��Ŀ����
TMB��һ������ָ�Ƽ���ɫԭ�Լ�����̼���⡢������Ԫ����ɣ�����������Է�������֮��Ϊ120��ij�о���ѧϰС���ͬѧ���������������ⶨTMB�ķ���ʽ������Ҫ����Ϊ�������������н�4.80g TMB��Ʒ��������Ԫ��ת��ΪN2�������������ռ��ֱ�����ˮ������CO2�����ͼ��ѡ���ʵ���װ�ã�װ�÷���������Ҫ����װ�õ�����ȥ��������Ʒ����ѡ����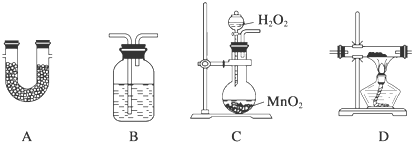
��1������ѡ�õ����������ظ�ѡ�ã�����˳�����ϵ������������±�����д����������Ӧ������Լ������Ƽ����ã�
��2��ʵ���Ƶ�A���Ժ��װ�ã�����Aװ�ò�����ÿ�����վ�����ȫ�ģ�������������3.60g��14.08g��0.14g����TMB�ķ���ʽΪ ��

��1������ѡ�õ����������ظ�ѡ�ã�����˳�����ϵ������������±�����д����������Ӧ������Լ������Ƽ����ã�
| ѡ�õ����� | �����Լ� | �����Լ������� |
| C | H2O2��Һ��MnO2 | �������� |
| D | CuO��ĩ | ���������ɵ�COת��ΪCO2 |
���㣺�л���ʵ��ʽ�ͷ���ʽ��ȷ��
ר�⣺ʵ��̽�������ݴ�����
��������1��ʵ��ԭ������TMB����������Ϊ������̼��ˮ�͵��������������ռ��ֱ�����ˮ�Ͷ�����̼����ȷ����������̼���⡢��ԭ�ӵı�����ϵ��������ʵ���У�Ҫ��̼Ԫ��ȫ���Զ�����̼��ʽ���֣��������������Ҫ������ͭ������ϣ�Ϊ��ֹ��������ʱ����ˮ������Ӱ�죬Ҫ�ȶ��������и��ѡ�������迼������Ҫ��ʵ���Ƶ�Aװ�ô��õ��Ⱥ�˳�����������ֱ�����3.60g��14.08g������˸�������ѡ������B���������ռ��ֱ�����ˮ��CO2�迼���Լ�Ӱ�죬������ˮ������CO2��
��2������Aװ������14.08g���������Ʒ��CԪ�ص����ʵ�����Aװ������3.60g���������Ʒ��HԪ�ص����ʵ������ٸ�����Ʒ��������4.80g���������Ʒ��NԪ�ص����ʵ�������TMB������̼���⡢����ԭ�Ӹ����ȾͿ��������������Է�������֮�������Է������������ԭ�Ӹ����ȼ��������ʽ��
��2������Aװ������14.08g���������Ʒ��CԪ�ص����ʵ�����Aװ������3.60g���������Ʒ��HԪ�ص����ʵ������ٸ�����Ʒ��������4.80g���������Ʒ��NԪ�ص����ʵ�������TMB������̼���⡢����ԭ�Ӹ����ȾͿ��������������Է�������֮�������Է������������ԭ�Ӹ����ȼ��������ʽ��
���
�⣺��1��ʵ��ԭ������TMB����������Ϊ������̼��ˮ�͵��������������ռ��ֱ�����ˮ�Ͷ�����̼����ȷ����������̼���⡢��ԭ�ӵı�����ϵ��������ʵ���У�Ҫ��̼Ԫ��ȫ���Զ�����̼��ʽ���֣��������������Ҫ������ͭ������ϣ�Ϊ��ֹ��������ʱ����ˮ������Ӱ�죬Ҫ�ȶ��������и��ѡ�������迼������Ҫ��ʵ���Ƶ�Aװ�ô��õ��Ⱥ�˳�����������ֱ�����3.60g��14.08g������˸�������ѡ������B���������ռ��ֱ�����ˮ��CO2�迼���Լ�Ӱ�죬������ˮ������CO2��������˳��Ϊ��
�ʴ�Ϊ��
��2��ʵ���Ƶ�Aװ�ô��õ��Ⱥ�˳�����������ֱ�����3.60g��14.08g��0.14g����m��H2O��=3.60g��m��CO2��=14.08g����0.14gΪ�����е�CO2��H2O����װ���е���������
