��Ŀ����
18����֪ X��Y��Z��W ����Ԫ�طֱ���Ԫ�����ڱ������� 3 �������ڵ�Ԫ�أ���ԭ������ ��������X��W ͬ���壬Y��Z Ϊͬ���ڵ�����Ԫ�أ�W ԭ�ӵ����������� Y��Z ԭ���� ��������֮�ͣ�Y �� X �γɵķ������� 3 �����ۼ���Z ԭ�������������Ǵ������� ���� 3 �������ƶϣ���1��Z ���ӵĽṹʾ��ͼ
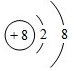 ��
����2��������Ԫ���������γɵĻ������У�����ˮ�Լ��Ե���̬�⻯��ĵ���ʽ Ϊ
 ���������Ӽ����ۼ��Ļ�����Ļ�ѧʽΪNa2O2��
���������Ӽ����ۼ��Ļ�����Ļ�ѧʽΪNa2O2����3���� X��Y��Z ���γɵij������ӻ�������NH4NO3
��д��ѧʽ�����û�������W������������Ӧ��ˮ�����Ũ��Һ����ʱ��Ӧ�����ӷ���ʽΪNH4++OH-$\frac{\underline{\;\;��\;\;}}{\;}$NH3��+H2O��
��4���õ���ʽ��ʾW��Z�γ�W2Z��������γɹ��̣�
 ��
��
���� Zԭ�������������Ǵ�����������3����Zԭ��ֻ����2�����Ӳ㣬����������Ϊ6����ZΪOԪ�أ�Y��X�γɵķ�������3�����ۼ�������Ԫ�طֱ���Ԫ�����ڱ������� 3 �������ڵ�Ԫ�أ�Y��ԭ������С��OԪ�أ���YΪNԪ�ء�XΪHԪ�أ�Wԭ�ӵ�����������Y��Zԭ������������֮�ͣ�Wԭ��������=5+6=11����WΪNaԪ�أ��ݴ˽��н��
��� �⣺Zԭ�������������Ǵ�����������3����Zԭ��ֻ����2�����Ӳ㣬����������Ϊ6����ZΪOԪ�أ�Y��X�γɵķ�������3�����ۼ�������Ԫ�طֱ���Ԫ�����ڱ������� 3 �������ڵ�Ԫ�أ�Y��ԭ������С��OԪ�أ���YΪNԪ�ء�XΪHԪ�أ�Wԭ�ӵ�����������Y��Zԭ������������֮�ͣ�Wԭ��������=5+6=11����WΪNaԪ�أ�
��1��ZΪOԪ�أ������ӵĺ˵����Ϊ8�������������Ϊ10�������ӽṹʾ��ͼΪ��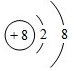 ��
��
�ʴ�Ϊ��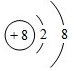 ��
��
��2��������Ԫ�������γɵĻ������У�����ˮ�Լ��Ե���̬�⻯���ǰ����������ĵ���ʽΪ ��
��
�������Ӽ��ͷǼ��Թ��ۼ��Ļ�����Ϊ�������ƣ��������ƵĻ�ѧʽΪ��Na2O2��
�ʴ�Ϊ�� ��Na2O2��
��Na2O2��
��3����X��Y��Z���γɵij������ӻ�������NH4NO3��W������������ˮ����ΪNaOH�����߷�Ӧ�����ӷ���ʽΪ��NH4++OH-$\frac{\underline{\;\;��\;\;}}{\;}$NH3��+H2O��
�ʴ�Ϊ��NH4NO3��NH4++OH-$\frac{\underline{\;\;��\;\;}}{\;}$NH3��+H2O��
��4��W2ZΪNa2O���������������ӻ�����õ���ʽ��ʾ�����Ƶ��γɹ���Ϊ ��
��
�ʴ�Ϊ�� ��
��
���� ���⿼��λ�á��ṹ�����ʹ�ϵ���ۺ�Ӧ�ã��ۺϿ���ѧ�������������������Ŀ�Ѷ��еȣ���ȷ�ƶϸ�Ԫ������Ϊ���ؼ�������ע�����ʽ����д�Լ�������ʵ����ʣ�Ϊ�״��㣮

 �������ͬ������ϵ�д�
�������ͬ������ϵ�д���NH3���ۡ��е��VA������Ԫ���⻯��ĸ�
��С���ӵĴ���������Ժ�ˮ������Ȼ���
�۱����ܶȱ�Һ̬ˮ���ܶ�С
�����ص��ۡ��е�ȴ���ĸ�
��Һ̬������Ļ�ѧʽ��ʱ����д�ɣ�HF��n ����ʽ
��H2O��H2S�ȶ���
| A�� | �٢ڢۢܢݢ� | B�� | �٢ڢۢܢ� | C�� | �٢ڢۢ� | D�� | �٢ڢ� |
| A�� | ��ʽ�ζ��ܿ�������װ���е����������Һ | |
| B�� | ����Na2CO3•10H2O ����ʱӦѡ�����µ�ʯӢ���� | |
| C�� | ʵ������ȡ��������ʱ�ñ���NaHCO3��Һ�ռ����� | |
| D�� | �ⶨ��Һ��pHʱ���ڲ������Ϸ�һƬ�����pH��ֽ���ò�����պȡȡ����Һ�ε�pH��ֽ�ϣ�����ֽ�����ɫ���Ƚϣ�������Һ��pH |
| A�� | ������ͭ��ϡ���ᷴӦ��OH-+H+�TH2O | |
| B�� | ���ۼ���ϡ�����У�2Fe+6H+�T2Fe3++3H2�� | |
| C�� | ̼�������ᷴӦ��CaCO3+2H+�TCa2++CO2��+H2O | |
| D�� | �Ȼ�����Һ�м��������ˮ��Al3++3NH3•H2O�TAl��OH��3��+3NH${\;}_{4}^{+}$ |
| Zn | A | B | C | D |
| ���� | Fe | C��ʯī�� | Ag | Zn |
| ���� | Cu | Fe | Fe | Fe |
| �������Һ | FeCl3 | Fe2 ��SO4��3 | H2SO4 | FeCl3 |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | 1��2 | B�� | 3��1 | C�� | 3��2 | D�� | 4��3 |
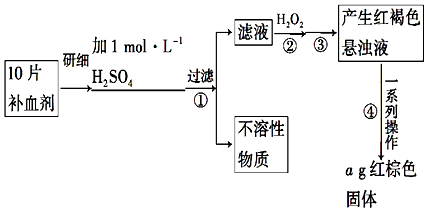
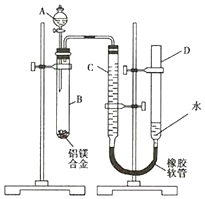 ijѧϰС������ͼװ�òⶨþ���Ͻ�����������������
ijѧϰС������ͼװ�òⶨþ���Ͻ����������������� һλͬѧΪ̽��Ԫ�����ڱ���Ԫ�����ʵĵݱ���ɣ������������ʵ�飮
һλͬѧΪ̽��Ԫ�����ڱ���Ԫ�����ʵĵݱ���ɣ������������ʵ�飮