��Ŀ����
 �����й�֪ʶ�ش��������⣺
�����й�֪ʶ�ش��������⣺��1��B��F��Ԫ���γɵĻ�����Ŀռ乹��Ϊ
��2��CH4��
��3��д��Ԫ��Cu��̬ԭ�ӵĵ����Ų�ʽ
��4��Fe��CO���γɵ�Fe��CO��5�ͻ�����û����ﳣ���³�Һ̬���۵�Ϊ-20.5�棬�е�Ϊ103�棬�����ڷǼ����ܼ����ݴ˿��жϸû����ᄃ������
��5��Zn�����ӵ��������ﲻ����ˮ���������ڰ�ˮ�У���������NH3���ϵ�������Ϊ
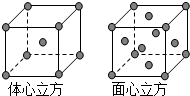
��6������Fe�ľ����ڲ�ͬ�¶��������ֶѻ���ʽ�������ֱ���ͼ��ʾ��������������������������������ʵ�ʺ��е�ԭ�Ӹ���֮��Ϊ
���㣺�����ļ���,�жϼ��ӻ����ӵĹ���,���ȵ���ԭ������Ӧ��,ԭ�ӹ���ӻ���ʽ���ӻ������ж�
ר�⣺��ѧ���뾧��ṹ
��������1����������ԭ�ӵ��Ӷ����ж��ӻ����ͺͿռ乹�ͣ�
��2�����ݵȵ�������ָ������ͬ������Ŀ��ԭ����Ŀ�ķ��ӻ��������ش�
��3������ԭ�ӵ����Ų�����ԭ���Ų���
��4��Fe��CO��5���۷е�ͣ������ڷǼ����ܼ���Ӧ���ڷ��Ӿ��壻
��5��Zn�����ӵ�������������ڰ�ˮ�У��γɵ�����Ϊ[Zn��NH3��4]��OH��2��
��6�����þ�̯�����㾧���к��е�ԭ�Ӹ������������ܶȵ��ھ�������ԭ�ӵ�����������ıȣ�
��2�����ݵȵ�������ָ������ͬ������Ŀ��ԭ����Ŀ�ķ��ӻ��������ش�
��3������ԭ�ӵ����Ų�����ԭ���Ų���
��4��Fe��CO��5���۷е�ͣ������ڷǼ����ܼ���Ӧ���ڷ��Ӿ��壻
��5��Zn�����ӵ�������������ڰ�ˮ�У��γɵ�����Ϊ[Zn��NH3��4]��OH��2��
��6�����þ�̯�����㾧���к��е�ԭ�Ӹ������������ܶȵ��ھ�������ԭ�ӵ�����������ıȣ�
���
�⣺��1��B��F��Ԫ���γɵĻ���������ԭ��B�ĵ��Ӷ���Ϊ3+
=3�����ӻ�����Ϊsp2�ӻ������ӿռ乹��Ϊƽ�������Σ��ʴ�Ϊ��ƽ�������Σ�sp2�ӻ���
��2��NH4+�е�����Ϊ6+4=10��CH4�ǵȵ����壬�ʴ�Ϊ��NH4+��
��3��CuԪ��Ϊ29��Ԫ�أ�ԭ�Ӻ�����29�����ӣ����Ժ�������Ų�ʽΪ��1s22s22p63s23p63d104s1��[Ar]3d104s1���ʴ�Ϊ��1s22s22p63s23p63d104s1��
��4��Fe��CO��5��������ΪҺ̬���۷е�ͣ������ڷǼ����ܼ���Ӧ���ڷ��Ӿ��壬�ʴ�Ϊ�����ӣ�
��5��Zn�����ӵ��������ﲻ����ˮ���������ڰ�ˮ�У���Ӧ����ʽΪZn��OH��2+4NH3=[Zn��NH3��4]��OH��2��[Zn��NH3��4]��OH��2�Ƿ������������������γɵ������Ϊ��λ�����ʴ�Ϊ����λ����
��6������������������ԭ�Ӹ���=1+8��
=2����������������ʵ�ʺ��е�Feԭ�Ӹ���=6��
+8��
=4����������������������������������ʵ�ʺ��е�Feԭ�Ӹ���֮��2��1���ʴ�Ϊ��2��1��
| 3-1��3 |
| 2 |
��2��NH4+�е�����Ϊ6+4=10��CH4�ǵȵ����壬�ʴ�Ϊ��NH4+��
��3��CuԪ��Ϊ29��Ԫ�أ�ԭ�Ӻ�����29�����ӣ����Ժ�������Ų�ʽΪ��1s22s22p63s23p63d104s1��[Ar]3d104s1���ʴ�Ϊ��1s22s22p63s23p63d104s1��
��4��Fe��CO��5��������ΪҺ̬���۷е�ͣ������ڷǼ����ܼ���Ӧ���ڷ��Ӿ��壬�ʴ�Ϊ�����ӣ�
��5��Zn�����ӵ��������ﲻ����ˮ���������ڰ�ˮ�У���Ӧ����ʽΪZn��OH��2+4NH3=[Zn��NH3��4]��OH��2��[Zn��NH3��4]��OH��2�Ƿ������������������γɵ������Ϊ��λ�����ʴ�Ϊ����λ����
��6������������������ԭ�Ӹ���=1+8��
| 1 |
| 8 |
| 1 |
| 6 |
| 1 |
| 8 |
���������⿼���˾������㡢�ȵ���ԭ�����ӻ���ʽ�����ݣ��ѶȽϴ����ڿ���ѧ���������⣬��������������

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
������Һ�������ɺ����������ʹ�����ǣ�������
| A��AlCl3 |
| B��KHCO3 |
| C�������� |
| D��NH4HCO3 |
ˮ��ҺX��ֻ��������K+��Ca2+��AlO2-��SiO32-��CO32-��SO42-�е����������ӣ�ijͬѧ�Ը����н���������ʵ�������ж���ȷ���ǣ�������


| A�������һ���Ǵ����� |
| B���������ǹ������ƵĻ���� |
| C��K+��AlO2-��SiO32-����CO32-һ����������ҺX�� |
| D��SO42-һ������������ҺX�� |
��ͼΪˮ����ص�ʾ��ͼ������˵����ȷ���ǣ�������


| A��пƬ���������������� |
| B��������ͭƬ�ص�������пƬ |
| C��ͭ�缫���ܽ� |
| D����װ���ܽ�����ת��Ϊ��ѧ�� |
���и�����������ͬһ��Һ�����������ǣ�������
| A��Na+��Al3+��Cl-��AlO2- |
| B��Mg2+ NO3- Fe2+��H+ |
| C��Fe3+��SO42-��Na+��SCN- |
| D��K+��Na+��AlO2-��CO32- |
 ��һ��������Mg��Al�Ļ����Ͷ��500mLϡ�����У�����ȫ���ܽⲢ�������壮����Ӧ��ȫ����������Һ�м���NaOH��Һ�����ɳ��������ʵ��������NaOH��Һ�������ϵ��ͼ��ʾ��
��һ��������Mg��Al�Ļ����Ͷ��500mLϡ�����У�����ȫ���ܽⲢ�������壮����Ӧ��ȫ����������Һ�м���NaOH��Һ�����ɳ��������ʵ��������NaOH��Һ�������ϵ��ͼ��ʾ��