��Ŀ����
9��������ͼ��ʾ��ʵ��װ�ã��ش��������⣺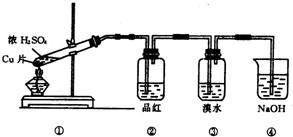
��1��д��ͭ��Ũ���ᷴӦ�Ļ�ѧ����ʽCu+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+SO2��+2H2O
��2��֤��SO2����Ư���Ե���������ɫ���ټ��ȸ���Һ���������ֱ�Ϊ��ɫ��
��3��װ�â��е���������ɫ��֤��SO2���л�ԭ�ԣ�
��4��װ�âܵ����������ն���Ķ����������壬��ֹ��Ⱦ������
��5����ʵ��������õ�25g CuSO4•5H2O���壬��ԭ�������������9.8g��
���� ��ʵ��װ��ͼ��֪��װ�â�ͭ��Ũ���ᷴӦ���ɶ�������װ�â���֤���������Ư���ԡ�װ�â���֤��������Ļ�ԭ�ԡ�װ�â�β�����գ�
��1��ͭ��Ũ�����ڼ��������·�Ӧ��������ͭ�����������ˮ��
��2��SO2����ʹƷ����Һ��ɫ��������SO2����Ư���ԣ�SO2��Ư������ʱ�Ե�Ư�ף�
��3������������н�ǿ�Ļ�ԭ�ԣ������嵥�ʷ�Ӧ��������������
��4������������Ⱦ������װ�â�Ϊβ������װ�ã�
��5����ʵ��������õ�25g CuSO4•5H2O��������ʵ���Ϊ��$\frac{25g}{250g/mol}$=0.1mol������ԭ��������ʵ�����������ͭ��������ʵ���������m=nM�����м��㣮
��� �⣺��1��ͭ��Ũ�����ڼ��������·�Ӧ��������ͭ�����������ˮ����Ӧ�ķ���ʽΪ��Cu+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+SO2��+2H2O��
�ʴ�Ϊ��Cu+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+SO2��+2H2O��
��2��SO2����ʹƷ����Һ��ɫ��������SO2����Ư���ԣ�SO2��Ư������ʱ�Ե�Ư�ף����ɵ����ʲ��ȶ��������ֽ⣬���±�Ϊ��ɫ��
�ʴ�Ϊ����ɫ���ֱ�Ϊ��ɫ��
��3������������н�ǿ�Ļ�ԭ�ԣ������嵥�ʷ�Ӧ��������������ᣬ���װ�â��е�����Ϊ��Һ��ɫ��
�ʴ�Ϊ����ɫ����ԭ�ԣ�
��4������������Ⱦ����������������Ա�����������Һ���գ����ٿ�����Ⱦ�����װ�âܵ������ǣ����ն���Ķ����������壬��ֹ��Ⱦ������
�ʴ�Ϊ�����ն���Ķ����������壬��ֹ��Ⱦ������
��5����ʵ��������õ�25g CuSO4•5H2O��������ʵ���Ϊ��$\frac{25g}{250g/mol}$=0.1mol������ԭ��������ʵ�����������ͭ��������ʵ�����m=nM=0.1mol��98g/mol=9.8g���ʴ�Ϊ��9.8��
���� ���⿼��Ũ����Ͷ�����������ʣ��漰Ũ�����ǿ�����ԺͶ��������Ư���ԡ���ԭ�ԣ�����ʵ��װ��ͼ�ǽ���Ĺؼ�������Ƚϻ������ѶȲ���

 ��У����ϵ�д�
��У����ϵ�д�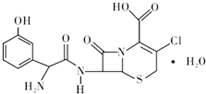 ͷ�߿������˹��ϳɵĵڶ����ڷ�ͷ�߾������أ���Ҫ�����ϡ��º�������Ⱦ���ж��ס�Ƥ���������Ⱦ��֢״����ṹ��ͼ��ʾ�����й���ͷ�߿����˵��������ǣ�������
ͷ�߿������˹��ϳɵĵڶ����ڷ�ͷ�߾������أ���Ҫ�����ϡ��º�������Ⱦ���ж��ס�Ƥ���������Ⱦ��֢״����ṹ��ͼ��ʾ�����й���ͷ�߿����˵��������ǣ�������| A�� | �����ʽ���Ա�ʾΪC15H16ClN3O6S | |
| B�� | �����ʾ������� | |
| C�� | 1 mol�û�������NaOH��Һ����ʱ����NaOH�����ʵ������Ϊ 4 mol | |
| D�� | ��������FeCl3��Һ����ɫ |
| A�� | ��ɫ��ѧ�ĺ��������û�ѧԭ����Դͷ�ϼ��ٺ�������ҵ�����Ի�������Ⱦ | |
| B�� | ������ġ�ԭ�Ӿ��á����Ƿ�Ӧ���ԭ��ȫ��ת��Ϊ���������ղ��� | |
| C�� | ��ɫ��ѧ��Ӧѡ���ԭ�ϡ��������ܼ���Ӧ���������� | |
| D�� | ��ϩ�ֱ�����ˮ��������Ȼ�̼��Һ��Ӧ�Ʊ�1��2-���������ԭ�������ʾ�Ϊ100% |
| A�� | ���õ�ⱥ��MgCl2��Һ�ķ�����ý���þ | |
| B�� | ��ⷨ������ͭ���ô�ͭ����������ͭ������ | |
| C�� | �������Al2O3�Ʊ�������ʱ���������ʯʹAl2O3�����ۻ� | |
| D�� | �ȼҵ�е��ʳ��ˮʱ�������ӽ���Ĥ�ɷ�ֹ������NaOH��Һ��Ӧ |
| A�� | Y��Z��W�������Ӿ����ƻ�ˮ�ĵ���ƽ�� | |
| B�� | WԪ���������Ӧˮ���������һ��ǿ��Z | |
| C�� | ��X��Y��Z����Ԫ����ɵĻ����ﲻֹ2�� | |
| D�� | ��ΪX���⻯����Ӽ������������X���⻯���Z���⻯���ȶ� |
 �����ѣ�Voltaic pile������Ƭ�ͽṹ��ͼ������Բ�ε�п�塢ͭ�����ѻ����ɣ����е�Բ��֮��зż�����ˮ�ݹ��IJ�������˵����ȷ���ǣ�������
�����ѣ�Voltaic pile������Ƭ�ͽṹ��ͼ������Բ�ε�п�塢ͭ�����ѻ����ɣ����е�Բ��֮��зż�����ˮ�ݹ��IJ�������˵����ȷ���ǣ�������| A�� | �������ܽ�����ת���ɻ�ѧ�� | B�� | п������ | ||
| C�� | ��ˮ�ǵ������Һ | D�� | �����ѹ���ʱͭ����ԭ |
| A�� | ��֪K=$\frac{{c}^{2}C{O}_{2}•{c}^{6}•{H}_{2}}{{c}^{3}{H}_{2}O•cC{H}_{3}C{H}_{2}OH}$�����Ӧ�Ļ�ѧ��Ӧ���Ա�ʾΪ2CO2��g��+6H2��g��?CH3CH2OH��g��+3H2O��g�� | |
| B�� | ��ijŨ���������Һ�����¶ȣ������ĵ���ȣ������͵���ƽ�ⳣ��Ka����� | |
| C�� | �����£���ˮ�м���̼���ƣ�ˮ�ĵ���̶ȱ��KW��� | |
| D�� | ��ѧƽ�ⳣ��K���¶��йأ����¶ȵ����ߣ�K��������Ҳ���ܼ�С�����߲��� |

 ��
��