��Ŀ����
15�������Ѷ���Һ���Ҷ��ԣ����뼡�����������һ���С����������֮�ƣ������й�${\;}_{22}^{18}$Ti��${\;}_{22}^{50}$Ti��˵����ȷ���ǣ�������| A�� | ${\;}_{22}^{18}$Ti��${\;}_{22}^{50}$Tiԭ���о�����22������ | |
| B�� | ${\;}_{22}^{18}$Ti��${\;}_{22}^{50}$Ti�����ڱ���λ����ͬ�����ڵ�4���� | |
| C�� | ${\;}_{22}^{18}$Ti��${\;}_{22}^{50}$Ti������������ͬ | |
| D�� | ${\;}_{22}^{18}$Ti��${\;}_{22}^{50}$TiΪͬһ���� |
���� A������������=������+��������ZAX�е�Z��ʾ��������A��ʾ�������������������
B����������ͬ���������ڱ���λ����ͬ��22��Ԫ���ǵ�4���ڵ�IVB�壻
C��${\;}_{22}^{18}$Ti��${\;}_{22}^{50}$Ti��Ϊͬλ�أ��������ʲ�ͬ��
D�����ص���������������������������
��� �⣺A��ZAX�е�Z��ʾ��������A��ʾ��������������=������+������������2248Ti��2250Tiԭ�����������ֱ�Ϊ26��28����A����
B����������ͬ���������ڱ���λ����ͬ��22��Ԫ���ǵ�4���ڵ�IVB�壬���ڵ�4���У���B��ȷ��
C��${\;}_{22}^{18}$Ti��${\;}_{22}^{50}$Ti��Ϊͬλ�أ��������ʲ�ͬ����ѧ������ͬ����C����
D��2248Ti��2250Ti����������ͬ������������ͬ���Dz�ͬ�ĺ��أ���D����
��ѡB��
���� ���⿼���˺��ء�ͬλ�أ�����������������������֮��Ĺ�ϵ���ѶȲ���ע��������=������+��������

��ϰ��ϵ�д�
�����Ŀ
5����pHΪ3�Ĵ�����Һ��ˮϡ��1000����ϡ�ͺ���Һ��pHΪ��������
| A�� | ����6 | B�� | ����6 | C�� | ��6 | D�� | ����0 |
6��������Ԫ��X��Y��Z��W��Q��Ԫ�����ڱ��е����λ����ͼ��ʾ������˵����ȷ���ǣ������� 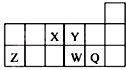

| A�� | ԭ�Ӱ뾶�Ĵ�С˳��Ϊ��rX��rY��rZ��rW��rQ | |
| B�� | ����Y2-��Z3+�ĺ���������͵��Ӳ���������ͬ | |
| C�� | Ԫ��W������������Ӧ��ˮ��������Ա�Q��ǿ | |
| D�� | Ԫ��Y�ĵ��ʱ�Ԫ��X�ĵ��ʸ�������������Ӧ |
3���þ۹��ֵ�Ͳ�������з�ɢϵ�����ܹ۲쵽�����ЧӦ���ǣ�������
| A�� | ������Һ | B�� | Fe��OH��3���� | C�� | ��������Һ | D�� | ��������Һ |
10���йػ�ѧ������ȷ�ģ�������
| A�� | NH3�ĽṹʽΪ��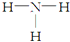 | B�� | NH4Cl�ĵ���ʽ��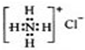 | ||
| C�� | ��ϩ�Ľṹ��ʽ��CH2CH2 | D�� | -CH3�������ĵ���ʽΪ��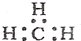 |
20�������뽺����й����ʵ�����������ǣ�������
