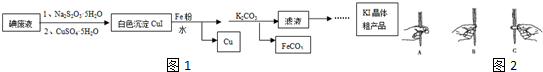
��Ŀ����
9��ijѧ����0.1mol•L-1��KOH����Һ�ζ�δ֪Ũ�ȵ����ᣬ������ֽ�Ϊ���¼�����A����ȡ20mL��������ע��ྻ����ƿ��������2��3�η�̪
B���ñ���Һ��ϴ�ζ���2��3��
C����ʢ�б���Һ�ļ�ʽ�ζ��̶ܹ��ã����ڵζ��ܼ���ʹ֮������Һ
D��ȡ��KOH��Һע���ʽ�ζ�������0���̶�����1��2cm
E������Һ������0����0�����¿̶ȣ����¶���
F������ƿ�µ�һ�Ű�ֽ������ƿ���ڵζ��ܵ����棬�ñ�KOH��Һ�ζ����յ㲢���µζ���Һ��Ŀ̶ȣ�
�ʹ�ʵ�������գ�
��1����ȷ���������˳���ǣ��������ĸ��д��BDCEAF��
��2������B���������Ŀ����ϴȥ�ζ����ڱڸ��ŵ�ˮ����ֹ����Һϡ�Ͷ�������
��3������A�������֮ǰ�����ô���Һ��ϴ��ƿ����Եζ������Ӱ����ʹ���δ֪Һ��Ũ��ƫ��
��4����������ƿ�µ�һ�Ű�ֽ�������DZ��ڹ۲���Һ��ɫ�任��ȷ�жϵζ��յ㣻
��5���жϵ���ζ��յ��ʵ�������ǵ���Һ����ɫ��Ϊdz��ɫ�����ڰ�����ڲ���ɫ��
��6������ȡһ������KOH���壨������NaOH�����Ʊ���Һ�������ζ��������ᣬ��Եζ����������Ӱ����ʹ���δ֪ҺŨ��ƫС��
���� ��1�������к͵ζ��м�©��ϴ�ӡ���ϴ��װҺ�����㡢ȡ����Һ��ָʾ�����ζ��Ȳ������̣�
��2��ֱ��װ�����Һ����Һ��Ũ��ƫ�ͣ�
��3������c�����⣩=$\frac{c����ע����V������}{V�����⣩}$��������������V��������Ӱ�죬�Դ��ж�Ũ�ȵ���
��4����ƿ�µ�һ�Ű�ֽʹ�ζ��յ���ɫ�仯�����ԣ����ڷֱ棻
��5������Һ��ɫ�仯�Ұ�����ڲ���ɫ����˵���ﵽ�ζ��յ㣻
��6������c�����⣩=$\frac{c����ע����V������}{V�����⣩}$��������������V��������Ӱ�죬�Դ��ж�Ũ�ȵ���
��� �⣺��1���к͵ζ��м�©��ϴ�ӡ���ϴ��װҺ�����㡢ȡ����Һ��ָʾ�����ζ��Ȳ�����
�ʴ�Ϊ��BDCEAF��
��2���ζ���������ˮϴ�Ӻ��ڱ���һ��ˮĤ�����ֱ��װҺ��ʹŨ�Ƚ��ͣ����Ա����ñ���Һ��ϴ�ζ���2-3�Σ�
�ʴ�Ϊ��ϴȥ�ζ����ڱڸ��ŵ�ˮ����ֹ����Һϡ�Ͷ�������
��3����ƿ������ˮϴ�Ӻ�������ô���Һ��ϴ����ʹ��ƿ�����ʵ����ʵ����������V������ƫ����c�����⣩=$\frac{c����ע����V������}{V�����⣩}$�������ⶨ���ƫ�ߣ�
�ʴ�Ϊ��ʹ���δ֪Һ��Ũ��ƫ��
��4������ƿ�µ�һ�Ű�ֽʹ�ζ��յ���ɫ�仯�����ԣ����ڷֱ棬
�ʴ�Ϊ�����ڹ۲���Һ��ɫ�任��ȷ�жϵζ��յ㣻
��5����0.1mol•L-1��KOH����Һ�ζ�δ֪Ũ�ȵ����ᣬ�÷�̪��ָʾ���������յ�ʱ�����ǵ���Һ����ɫ��Ϊdz��ɫ�����ڰ�����ڲ���ɫ��
�ʴ�Ϊ������Һ����ɫ��Ϊdz��ɫ�����ڰ�����ڲ���ɫ��
��6��������KOH��������NaOH�����ʹ���ƫС����Ϊͬ������NaOH��KOH�к�����ǿ��������Һ������٣�����ƫС��
�ʴ�Ϊ��ʹ���δ֪ҺŨ��ƫС��
���� ���⿼�����ʵĺ����IJⶨ��Ϊ��Ƶ���㣬���ؿ��黯ѧʵ������е�����������Ŀ�Ѷ��еȣ�����ʵ���ԭ������ȷ���������ǽ���Ĺؼ�������������ѧ�����Ӧ����ѧ֪ʶ��������

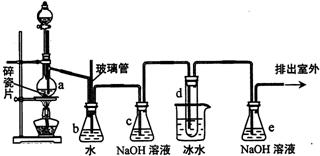
��֪��
CH3CH2OH$��_{170��}^{Ũ����}$CH2�TCH2��+H2O
CH3CH2OH$��_{140��}^{Ũ����}$CH3CH2OCH2CH3+H2O
