��Ŀ����
5������ƽ������Һ��ѧ�е���Ҫ���ݣ�ij��ѧ��ȤС����������Ϊ��̽����������ʣ���1��ʵ��һ���������Ʊ���Һ�����ƣ�
��Ҫ����0.1000mol/LNaOH����Һ250mL����Ҫ�õ��IJ��������в��������ձ�����ͷ�ιܺ�250��������ƿ���������������û�н��ձ��ȵ�ϴ��Һһ��ת������ƿ����������������ȷ������£����ñ���Һ�����ζ����У�2���е�δ֪Ũ�ȵ�������Һ������ʹ�ඨ�Ľ��ƫ�ߣ���ߡ������͡�������Ӱ�족��
��2��ʵ���������һƿ������Һ�������²ⶨ��������ĵ���ƽ�ⳣ�������ʵ�鷽�����������������Ͷ�Ӧ�IJⶨ������д�ڱ�һ�У�
��һ��
| ���������� | �ⶨ���� |
| ��������Һ�����ʵ���Ũ�� | ��ȡ25.00mL������Һ����ƿ�У��μ�ָʾ������0.1000mol/LNaOH����Һװ���ʽ�ζ��ܣ��ζ����յ㣬��¼���ݣ��ظ��ζ�2�Σ� |
| ��H+�����ʵ���Ũ�� | ȡ����������Һ���ձ��У���pH�ƻ���pH��ֽ�ⶨ��ҺpH�� |
��3��ʵ������̽�����ǿ��������þ����Ӧ���ʵ�Ӱ�죮
�����ʵ�鷽���������������c=2.0g��
������
| ��� | ������� | ���Ũ�ȣ�mol/L�� | ������/mL | þ������/g |
| l | ���� | 0.5 | 17.0 | 2.0 |
| 2 | ���� | 0.5 | 17.0 | c |
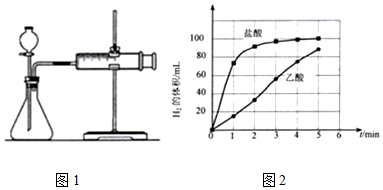
a��ͼ1��װ���У�������ҩƷ֮ǰ������װ�õ������ԣ�
b����Ӧ��ʼ��ÿ��1min��¼һ������H2�������
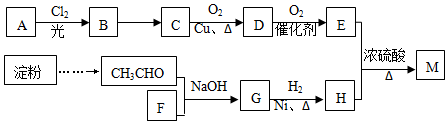
c��������¼�����ݻ��Ƴ�����ͼ��ͼ2����
��д��þ��������Һ��Ӧ�����ӷ���ʽ��Mg+2CH3COOH=Mg2++2CH3COO-+H2����
������0��5min������þ����Ӧ�ķ�Ӧ���ʱ仯���ɣ�������þ����Ӧ��������ʱ��仯�����ԣ�������þ����Ӧ��ʼ�η�Ӧ���ʺܿ죬һ��ʱ���Ӧ�������Լ�С��
���� ��1��Ҫ����0.1000mol/LNaOH����Һ250mL���������ܽ⡢ת�Ƶ�����ƿ��ϴ��ת�ơ����ݵȲ���ѡ�����������������������û�н��ձ��ȵ�ϴ��Һһ��ת������ƿ��������ҺŨ�ȼ�С���ζ�ʵ��������Һ�������c�����⣩=$\frac{c������V������}{V�����⣩}$������
��2������һƿ������Һ�������²ⶨ��������ĵ���ƽ�ⳣ������Ҫ�ⶨ��ҺPH���㣻
�������ij�¶�ʱ���������Һ�����ʵ���Ũ��Ϊ0.1000mol/L��pH=3�����ڸ��¶�ʱ����ĵ���ƽ�ⳣ��K=$\frac{c��{H}^{+}��c��C{H}_{3}CO{O}^{-}��}{c��C{H}_{3}COOH��}$��
��3����̽�����ǿ��������þ����Ӧ���ʵ�Ӱ����Ҫ��������Ũ����ͬ��þ����С�������������ͬ��Ҫ̽�����ǿ��������þ����Ӧ���ʵ�Ӱ�죬����뱣������Ӱ�췴Ӧ���ʵ����ر���һ�£�
��a������ⶨװ�ã�������ҩƷ֮ǰ������װ�������ԣ�
b����ͼ��֪����Ҫͨ����������ͬ��ʱ�����ռ�������������Ĵ�С��������Ӧ���ʣ��ݴ˷�����Ӧ��ʼ��IJ�����
��þ�ʹ��ᷴӦ���ɴ���þ��������
��ͨ��ͼ����������λʱ��������������ı仯��֪���ᡢ������þ����Ӧ�����ʵı仯�����
��� �⣺��1����Ҫ����0.1000mol/LNaOH����Һ250mL����Ҫ�õ��IJ��������в��������ձ�����ͷ�ιܺ�250ml����ƿ���������������û�н��ձ��ȵ�ϴ��Һһ��ת������ƿ��������ҺŨ�ȼ�С���ζ�ʵ��������Һ�������c�����⣩=$\frac{c������V������}{V�����⣩}$��������ʹ�ඨ�Ľ��ƫ�ߣ�
�ʴ�Ϊ��250ml����ƿ���ߣ�
��2������һƿ������Һ�������²ⶨ��������ĵ���ƽ�ⳣ�������ʵ�鷽������ȡ25.00mL������Һ����ƿ�У��μ�ָʾ������0.1000mol/LNaOH����Һװ���ʽ�ζ��ܣ��ζ����յ㣬��¼���ݣ��ظ��ζ�2�Σ��ⶨ������ҺŨ�ȣ�Ҳ����ȡ����������Һ���ձ��У���pH�ƻ���pH��ֽ�ⶨ��ҺpH���õ�H+�����ʵ���Ũ�ȣ�����ƽ�ⳣ����
�ʴ�Ϊ��
| ���������� | �ⶨ���� |
| ��������Һ�����ʵ���Ũ�� | |
| pH�ƻ���pH��ֽ |
�ʴ�Ϊ��1��10-5��
��3����̽�����ǿ��������þ����Ӧ���ʵ�Ӱ����Ҫ��������Ũ����ͬ��þ����С�������������ͬ��������c=2.0g��
�ʴ�Ϊ��2.0��
��a������ⶨ����©����������ҩƷ֮ǰ������װ�������ԣ�
�ʴ�Ϊ�����װ�õ������ԣ�
b����ͼ��֪����Ҫͨ����������ͬ��ʱ������ռ�������������Ĵ�С��������Ӧ���ʣ����ڷ�Ӧ��ʼ��Ӧÿ��1min��¼һ������H2�������
�ʴ�Ϊ��ÿ��1min��¼һ������H2�������
��þ�ʹ��ᷴӦ���ɴ���þ����������Ӧ�����ӷ���ʽΪ��Mg+2CH3COOH=Mg2++2CH3COO-+H2����
�ʴ�Ϊ��Mg+2CH3COOH=Mg2++2CH3COO-+H2����
��ͨ��ͼ�������֪����λʱ���ڴ�����þ����Ӧ��������������仯��С����������þ���ķ�Ӧ���ʱ仯����������þ���ķ�Ӧһ��ʼ�ܿ죬һ��ʱ���λʱ�������ɵ�������������Ա�С������Ӧ�������Լ�С��
�ʴ�Ϊ��������þ����Ӧ��������ʱ��仯�����ԣ�������þ����Ӧ��ʼ�η�Ӧ���ʺܿ죬һ��ʱ���Ӧ�������Լ�С��
���� ���⿼����Ӱ�췴Ӧ���ʵ����ء�ʵ�鷽��������Լ�ͼ�����⣬�ۺ��Խ�ǿ���Ǹ߿��ĸ�Ƶ���㣬��Ŀ�Ѷ��еȣ�

| A�� | �ȵĴ�ˮ | B�� | �ȵ��ռ� | C�� | ���Ũ���� | D�� | �������ͭ��Һ |
| A�� | ��ĭ��������õ����մ�������� | |
| B�� | ��ˮ�еμ���������FeCl3��Һ���γɴ���Ľ��壬����������ǿ | |
| C�� | ����Ĵ������Դ�������Ŀ����������ղ���ı��� | |
| D�� | Na��H2O�ķ�Ӧ���ؼ��ķ��ȷ�Ӧ���÷�Ӧ���Է����� |
| A�� | ���ࡢ��֬�������ʵ�ˮ����ﶼ�Ƿǵ���� | |
| B�� | ���ǡ���ѿ�ǵķ���ʽ��ΪC12H22O11������Ϊͬ���칹�� | |
| C�� | �����Cl2�ķ�Ӧ����ϩ��Br2�ķ�Ӧ����ͬһ���͵ķ�Ӧ | |
| D�� | �Ҵ�����������о����й�����-OH�����Ծ�����NaOH��Һ��Ӧ |
| A�� | Fe+CuSO4�TFeSO4+Cu | B�� | 2KClO3$\frac{\underline{\;MnO_{2}\;}}{��}$2KCl+3O2�� | ||
| C�� | S+O2$\frac{\underline{\;��ȼ\;}}{\;}$SO2 | D�� | NaOH+HCl�TNaCl+H2O |
| A�� | 2-�ȶ������������ơ��Ҵ��ڼ��������µ���ȥ��Ӧ | |
| B�� | ��ϩͨ����ˮ�еļӳɷ�Ӧ | |
| C�� | ���ȵ�ͭ˿���������Ҵ��е�������Ӧ | |
| D�� | ������������������һ�������µļӳɷ�Ӧ |
| A�� | �մɵ����� | B�� | ����ӡˢ�Ű��� | ||
| C�� | ���ں��̻���ȼ�� | D�� | ˾ĸ�춦�������ͭ�� |

| A�� | ����1 molHF����ų�������Ϊ270 KJ | |
| B�� | H2��g��+F2��g����2HF��l��+270 KJ | |
| C�� | ��Ӧ�������������������������� | |
| D�� | �÷�Ӧ�����ȷ�Ӧ |
 ��
�� ��
�� ��
��