��Ŀ����
��������Ҫ�ɷ�ΪFeS2�����ҹ���������᳧��ȡ�������Ҫԭ�ϡ�ij��ѧѧϰС���ij������ʯ��������ʵ��̽����
[ʵ��һ]�ⶨ��Ԫ�صĺ�����
��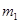 g�û�������Ʒ������ͼ1��ʾװ�ã��гֺͼ���װ��ʡ�ԣ���ʯӢ���У���a�����ϵػ���ͨ���������������ʯӢ���еĻ�������Ʒ����Ӧ��ȫ��ʯӢ���з�����Ӧ�Ļ�ѧ����ʽΪ��4FeS2+11O2
g�û�������Ʒ������ͼ1��ʾװ�ã��гֺͼ���װ��ʡ�ԣ���ʯӢ���У���a�����ϵػ���ͨ���������������ʯӢ���еĻ�������Ʒ����Ӧ��ȫ��ʯӢ���з�����Ӧ�Ļ�ѧ����ʽΪ��4FeS2+11O2 2Fe2O3+8SO2
2Fe2O3+8SO2
��Ӧ��������ƿ�е���Һ������ͼ2��ʾ������
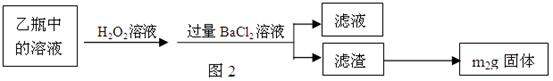
[ʵ���]�ⶨ��Ԫ�صĺ�����
�ⶨ��Ԫ�غ�����ʵ�鲽����ͼ3��ʾ��
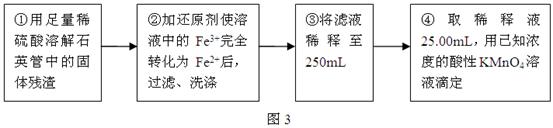
�������ۣ�
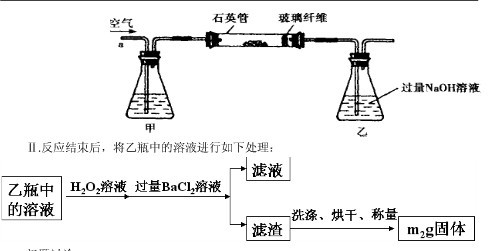
��1�����У���ƿ����ʢ�Լ��� ��Һ����ƿ�ڷ�����Ӧ�����ӷ���ʽ�У�
�� ��
��2�����е������ڳ���ǰ��Ӧ���еIJ����� ��
��3�����У�����H2O2��Һ������������������������ ��
��4����IJ�����У���Ҫ�õ����������ձ�������������ͷ�ι��⣬���� ��
д�����з�Ӧ�����ӷ���ʽ ��
��5���û���������Ԫ�ص���������Ϊ ��
��1��NaOH (2��)  ��
��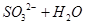 (2��)��
(2��)��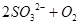 ��
��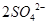 (2��) ��2��ϴ�ӡ�����(2��) ��3��ʹ
(2��) ��2��ϴ�ӡ�����(2��) ��3��ʹ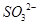 ��ȫת����
��ȫת����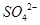 (2��)
(2��)
��4��250ml ����ƿ(2��) 5Fe2+ + 2MnO4-+16H+==5Fe3++2Mn2++8H2O(2��)
��5��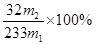 (2��)
(2��)
����������1�����ڿ����к���CO2��ˮ���������Ա����ȥ���Է�ֹ���ź����SO2�����գ����ʢ�ŵ�������������Һ��SO2������������ܱ�����������Һ���գ���Ӧ�ķ���ʽ��SO2��2OH��=SO32����H2O������Ϊ�������Ƽ��ױ��������������Ի����ܷ�����Ӧ2SO32����O2=2SO42����
��2�������ڳ���֮ǰ����Ҫϴ�Ӻ��
��3��Ϊ��ʹ��Һ�е�SO32����ȫ����������SO42��������Ĺ�������Ӧ���ǹ����ġ�
��4������Ҫϡ����250ml�����Ի���Ҫ250ml����ƿ�����Ը��������Һ���������ԣ��������������ӣ���Ӧ�ķ���ʽ��5Fe2+ + 2MnO4-+16H+==5Fe3++2Mn2++8H2O��
��5�����ᱵ��������m2g�����ʵ�����m2/233mol����SO2�����ʵ�����m2/233mol��������Ԫ�ص�����������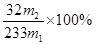 ��
��

| |||||||||||||||||||
���������û�������������������ų��ķ�������Ҫ��ѧ�ɷ�ΪSiO2Լ45%��Fe2O3Լ40%��Al2O3Լ10%��MgOԼ5%��Ŀǰ�ҹ��Ѿ��ڼ�����ȡ��ͻ�ơ������������з�������ֳɷֲ��������á������̺�����������£�

�����ϵ�֪��
|
�������� |
�ܶȻ�(Ksp) |
pHֵ |
|
|
��ʼ���� |
��ȫ���� |
||
|
Mg(OH)2 |
5.6��10��12 |
9.3 |
10.8 |
|
Fe(OH)3 |
2.8��10��16 |
2.7 |
3.7 |
|
Al(OH)3 |
1.3��10��33 |
3.7 |
4.7 |
��ش��������⣺
��1��д������A�Ļ�ѧʽΪ ��
��2����Ҫ�ⶨ��Һ��pH�Ƿ�ﵽ3.7������ʵ����Ʒ�п�ѡ�õ��� ��
A��ʯ����Һ B���㷺pH��ֽ C������pH��ֽ D��pH��
��3������������ӷ�Ӧ����ʽ
����ҺD���ɹ���E �� ����ҺF���ɹ���G ��
��4��Ҫ������C������E����G��ת��Ϊ��Ӧ���ȶ����������е�ʵ�����Ϊ ��
��5������������Һ����ı仯���������ҺH��c��Mg2����= ��