��Ŀ����
�Ȼ���������ض��dz�����ˮ����������ͼΪ�Ʊ��Ȼ�������һ�������Ʊ�������صĹ������̣�
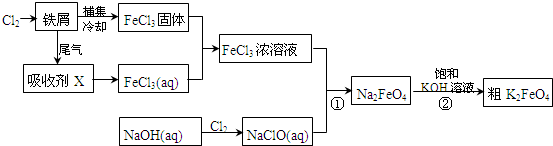
��ش��������⣺
��1���Ȼ����ж�����;��
���Ȼ�������ˮ�����������ӷ���ʽ��ʾ��ԭ�� ��
�ڹ�ҵ�ϳ���FeCl3��Һ��ʴͭӡˢ��·�壮����������FeCl3��ǿ�� �ԣ�
��2�����ռ�X�Ļ�ѧʽΪ ��Ϊ�������ռ��Ƿ�����ȫת��ΪFeCl3��Һ��������������Ը��������Һ�����ܿ챻�������ǣ������ӷ���ʽ��ʾ�� ����ȷ�ļ��鷽���ǣ�ȡ��������Һ������ ��д��ѧʽ����Һ����������ɫ������������֤��ת����ȫ��
��3�����������·�Ӧ�ٵ����ӷ���ʽΪ ��
��4�����̢ڽ������Һ�����Сʱ�����ã����˻�ôֲ�Ʒ���÷�Ӧ�Ļ�ѧ����ʽΪ2KOH+Na2FeO4�TK2FeO4+2NaOH���������ط�Ӧԭ��������Ӧ�ܷ�����ԭ�� ��
��5������K2FeO4��Ʒ���ؽᾧ�����ˡ� �� �����ýϴ�����K2FeO4��

��ش��������⣺
��1���Ȼ����ж�����;��
���Ȼ�������ˮ�����������ӷ���ʽ��ʾ��ԭ��
�ڹ�ҵ�ϳ���FeCl3��Һ��ʴͭӡˢ��·�壮����������FeCl3��ǿ��
��2�����ռ�X�Ļ�ѧʽΪ
��3�����������·�Ӧ�ٵ����ӷ���ʽΪ
��4�����̢ڽ������Һ�����Сʱ�����ã����˻�ôֲ�Ʒ���÷�Ӧ�Ļ�ѧ����ʽΪ2KOH+Na2FeO4�TK2FeO4+2NaOH���������ط�Ӧԭ��������Ӧ�ܷ�����ԭ��
��5������K2FeO4��Ʒ���ؽᾧ�����ˡ�
���㣺�Ʊ�ʵ�鷽�������,���κ������ε��ת��
ר�⣺������Ҫ�Ľ������仯����
��������1�����Ȼ�������ˮ������ΪFe3+ˮΪFe��OH��3�����Ե�ʣ��ڹ�ҵ�ϳ���FeCl3��Һ��ʴͭӡˢ��·����Fe3+������Cu��Ե�ʣ�
��2�����ռ�X����Cl2��Ӧ�IJ�����FeCl3��X��FeCl2��Ϊ�������ռ��Ƿ�����ȫת��ΪFeCl3��Һ�������Ը��������Һ������֤����ΪCl-�ܱ����Ը����������ΪCl2��Fe2+�����軯����Һ������ɫ�������������ô���ɫ��Ӧ����֤�Ƿ����Fe2+��
��3�����������¸��ݷ�Ӧ��Ͳ����֪����Ӧ�ٵ����ӷ���ʽΪ3ClO-+2Fe3++10OH-=2FeO42-+3Cl-+5H2O��
��4�����̢ڽ������Һ�����Сʱ�����ã����˻�ôֲ�Ʒ���÷�Ӧ�Ļ�ѧ����ʽΪ2KOH+Na2FeO4 =K2FeO4+2NaOH���������ط�Ӧԭ��������Ӧ�ܷ�����ԭ��K2FeO4���ܽ�ȱ�Na2FeO4С����Һ��K+��FeO42-��Ũ�ȱȽϴ�
