��Ŀ����
20��ij��ѧ��ȤС��ⶨijFeCl3��Ʒ��������FeCl2���ʣ�����Ԫ�ص�����������ʵ���Ұ����²�����У��ٳ���a g��Ʒ�������ձ��У�
�ڼ��������������������ˮ��ʹ��Ʒ�ܽ⣬Ȼ��ȷ���Ƴ�250mL��Һ��
��ȷ��ȡ25.00mL���������õ���Һ�������ձ��У�������������ˮ������ʹ��Ӧ��ȫ��
�ܳ���Ѹ�ټ��������ˮ����ֽ��裬ʹ������ȫ��
�ݹ��ˡ�ϴ�ӳ�����
������ת�Ƶ������ڣ����ȡ����裬ֱ������ȫ���ɺ��ɫ��Ϊ����ɫ���ڸ���������ȴ�����º�����
�ߡ�
����������������ش�
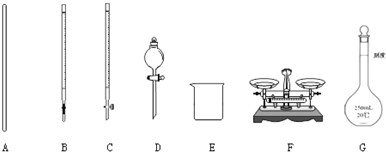
��1����ͼ��ʾ�����У���ʵ�鲽��٢ڢ��б����õ���������E��A��C��F��G������ĸ����
��2��д��������з�����Ӧ�����ӷ���ʽ2Fe2++Cl2=2Fe3++2Cl-����
��3��ϴ����ϴȥ�����ڳ����ϵ�NH4+��Cl-��OH-��д���ӷ��ţ���ϴ�ӳ����IJ�������©����ע������ˮ��ʹˮ��û����������ˮ��Ȼ������ظ�����2��3�Σ�
��4�����ȷ�������Ƿ�ϴ�Ӹɾ��IJ�����ȡ���һ��ϴ��Һ��������������Һ�����û�г������ɣ���֤��ϴ�Ӹɾ���
��5��������������W1����������Ⱥ������������W2������Ʒ����Ԫ�ص���������Ϊ��$\frac{��{W}_{2}-{W}_{1}��g��\frac{112}{160}��\frac{250}{25}}{ag}$��100%�����г���ʽ�����軯��
��6������ȤС���м�ѧ����Ϊ��ʵ�鲽����в�������ˮ�����ಽ�費�䣬�Կɴﵽʵ��Ŀ�ģ�����Ϊ��ѧ���Ĺ۵��Ƿ���ȷ����ȷ������ȷ�������˵�����ɣ�FeCl3��FeCl2��Һ�м��������ˮ������Fe��OH��3��Fe��OH��2�������ڲ���������Fe��OH��2�ᱻ������һ����������Fe��OH��3���������������ֻ��Fe2O3����ʵ������Ӱ�죬�ʼ�ͬѧ�Ĺ۵���ȷ��
���� ��1�����ݳ������ܽ⡢����һ�����ʵ���Ũ�ȵ���Һ��ȷ��ȡ��Һ�Ȳ���ȷ������������
��2��������ˮ�ܽ�FeCl2��������FeCl3��
��3��������Һ�к��а�ˮ���Ȼ����ȷ�����ӣ�����ϴ�ӳ����ķ�����
��4����Ϊ�����к���Cl-���ݴ˼�������Ƿ�ϴ�Ӹɾ���
��5�����������������պ�����������������������W1����������Ⱥ������������W2����������������Ϊ��W2-W1��g������������Ԫ�ص�����������Ʒ����Ԫ�ص�����������������Ԫ�ص�����������
��6������FeCl3��FeCl2��Һ�м��������ˮ������Fe��OH��3��Fe��OH��2�������ڲ���������Fe��OH��2�ᱻ������һ����������Fe��OH��3���������������ֻ��Fe2O3��
��� �⣺��1������ʱ��Ҫ��ƽ��Կ�ף��ܽ�ʱ��Ҫ�ձ���������������һ�����ʵ���Ũ�ȵ���Һʱ��Ҫ��Ͳ���ձ�������������ͷ�ιܡ�һ����������ƿ��
ȷ��ȡ��Һ��Ҫ��ʽ�ζ��ܡ�ϴ����
�ʴ�Ϊ��A��C��F��G��
��2����ˮ�ܽ�FeCl2��������FeCl3�����ӷ���ʽΪ��2Fe2++Cl2=2Fe3++2Cl-���ʴ�Ϊ��2Fe2++Cl2=2Fe3++2Cl-��
��3����Һ�к��а�ˮ���Ȼ�泥����������У�NH4+��Cl-��OH-��ϴ�ӳ����ķ�������©����ע������ˮ��ʹˮ��û����������ˮ��Ȼ������ظ�����2��3�Σ�
�ʴ�Ϊ��NH4+��Cl-��OH-����©����ע������ˮ��ʹˮ��û����������ˮ��Ȼ������ظ�����2��3�Σ�
��4����Ϊ�����к���Cl-��ȷ�������Ƿ�ϴ�Ӹɾ��IJ����ǣ�ȡ���һ��ϴ��Һ��������������Һ�����û�г������ɣ���֤��ϴ�Ӹɾ���
�ʴ�Ϊ��ȡ���һ��ϴ��Һ��������������Һ�����û�г������ɣ���֤��ϴ�Ӹɾ���
��5����Ԫ�������غ㣬������ɫ�����е���������Ʒ������Fe2O3����Ԫ�ص�����Ϊ��W2-W1��g��$\frac{112}{160}$����Ʒ����Ԫ�ص�������W2-W1��g��$\frac{112}{160}$��$\frac{250}{25}$����Ʒ����Ԫ�ص���������$\frac{��{W}_{2}-{W}_{1}��g��\frac{112}{160}��\frac{250}{25}}{ag}$��100%��
�ʴ�Ϊ��$\frac{��{W}_{2}-{W}_{1}��g��\frac{112}{160}��\frac{250}{25}}{ag}$��100%��
��6����FeCl3��FeCl2��Һ�м��������ˮ������Fe��OH��3��Fe��OH��2�������ڲ���������Fe��OH��2�ᱻ������һ����������Fe��OH��3���������������ֻ��Fe2O3���Կɴﵽʵ��Ŀ�ģ��ʼ�ͬѧ�Ĺ۵���ȷ��
�ʴ�Ϊ����ȷ��FeCl3��FeCl2��Һ�м��������ˮ������Fe��OH��3��Fe��OH��2�������ڲ���������Fe��OH��2�ᱻ������һ����������Fe��OH��3���������������ֻ��Fe2O3����ʵ������Ӱ�죬�ʼ�ͬѧ�Ĺ۵���ȷ��
���� ������Ҫ����ѧ����Һ�����ơ���ʵ��ԭ�����⡢��ʵ����������ۡ����ʺ����IJⶨ����ѧ����ȣ���Ŀ�Ѷ��еȣ���Ҫѧ���߱���ʵ�Ļ���֪ʶ��������⡢�������������

 ��ĩ���100�ִ��½����ȫ�Ծ�ϵ�д�
��ĩ���100�ִ��½����ȫ�Ծ�ϵ�д�
| A�� | X��Y��ɵ����ֻ����������������ӵĸ����Ⱦ�Ϊ1��2 | |
| B�� | W��R����Ԫ�ض�Ӧ�ļ����Ӷ���Ӱ��ˮ�ĵ���ƽ�� | |
| C�� | ��X��Y��Z����Ԫ����ɵĻ������ˮ��Һ�Լ��� | |
| D�� | ��ҵ���õ�����ڵ�Z��X��ɵĻ���������ȡZ�ĵ��� |
| A�� | CH4+Cl2 $\stackrel{����}{��}$CH2Cl2+H2 | |
| B�� | 2CH3CH2OH+O2 $��_{��}^{����}$2CH3CHO+H2O | |
| C�� | CH3CH2OH+CH3COOH $\stackrel{Ũ����}{��}$CH3COOCH2CH3 | |
| D�� | H2C�TCH2+Br2��CH3CHBr2 |
| A�� | AgCl���������ɺ��ܽⲻ�Ͻ��У���������� | |
| B�� | AgCl������ˮ����Һ��û��Ag +��Cl - | |
| C�� | �����¶ȣ�AgCl���ܽ�Ȳ��� | |
| D�� | ��AgCl�������ܽ�ƽ����ϵ�м���NaCl���壬AgCl���ܽ�Ȳ��� |
 ��ˮ����Ҫ���ӵĺ������£�
��ˮ����Ҫ���ӵĺ������£�| �ɷ� | ����/��mg/L�� | �ɷ� | ����/��mg/L�� |
| Cl- | 18980 | Ca2+ | 400 |
| Na+ | 10560 | HCO3- | 142 |
| SO42- | 2560 | Mg2+ | 1272 |
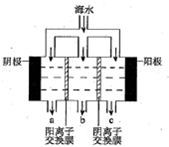
��2����������������ˮʾ��ͼ��ͼ��ʾ�����������������ӽ���Ĥ������������������ͨ���������ϲ�����������������������������ɫ��������ɷ���Mg��OH��2��CaCO3������CaCO3�����ӷ���ʽ��Ca2++OH-+HCO3-=CaCO3��+H2O��
��3���ú�ˮ��ͬʱ�����Ȼ��ƺͽ���þ��þ�Ļ��������������ͼ��ʾ��
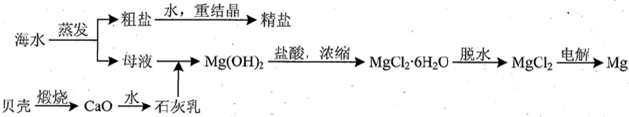
����ʵ�������ɴ��Ρ��ؽᾧ���ƾ��εIJ��������ܽ⡢���ˡ�������ϴ�ӵȲ��裻�й����С������������������ȷ����bd��
a��������Ŀ���ǵõ��ȱ�����Һ
b��������Ŀ������������
c��Ӧ������������Һ
d��Ӧ�������н϶ྦྷ������ʱΪֹ
����MgCl2��Һ�õ�MgCl2•6H2O����ʱ��Ҳ��Ҫ������������Ŀ���ǵõ��ȱ�����Һ���ж���Һ�ѱ��͵���������Һ�����γɾ�Ĥ��
��4��25��ʱ������Mg��OH��2��Һ��Ũ��Ϊ5��10-4 mol/L��
�ٱ���Mg��OH��2��Һ�еμӷ�̪����������Һ��죮
��ijѧϰС��⺣ˮ��Mg2+������mg/L���ķ����ǣ�ȡһ������ĺ�ˮ�������������ᣬ�ټ�������NaOH����Mg2+תΪMg��OH��2��25�棬�÷�����õ�Mg2+���������1272mg/L�ġ���ֵ���ȣ�������ԼΪ-5.6%[����2λС������ˮ�б���Mg��OH��2��Һ���ܶȶ���l g/cm3��]��
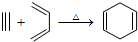 �����Ҫ�ϳ�
�����Ҫ�ϳ� ���õ�ԭʼԭ�Ͽ����ǣ�������
���õ�ԭʼԭ�Ͽ����ǣ�������| A�� | 2-��-1��3-����ϩ��2-��Ȳ | B�� | 1��3-���ϩ��2-��Ȳ | ||
| C�� | 2��3-����-1��3-���ϩ����Ȳ | D�� | 2��3-����-1��3-����ϩ��1-��Ȳ |
 ������ԭ��Ӧ�������������о��й㷺����;���ᴩ�Ž�
������ԭ��Ӧ�������������о��й㷺����;���ᴩ�Ž�
 ��
��