| A�� | ����±��ʯ����붹�����Ƴ���ζ�ɿڵĶ��� | |
| B�� | ��FeCl3��Һ�м���NaOH��Һ������ֺ��ɫ�����������Ƶ�Fe��OH��3���� | |
| C�� | �ŵ�Ӱʱ����ӳ���䵽��Ļ�ϵĹ��� | |
| D�� | ��ʢ�к��ɫFe��OH��3�����U�ι����˼���ֱ����ѹ������������ɫ���� |
4���ɻ������ֳ�Ƥ��������һ�ּӹ����յ���Ҫԭ���䷽Ϊ�������ʳ�μ��������У��ӷ�ˮ�ܽ⣬������������ʯ�ҳ�ַ�Ӧ����ȴ��õ���Һ���ټ���Ѽ�����գ�
��ش��������⣺
��1����ʯ�Ҽ����ˮ�У�д���û�ѧ��Ӧ�Ļ�ѧ����ʽCaO+H2O=Ca��OH��2��ˮ�ܳ������ַ��ڣ���ԭ���Ƿ�Ӧ���̷��ȣ�
��2�����ú�ȡ�������ϲ���Һ��ϡ�ͺ�μ���ɫ��̪��Һ����ҺӦ��Ϊ��ɫ��
��3��ȡ�ϲ���Һ������һ�����е�����NaOH��NaCl�����ܻ������������ʣ���������Ca��OH��2��Na2CO3ͬʱ���ڣ�������Ca��OH��2+Na2CO3=2NaOH+CaCO3�����û�ѧ����ʽ��ʾ����
��4������Ϊ���ܺ��е�����Na2CO3��Ca��OH��2������ʵ��֤����
��5��Ƥ��ʳ��ʱ����һЩʳ�ף�����ȥ����ζ��ɬ�У�ԭ���Ƿ�������кͷ�Ӧ��
��6����������Ӧ�����У�û���漰���Ļ�ѧ��Ӧ���ͷֽⷴӦ���û���Ӧ��
��ش��������⣺
��1����ʯ�Ҽ����ˮ�У�д���û�ѧ��Ӧ�Ļ�ѧ����ʽCaO+H2O=Ca��OH��2��ˮ�ܳ������ַ��ڣ���ԭ���Ƿ�Ӧ���̷��ȣ�
��2�����ú�ȡ�������ϲ���Һ��ϡ�ͺ�μ���ɫ��̪��Һ����ҺӦ��Ϊ��ɫ��
��3��ȡ�ϲ���Һ������һ�����е�����NaOH��NaCl�����ܻ������������ʣ���������Ca��OH��2��Na2CO3ͬʱ���ڣ�������Ca��OH��2+Na2CO3=2NaOH+CaCO3�����û�ѧ����ʽ��ʾ����
��4������Ϊ���ܺ��е�����Na2CO3��Ca��OH��2������ʵ��֤����
| ʵ�鲽�� | ʵ������ |
��6����������Ӧ�����У�û���漰���Ļ�ѧ��Ӧ���ͷֽⷴӦ���û���Ӧ��
5������ָ�����ӵĸ�����Ϊ1��1���ǣ�������
| A�� | NH4+��������������� | B�� | NH4HSO4�������������������� | ||
| C�� | OH-������������� | D�� | Na2O2���������������� |
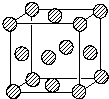 ǰ������ԭ�������������������Ԫ�أ�A��B��C��D��E��H�У�AԪ���������к�����ḻ��BԪ�ػ�̬ԭ�ӵĺ�����3��������ͬ��ԭ�ӹ������ÿ�ֹ���еĵ�����Ŀ��ͬ��DԪ���ǵؿ��к�������Ԫ�أ�EΪd��Ԫ�أ�����Χ�����Ų�����4�ԳɶԵ��ӣ�HԪ�ػ�̬ԭ�������ֻ��һ�����ӣ���������ѳ������ӣ�
ǰ������ԭ�������������������Ԫ�أ�A��B��C��D��E��H�У�AԪ���������к�����ḻ��BԪ�ػ�̬ԭ�ӵĺ�����3��������ͬ��ԭ�ӹ������ÿ�ֹ���еĵ�����Ŀ��ͬ��DԪ���ǵؿ��к�������Ԫ�أ�EΪd��Ԫ�أ�����Χ�����Ų�����4�ԳɶԵ��ӣ�HԪ�ػ�̬ԭ�������ֻ��һ�����ӣ���������ѳ������ӣ