з�Ӧ�����������������
�ڼ������п�ۻ�ԭ�������Ӻ������ӣ�
�۷�����֪����AΪп������
�ܲ���b����Һ�еõ�����𩷯���壬��ȡ��ȴ�ᾧ������ϴ�Ӹ���Ȳ���õ���
���
�⣺��1�����Ṥҵ�����ķ���A����Ҫ�ɷ֣�SO2��O2��N2��CO2�ȣ��ŷŵ������л���Ⱦ���������ʵ��������A�к��е�CO2����Ҫ��ȥ������������Ŷ�����̼�ļ��飬�����������������ͨ������ʯ��ˮ���������̼�Ĵ��ڣ�ʯ��ˮ�����֤�����ж�����̼������A��Ϊ���������Һ��B��Ϊ����ʯ��ˮ��
�ʴ�Ϊ��BC��
��2����Ϊ�˱�֤ʵ��ɹ���Ҫ��Aװ�����ø��������Һȫ�����գ����������Һ����ɫ֤����������������ȫ���������Ҫ�����dz�����ն���������������ʣ�
�ʴ�Ϊ��KMnO4��Һ���Ϻ�ɫ������ȫ��ɫ�����SO2�������ʣ�
�ڰ���ͬѧ����ʵ�飬��������SO2��ȫ�����գ��ⶨ�Ľ��Ӧ��ƫ���������ɵ������ᱻ����������������ʵ������з���2H2SO3+O2=2H2SO4�����²ⶨ������������
�ʴ�Ϊ��2H2SO3+O2=2H2SO4��
��3���������������������������������Ϊ���ᣬ����������ԭΪ�����������������п�ۻ�ԭ�������Ӻ������ӣ��õ�����AΪп��������ҺΪ����п��Һ��ͨ������Ũ������ȴ�ᾧ������ϴ�Ӹ���Ȳ���õ�����п���壻
�١��������м���������������Ӧ�Ǻ������ж��������������з�Ӧ�����������������ᣬ��Ӧ�����ӷ���ʽΪ��2Fe3++SO2+2H2O=2Fe2++SO42-+4H+��
�ʴ�Ϊ��2Fe3++SO2+2H2O=2Fe2++SO42-+4H+��
�ڼ������п�ۻ�ԭ�������Ӻ������ӣ�����Һ�е�Fe2+��Fe3+��
�ʴ�Ϊ������Һ�е�Fe2+��Fe3+�ȣ�
�۷�����֪����AΪп������
�ʴ�Ϊ��Zn��Fe��
�ܲ���b����Һ�еõ�����𩷯���壬��Ҫ�IJ���Ϊ����ȴ�ᾧ������ϴ�ӣ�����Ȳ���õ���
�ʴ�Ϊ��������Ũ���������£��ᾧ����
�ʴ�Ϊ��BC��
��2����Ϊ�˱�֤ʵ��ɹ���Ҫ��Aװ�����ø��������Һȫ�����գ����������Һ����ɫ֤����������������ȫ���������Ҫ�����dz�����ն���������������ʣ�
�ʴ�Ϊ��KMnO4��Һ���Ϻ�ɫ������ȫ��ɫ�����SO2�������ʣ�
�ڰ���ͬѧ����ʵ�飬��������SO2��ȫ�����գ��ⶨ�Ľ��Ӧ��ƫ���������ɵ������ᱻ����������������ʵ������з���2H2SO3+O2=2H2SO4�����²ⶨ������������
�ʴ�Ϊ��2H2SO3+O2=2H2SO4��
��3���������������������������������Ϊ���ᣬ����������ԭΪ�����������������п�ۻ�ԭ�������Ӻ������ӣ��õ�����AΪп��������ҺΪ����п��Һ��ͨ������Ũ������ȴ�ᾧ������ϴ�Ӹ���Ȳ���õ�����п���壻
�١��������м���������������Ӧ�Ǻ������ж��������������з�Ӧ�����������������ᣬ��Ӧ�����ӷ���ʽΪ��2Fe3++SO2+2H2O=2Fe2++SO42-+4H+��
�ʴ�Ϊ��2Fe3++SO2+2H2O=2Fe2++SO42-+4H+��
�ڼ������п�ۻ�ԭ�������Ӻ������ӣ�����Һ�е�Fe2+��Fe3+��
�ʴ�Ϊ������Һ�е�Fe2+��Fe3+�ȣ�
�۷�����֪����AΪп������
�ʴ�Ϊ��Zn��Fe��
�ܲ���b����Һ�еõ�����𩷯���壬��Ҫ�IJ���Ϊ����ȴ�ᾧ������ϴ�ӣ�����Ȳ���õ���
�ʴ�Ϊ��������Ũ���������£��ᾧ����
���������⿼����������ɵ�ʵ��̽���������̷����жϣ���Ҫ���������ʵ�����Ӧ�ã����ջ����ǹؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
��֪����1mol H2�����л�ѧ������ʱ��Ҫ����436kJ����������1mol Cl2�����л�ѧ������ʱ��Ҫ����243kJ������������Hԭ�Ӻ�Clԭ���γ�1mol HCl����ʱҪ�ų�431kJ������������������ȷ���ǣ�������
| A��������������Ӧ�����Ȼ���������Ȼ�ѧ����ʽ��H2��g��+Cl2��g���T2HCl��g�� |
| B��������������Ӧ����2 mol�Ȼ������壬��Ӧ����183 kJ������ |
| C��������������Ӧ����1 mol�Ȼ������壬��Ӧ�ų�183 kJ������ |
| D��������������Ӧ����2 mol�Ȼ������壬��Ӧ�ų�183 kJ������ |
��֪�����Դ�ǿ������˳���ǣ�Cl2��Br2��Fe2+��I2�������л�ѧ����ʽ�������ӷ���ʽ���д�����ǣ�������
| A��Cl2+2FeCl2��2FeCl3 |
| B��Br2+2Fe2+��2Fe3++2Br- |
| C��2FeCl3+2KI��2FeCl2+2KCl+I2 |
| D��I2+2Fe2+��2I-+2Fe3+ |
�������ӷ���ʽ������ȷ���ǣ�������
| A�����ۼ��뵽FeCl3��Һ�У�Fe+Fe3+=2Fe2+ |
| B�������İ�ˮ�μӵ��Ȼ�����Һ��Al3++4OH-�TAlO2-+2H2O |
| C��������ˮ��Ӧ��Cl2+H2O�TCl-+2H++ClO- |
| D����������Һ��ͨ�����CO2��SiO32-+2CO2+2H2O�TH2SiO3��+2HCO3- |
����ʵ��װ��һ�㲻���ڷ������ʵ��ǣ�������
A�� |
B�� |
C�� |
D�� |
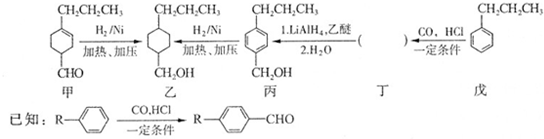
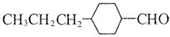 ��
��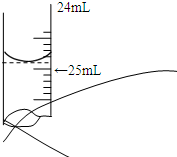 ���ⶨijNaOH��Һ�����ʵ���Ũ�ȣ�����0.1000mol?L-1 HCl����Һ�����к͵ζ����÷�̪��ָʾ��������ش��������⣺
���ⶨijNaOH��Һ�����ʵ���Ũ�ȣ�����0.1000mol?L-1 HCl����Һ�����к͵ζ����÷�̪��ָʾ��������ش��������⣺