��Ŀ����
8����ú��ʯ���п�������������ԭ��A��B��A��һ�ֹ�ʵ����������IJ�����������һ�����ҵ�ʯ�ͻ�����չˮƽ��B��һ�ֱ�ˮ�����״Һ�壬B����̼������Ԫ����ɣ�̼Ԫ������Ԫ�ص�������Ϊ12��1��B����Է�������Ϊ78���ش��������⣺��1��A�Ľṹ��ʽΪH2C=CH2��B�Ŀռ乹��Ϊƽ���������Σ�
��2���ڵ�ˮ�м���B���ú��������Һ�ֲ㣬�ϲ�Ϊ�л����ҳ��Ϻ�ɫ���²������ɫ��
��3��B��ŨH2SO4��ŨHNO3��50-60�淴Ӧ�Ļ�ѧ��Ӧ����ʽ
 ��
����4��B��H2���������������·�Ӧ�Ļ�ѧ����ʽ
 +3H2$��_{��}^{Ni}$
+3H2$��_{��}^{Ni}$ ��
����5����A���ڵ�ͬϵ��Cʹ������Ȼ�̼��Һ��ɫ�Ļ�ѧ��Ӧ����ʽH2C=CHCH3+Br2��BrCH2CHBrCH3��
���� ��ú��ʯ���п�������������ԭ��A��B��A��һ�ֹ�ʵ����������IJ�����������һ�����ҵ�ʯ�ͻ�����չˮƽ����AΪC2H4��B����̼������Ԫ����ɣ�̼Ԫ������Ԫ�ص�������Ϊ12��1���������C��Hԭ����Ŀ֮��Ϊ$\frac{12}{12}$��$\frac{1}{1}$=1��1�����ʽΪCH��B����Է�������Ϊ78���������ʽΪCnHn����13n=78�����n=6����B�ķ���ʽΪC6H6����BΪ ���ݴ˽��
���ݴ˽��
��� �⣺��ú��ʯ���п�������������ԭ��A��B��A��һ�ֹ�ʵ����������IJ�����������һ�����ҵ�ʯ�ͻ�����չˮƽ����AΪC2H4��B����̼������Ԫ����ɣ�̼Ԫ������Ԫ�ص�������Ϊ12��1���������C��Hԭ����Ŀ֮��Ϊ$\frac{12}{12}$��$\frac{1}{1}$=1��1�����ʽΪCH��B����Է�������Ϊ78���������ʽΪCnHn����13n=78�����n=6����B�ķ���ʽΪC6H6����BΪ ��
��
��1��AΪ��ϩ���ṹ��ʽΪH2C=CH2��BΪ ��Ϊƽ���������νṹ��
��Ϊƽ���������νṹ��
�ʴ�Ϊ��H2C=CH2��ƽ���������Σ�
��2���ڵ�ˮ�м���B�� �����ã��������Һ��������ȡ��Һ�еĵ⣬�����ܶ�С��ˮ������Ϊ����Һ�ֲ㣬�ϲ�Ϊ�л����ҳ��Ϻ�ɫ���²������ɫ��
�����ã��������Һ��������ȡ��Һ�еĵ⣬�����ܶ�С��ˮ������Ϊ����Һ�ֲ㣬�ϲ�Ϊ�л����ҳ��Ϻ�ɫ���²������ɫ��
�ʴ�Ϊ����Һ�ֲ㣬�ϲ�Ϊ�л����ҳ��Ϻ�ɫ���²������ɫ��
��3��B�� ����Ũ������Ũ������50��60�淴Ӧ������������ˮ����Ӧ��ѧ��Ӧ����ʽ��
����Ũ������Ũ������50��60�淴Ӧ������������ˮ����Ӧ��ѧ��Ӧ����ʽ�� ��
��
�ʴ�Ϊ�� ��
��
��4������H2���������������·�Ӧ�Ļ�ѧ����ʽ�� +3H2$��_{��}^{Ni}$
+3H2$��_{��}^{Ni}$ ��
��
�ʴ�Ϊ�� +3H2$��_{��}^{Ni}$
+3H2$��_{��}^{Ni}$ ��
��
��5����A��H2C=CH2�����ڵ�ͬϵ��CΪH2C=CHCH3�����巢���ӳɷ�Ӧʹ������Ȼ�̼��Һ��ɫ����ѧ��Ӧ����ʽΪ��H2C=CHCH3+Br2��BrCH2CHBrCH3��
�ʴ�Ϊ��H2C=CHCH3+Br2��BrCH2CHBrCH3��
���� ���⿼���л�����ƶϣ��漰����ϩ�������ʡ��л���Ӧ����ʽ��д����ȡ�ȣ�ע��Ի���֪ʶ���������գ�

| A�� | CH��CH | B�� | CH2=CH2 | C�� | CH2=C��CH3��CH3 | D�� | CH��C-CH3 |
| A�� | 1mol�û�������������3molNaOH��Ӧ | |
| B�� | �ȿ�����Br2��CCl4��Һ�����ӳɷ�Ӧ���ֿ��ڹ�������Br2����ȡ����Ӧ | |
| C�� | �ȿ��Դ����⣬�ֿ���ʹ����KMnO4��Һ��ɫ | |
| D�� | �ȿ�����FeCl3��Һ������ɫ��Ӧ���ֿ���NaHCO3��Һ��Ӧ�ų�CO2���� |
 �������������Ϣ���ش����⣮
�������������Ϣ���ش����⣮| A | ��̬ԭ�������������Ǵ��������� |
| B | ��̬ԭ�Ӻ�����13�ֲ�ͬ�˶�״̬�ĵ��� |
| C | ��Bͬһ���ڣ�ԭ����δ�ɶԵ�������ͬ��������� |
| D | D2-�ĺ�������Ų����ԭ����ͬ |
| E | ��ds��ԭ��������С��Ԫ�� |
��2��A3���ӵĿռ乹��ΪV�Σ����以Ϊ�ȵ�����ķ���ΪSO2��
��3��������ˮ�е��ܽ��C7H15OH���Ҵ��͵�ԭ���ǣ��Ҵ��е��ǻ���ˮ���ǻ��ṹ���������ܽ�ȴ�C7H15OH����������ˮ���ǻ��ṹ���Ƴ̶�С�����ܽ��С��C7H15OH �в���sp3�ӻ���ԭ�ӹ���8����
��4��E��NH3��42+�������д��ڵĻ�ѧ�������Т٢ۣ�����ţ���
����λ�� �ڽ����� �ۼ��Թ��ۼ� �ܷǼ��Թ��ۼ� �����Ӽ� �����
�� E��NH3��42+���жԳƵĿռ乹�ͣ��ҵ� E��NH3��42+�е�����NH3���ӱ�����Cl-ȡ��ʱ���ܵõ����ֲ�ͬ�ṹ�IJ���� E��NH3��42+�Ŀռ乹��Ϊa������ţ���
a��ƽ��������b���������� c�������� d��V��
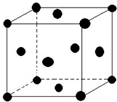
��5������E������ͼ��ʾ����֪EԪ�����ԭ������ΪM��ԭ�Ӱ뾶Ϊr pm���ܶ�Ϊ��g/cm3 ��1pm=10-10cm����ôд������٤������NA�ı���ʽ$\frac{\sqrt{2}��10{\;}^{30}M}{8��r{\;}^{3}}$����M��r���ѱ�ʾ����
��1������Ԫ��ԭ�Ӱ뾶�Ӵ�С��˳��ΪNa��Mg��C��O����Ԫ�ط��ű�ʾ����
��2�����á�����������գ�
| ��һ������ | �縺�� | �⻯��е� | ����������Ӧˮ����ļ��� |
| W��X | Z��Y | Y��Z | W��X |
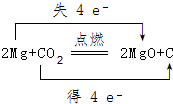 ��
�� | A�� | CH3CH��OH��COOH | B�� | HO��CH2��2CHO | C�� | HOOC-COOH | D�� | CH3CH2COOH |
| A�� | ���̼�����������������Һ | B�� | ���������������������Һ | ||
| C�� | ���Ũ��ˮ����������Һ | D�� | ����������������Һ |
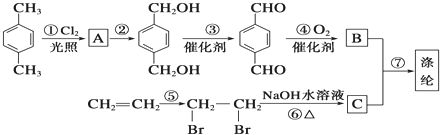
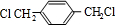 ��B
��B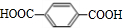 ��CHOCH2CH2OH��
��CHOCH2CH2OH�� ��
��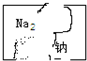 ʵ���ҷ���һƿ��ǩ�������Һ����ͼ��ʾ��ͬѧ���룬����Һ������C��������ĸ��ţ�
ʵ���ҷ���һƿ��ǩ�������Һ����ͼ��ʾ��ͬѧ���룬����Һ������C��������ĸ��ţ