n��H2O��=
=0.2mol��n��H��=0.4mol��m��H��=0.4g��
n��CO2��=
=0.32mol��n��C��=0.32mol��m��C��=0.32mol��12g/mol=3.84g��
����m��N��=4.80g-0.4g-3.84g=0.56g��
n��N��=
=0.04mol��
��n��C����n��H����n��N��=0.32mol��0.4mol��0.04mol=8��10��1��
TMB����Է�������Ϊ��2��120=240�������ʽΪ��C8nH10nNn�����У�12��8n+10n+14n=240�����n=2�����Է���ʽΪC16H20N2��
�ʴ�Ϊ��C16H20N2��
| ѡ�õ����� | �����Լ� | �����Լ������� |
| C | H2O2��Һ��MnO2 | �������� |
| B | ŨH2SO4 | �������� |
| D | TMB | TMB������CO2��H2O��N2 |
| D | CuO | ��֤Cȫ��ת��ΪCO2 |
| A | CaCl2 | ����ˮ�� |
| A | NaOH | ����CO2 |
| A | ��ʯ�� | ��ֹ�����е�CO2��H2O����װ���� |
| ѡ�õ����� | �����Լ� | �����Լ������� |
| C | H2O2��Һ��MnO2 | �������� |
| B | ŨH2SO4 | �������� |
| D | TMB | TMB������CO2��H2O��N2 |
| D | CuO | ��֤Cȫ��ת��ΪCO2 |
| A | CaCl2 | ����ˮ�� |
| A | NaOH | ����CO2 |
| A | ��ʯ�� | ��ֹ�����е�CO2��H2O����װ���� |
n��H2O��=
| 3.6g |
| 18g/mol |
n��CO2��=
| 14.08g |
| 44g/mol |
����m��N��=4.80g-0.4g-3.84g=0.56g��
n��N��=
| 0.56g |
| 14g/mol |
��n��C����n��H����n��N��=0.32mol��0.4mol��0.04mol=8��10��1��
TMB����Է�������Ϊ��2��120=240�������ʽΪ��C8nH10nNn�����У�12��8n+10n+14n=240�����n=2�����Է���ʽΪC16H20N2��
�ʴ�Ϊ��C16H20N2��
�����������Զ���ʵ��Ϊ���⣬ȷ���л���ķ�����ɣ���Ŀ�ѶȽϴ����״���Ϊ��1����ע��ʵ������ԭ���Ͳ������裮

��ϰ��ϵ�д�
�����Ŀ
��300mL KOH��Һ�л���ͨ��2.24L����״���£�CO2���壬��ַ�Ӧ���ڼ�ѹ������Һ���õ�11.9g��ɫ���壮������˵������ȷ���ǣ�������
| A���˰�ɫ����ΪKOH��K2CO3�Ļ���� |
| B���˰�ɫ�����к���K2CO3 6.90g |
| C��ԭKOH��Һ�е����ʵ���Ũ����0.500 mol?L-1 |
| D���˰�ɫ����KHCO3 |
����ܳʽ����ԣ�������ʵ��˵����һ��ʵ���ǣ�������
| A���Ѿ��Ƶ�I��NO3��3��I��ClO4��3?2H2O�Ⱥ�I3+�Ļ����� |
| B���Ѿ��Ƶ�IBr��ICl ��±�ػ����� |
| C����������KI��Һ�γ�I3- |
| D��I4O9���Կ����ǵ����� |
�������ʵ���;����ȷ���ǣ�������
| A��AgBr�����˹����� |
| B��MgO�������ͻ���� |
| C��BaSO4�������͡��Լ� |
| D��ҽ���ϳ���75%�ľƾ��������� |
����˵��������ǣ�������
| A��Ԫ�ط����ǿ���ȷ���������Ƿ���C��H��O��N��Cl��Br��Ԫ�� |
| B���ú�������ǿ���ȷ���������Ƿ����ijЩ�л�ԭ���� |
| C����ԭ�����չ�����ȷ�������к�����Щ����Ԫ�� |
| D��12C�ķ����Կ����ڿ��Ŷϴ� |