��Ŀ����
�йش����С�մ��������ȷ���ǣ�������
| A����������Na2CO3��NaHCO3�ֱ���������ϡ���ᷴӦ��NaHCO3������CO2�� |
| B����������Na2CO3��NaHCO3�ֱ���������ͬ�����ᷴӦ��NaHCO3��Ӧ�������ĵ������ |
| C����NaHCO3��Һ�е���Ba��OH��2��Һ��������Na2CO3��Һ�м���Ba��OH��2��Һ���ְ�ɫ���� |
| D��Na2CO3��NaHCO3����������������NaOH��Ӧ |
���㣺�Ƶ���Ҫ������
ר�⣺
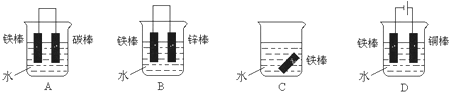
������A������̼���ƺ�̼�����Ƶĺ�̼��ȷ�����ɶ�����̼�Ķ��٣�
B�����ݼ��跨���������������Ķ��٣�
C��̼�����ƺ�����������Ӧ����̼�ᱵ������
D��������������Ʒ�Ӧ��
B�����ݼ��跨���������������Ķ��٣�
C��̼�����ƺ�����������Ӧ����̼�ᱵ������
D��������������Ʒ�Ӧ��
���
�⣺A������NaHCO3��NaCO3�Ļ�ѧʽ֪��̼�������к�̼���࣬���Ե������Ĵ��С�մ�ֱ�������ϡH2SO4��Ӧ��С�մ������CO2�࣬��A��ȷ��
B����̼���ƺ�̼�����Ƶ���������1g����̼���������ĵ���������ʵ�����x��
̼����������������ʵ�����y��
NaHCO3+HCl=NaCl+CO2��+H2O
84g 1mol
1g x
x=
mol��
Na2CO3+2HCl=2NaCl+CO2��+H2O
106g 2mol
1g y
y=
=
mol��
x��y������̼�������ĵ�����࣬��B����
C��NaHCO3+Ba��OH��2=BaCO3��+NaOH+H2O��������С�մ���Һ�е���Ba��OH��2��Һ�г�������C����
D������ֻ�����ᷴӦ�������������Ʒ�Ӧ����D����
��ѡA��
B����̼���ƺ�̼�����Ƶ���������1g����̼���������ĵ���������ʵ�����x��
̼����������������ʵ�����y��
NaHCO3+HCl=NaCl+CO2��+H2O
84g 1mol
1g x
x=
| 1g��1mol |
| 84g |
Na2CO3+2HCl=2NaCl+CO2��+H2O
106g 2mol
1g y
y=
| 1g��2mol |
| 106g |
| 1 |
| 53 |
x��y������̼�������ĵ�����࣬��B����
C��NaHCO3+Ba��OH��2=BaCO3��+NaOH+H2O��������С�մ���Һ�е���Ba��OH��2��Һ�г�������C����
D������ֻ�����ᷴӦ�������������Ʒ�Ӧ����D����
��ѡA��
���������⿼����̼���ƺ�̼�����Ƶ����ʣ��״�ѡ����C�����ݷ���ʽ���з������ϼ���Ҳ����׳�����

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
�ڢ�����ƿ �������� ���Թ� ����Ͳ ���ձ����������У����ܼ��ȵ��ǣ�������
| A���٢� | B���ڢۢ� |
| C���ۢ� | D���٢ڢ� |
Ϊ�˷�ֹFeSO4�����������ʣ�ʵ����������FeSO4��Һʱ��������Һ�м��������ģ�������
| A��ͭ�� |
| B������ |
| C��� |
| D��Fe2��SO4�� |
�����£�������������Һ����0.1mol/L NaOH��Һ ��pH=11��NaOH��Һ ��0.1mol/L CH3COOH��Һ ��pH=3��CH3COOH��Һ������˵����ȷ���ǣ�������
| A����ˮ�������c��H+�����٣��� |
| B����ϡ�͵�ԭ����100����pH�����ͬ |
| C������ۻ�ϣ�����ҺpH=7����V��NaOH����V��CH3COOH�� |
| D������ܻ�ϣ�����Һ�����ԣ���������Һ������Ũ�ȿ���Ϊ��c��CH3COO-����c��H+����c��Na+����c��OH-�� |
��һ�ܱ������г���һ������N2��H2��������ӦN2��g��+3H2��g��?2NH3��g�����ⶨ��Ӧ��ʼ���2s��������ƽ�����ʣ��ͣ�H2��=0.45mol/��L?s������2sĩNH3��Ũ��Ϊ��������
| A��0.50mol/L |
| B��0.60mol/L |
| C��0.45mol/L |
| D��0.55mol/L |
��֪1mol����ת��Ϊ1mol���ף�����18.39kJ��������4P��s���죩+5O2��g���T2P2O5��s����H1 ��4P��s���ף�+5O2��g���T2P2O5��s����H2�����H1���H2�Ĺ�ϵ��ȷ���ǣ�������
| A����H1=��H2 |
| B����H1����H2 |
| C����H1����H2 |
| D����ȷ�� |
ҽѧ��ͨ���÷�����14C��ǵ�C60������һ��C60���������������ض������¿�ͨ������DNAɱ��ϸ�����Ӷ����ư��̲������й�14C��������ȷ���ǣ�������
| A����14N ���е���������ͬ |
| B����C60��ͬ�������� |
| C����C60����̼ͨԭ�ӵĻ�ѧ���ʲ�ͬ |
| D����12C��Ϊͬλ�� |
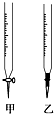 ijѧ����0.1000mol/L NaOH��Һ�ζ�δ֪Ũ�ȵ�������Һ��������ɷֽ�Ϊ���¼�����
ijѧ����0.1000mol/L NaOH��Һ�ζ�δ֪Ũ�ȵ�������Һ��������ɷֽ�Ϊ���¼�����