��������б����£�
| �Ҵ� | 1��2-�������� | ���� | �� | |
| ״̬ | ��ɫҺ�� | ��ɫҺ�� | ��ɫҺ�� | ����ɫҺ�� |
| �ܶȣ�g/mL�� | 0.79 | 2.18 | 0.71 | 3.10 |
| �е�/�� | 78.5 | 131.4 | 34.6 | 58.8 |
| �۵�/�� | -114.3 | 9.79 | -116.2 | -7.2 |
| ˮ���� | ���� | ���� | �� | ���� |
��2����ȫƿb��ʵ�����ж������ã���һ���Լ��ʵ������е���d�Ƿ�����������д����������ʱƿb�е����������е�Һ����������ƿ�е�Һ���½���
��ȫƿb�������������Ƿ�ֹ������
��3������c��e�ж�ʢ��NaOH��Һ��c��NaOH��Һ��������������ϩ�����к��е�CO2��SO2���������壮
��4����ȥ����������δ��Ӧ��Br2�����е���Ҫ����Ϊ���ѣ�Ҫ��һ���ᴿ�����в����б������D��������ȷѡ��ǰ����ĸ����
A���ؽᾧ B������ C����ȡ D������
��5��ʵ����Ҳ���Գ�ȥdװ����ʢ��ˮ���ձ�����Ϊ����ˮֱ�Ӽ��뵽dװ�õ��Թ��ڣ����ʱ��ˮ����������ȴ1��2һ��������������⣬��������������Һ��Br2��1��2-�������飮
| A�� | ��ϩ���Ҷ�����CH2�TCH2$\stackrel{�ӳ�}{��}$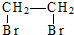 $\stackrel{ȡ��}{��}$ $\stackrel{ȡ��}{��}$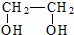 | |
| B�� | ��������Ҵ���CH3CH2Br$\stackrel{��ȥ}{��}$CH2�TCH2$\stackrel{�ӳ�}{��}$CH3CH2OH | |
| C�� | 1-�嶡���1��3-����ϩ��CH3CH2CH2CH2Br$\stackrel{��ȥ}{��}$CH3CH2CH=CH2$\stackrel{�ӳ�}{��}$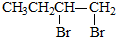 $\stackrel{��ȥ}{��}$CH2�TCH-CH�TCH2 $\stackrel{��ȥ}{��}$CH2�TCH-CH�TCH2 | |
| D�� | ��ϩ����Ȳ��CH2�TCH2$\stackrel{�ӳ�}{��}$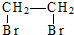 $\stackrel{��ȥ}{��}$CH��CH $\stackrel{��ȥ}{��}$CH��CH |
| ʵ�� | �¶�/�� | ��ʼʱ | ƽ��ʱ | |||
| n��CO��/mol | n��H2S��/mol | n��COS��/mol | n��H2��/mol | n��CO��/mol | ||
| 1 | 150 | 10.0 | 10.0 | 0 | 0 | 7.0 |
| 2 | 150 | 7.0 | 8.0 | 2.0 | 4.5 | a |
| 3 | 400 | 20.0 | 20.0 | 0 | 0 | 16.0 |
| A�� | ������Ӧ�����ȷ�Ӧ | |
| B�� | ʵ��1 ��ƽ��ʱ��CO ��ת����Ϊ70% | |
| C�� | ʵ��2 ��ƽ��ʱ��a��7.0 | |
| D�� | ʵ��3 ��ƽ����ٳ���1.0molH2��K ֵ����ƽ�������ƶ� |
| A�� | MnO2 | B�� | CO2 | C�� | H2O2 | D�� | ���� |
| A�� | 25��ʱ���ô�����Һ�ζ���Ũ��NaOH��Һ��pH=7��V�����ᣩ��V��NaOH�� | |
| B�� | ϡ�����ˮϡ�ͣ��������̶�������Һ��pH��С | |
| C�� | �����½�pH=3ϡ������Һϡ�͵�ԭ�����10������Һ��pH=4 | |
| D�� | 25��ʱ��amol/LһԪ����bmol/LNaOH�������Ϻ�pH=7�����û����Һ��һ����c��A-��=c��NA+�� |