��5������K2FeO4��Ʒ���ؽᾧ�����ˡ�ϴ�ӡ�������ýϴ�����K2FeO4��
��2�����ռ�X����Cl2��Ӧ�IJ�����FeCl3��X��FeCl2��Ϊ�������ռ��Ƿ�����ȫת��ΪFeCl3��Һ�������Ը��������Һ������֤����ΪCl-�ܱ����Ը����������ΪCl2��Fe2+�����軯����Һ������ɫ�������������ô���ɫ��Ӧ����֤�Ƿ����Fe2+��
��3�����������¸��ݷ�Ӧ��Ͳ����֪����Ӧ�ٵ����ӷ���ʽΪ3ClO-+2Fe3++10OH-=2FeO42-+3Cl-+5H2O��
��4�����̢ڽ������Һ�����Сʱ�����ã����˻�ôֲ�Ʒ���÷�Ӧ�Ļ�ѧ����ʽΪ2KOH+Na2FeO4 =K2FeO4+2NaOH���������ط�Ӧԭ��������Ӧ�ܷ�����ԭ��K2FeO4���ܽ�ȱ�Na2FeO4С����Һ��K+��FeO42-��Ũ�ȱȽϴ�
��5������K2FeO4��Ʒ���ؽᾧ�����ˡ�ϴ�ӡ�������ýϴ�����K2FeO4��
���
�⣺��1�����Ȼ�������ˮ������ΪFe3+ˮΪFe��OH��3�����Ե�ʣ�ԭ��ΪFe3++3H2O?Fe��OH��3�����壩+3H+��
�ʴ�Ϊ��Fe3++3H2O?Fe��OH��3�����壩+3H+��
�ڹ�ҵ�ϳ���FeCl3��Һ��ʴͭӡˢ��·�壬��Fe3+������Cu��Ե�ʣ�FeCl3��ǿ�������ԣ��ʴ�Ϊ��������
��2�����ռ�X����Cl2��Ӧ�IJ�����FeCl3��X��FeCl2��Ϊ�������ռ��Ƿ�����ȫת��ΪFeCl3��Һ�������Ը��������Һ������֤����ΪCl-�ܱ����Ը����������ΪCl2��Fe2+�����軯����Һ������ɫ�������������ô���ɫ��Ӧ����֤�Ƿ����Fe2+��
�ʴ�Ϊ��FeCl2��10Cl-+2MnO4-+16H+=5Cl2��+2Mn2++H2O��K3[Fe��CN��6]��
��3�����������¸��ݷ�Ӧ��Ͳ����֪����Ӧ�ٵ����ӷ���ʽΪ3ClO-+2Fe3++10OH-=2FeO42-+3Cl-+5H2O��
�ʴ�Ϊ��3ClO-+2Fe3++10OH-=2FeO42-+3Cl-+5H2O��
��4�����̢ڽ������Һ�����Сʱ�����ã����˻�ôֲ�Ʒ���÷�Ӧ�Ļ�ѧ����ʽΪ2KOH+Na2FeO4 =K2FeO4+2NaOH���������ط�Ӧԭ��������Ӧ�ܷ�����ԭ��K2FeO4���ܽ�ȱ�Na2FeO4С����Һ��K+��FeO42-��Ũ�ȱȽϴ�
�ʴ�Ϊ��K2FeO4���ܽ�ȱ�Na2FeO4С����Һ��K+��FeO42-��Ũ�ȱȽϴ�
��5������K2FeO4��Ʒ���ؽᾧ�����ˡ�ϴ�ӡ�������ýϴ�����K2FeO4���ʴ�Ϊ��ϴ�ӣ����
�ʴ�Ϊ��Fe3++3H2O?Fe��OH��3�����壩+3H+��
�ڹ�ҵ�ϳ���FeCl3��Һ��ʴͭӡˢ��·�壬��Fe3+������Cu��Ե�ʣ�FeCl3��ǿ�������ԣ��ʴ�Ϊ��������
��2�����ռ�X����Cl2��Ӧ�IJ�����FeCl3��X��FeCl2��Ϊ�������ռ��Ƿ�����ȫת��ΪFeCl3��Һ�������Ը��������Һ������֤����ΪCl-�ܱ����Ը����������ΪCl2��Fe2+�����軯����Һ������ɫ�������������ô���ɫ��Ӧ����֤�Ƿ����Fe2+��
�ʴ�Ϊ��FeCl2��10Cl-+2MnO4-+16H+=5Cl2��+2Mn2++H2O��K3[Fe��CN��6]��
��3�����������¸��ݷ�Ӧ��Ͳ����֪����Ӧ�ٵ����ӷ���ʽΪ3ClO-+2Fe3++10OH-=2FeO42-+3Cl-+5H2O��
�ʴ�Ϊ��3ClO-+2Fe3++10OH-=2FeO42-+3Cl-+5H2O��
��4�����̢ڽ������Һ�����Сʱ�����ã����˻�ôֲ�Ʒ���÷�Ӧ�Ļ�ѧ����ʽΪ2KOH+Na2FeO4 =K2FeO4+2NaOH���������ط�Ӧԭ��������Ӧ�ܷ�����ԭ��K2FeO4���ܽ�ȱ�Na2FeO4С����Һ��K+��FeO42-��Ũ�ȱȽϴ�
�ʴ�Ϊ��K2FeO4���ܽ�ȱ�Na2FeO4С����Һ��K+��FeO42-��Ũ�ȱȽϴ�
��5������K2FeO4��Ʒ���ؽᾧ�����ˡ�ϴ�ӡ�������ýϴ�����K2FeO4���ʴ�Ϊ��ϴ�ӣ����
���������⿼�������ʵ��Ʊ����̵�����Ӧ�á�ʵ������������������ʵķ���Ӧ�á���Ӧ�ȵļ��㡢�缫����ʽ����д�ȣ���Ŀ�漰��֪ʶ��϶࣬�����ڿ���ѧ����ʵ�������ͶԻ���֪ʶ���ۺ�Ӧ���������������ջ����ǹؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 �Ƹ�С״Ԫ���ֳ������ϵ�д�
�Ƹ�С״Ԫ���ֳ������ϵ�д� �¸��̵�ѧϵ�д�
�¸��̵�ѧϵ�д� ����ͬѧһ����ʦȫ�źþ�ϵ�д�
����ͬѧһ����ʦȫ�źþ�ϵ�д�
�����Ŀ
NAΪ�����ӵ�������ֵ������������ȷ���ǣ�������
| A����⾫��ͭʱ��ÿת��2mol�����������ܽ��ͭԭ����ΪNA |
| B��1.4g�ľ���ϩ��Լ����0.1NA̼ԭ�� |
| C��14gCnH2n�к��еĹ��õ��Ӷ���ΪNA |
| D����״���£�2.24 L�������к����ۼ���ĿΪ0.7NA |
 ͭ���ڲ����ý�������ȴ�ܱ�FeCl3��Һ��ʴ����д�������ӷ�Ӧ����ʽ
ͭ���ڲ����ý�������ȴ�ܱ�FeCl3��Һ��ʴ����д�������ӷ�Ӧ����ʽ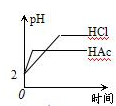
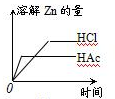
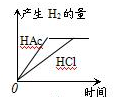
 VIA�������������Se�����ڣ�Te����Ԫ���ڻ������г����ֳ����ֻ��ϼۣ���
VIA�������������Se�����ڣ�Te����Ԫ���ڻ������г����ֳ����ֻ��ϼۣ���


