��Ŀ����
��1����֪�����Ȼ�ѧ����ʽ��
�� H2(g)��1/2O2(g) ��H2O(g) ��H����241.8 kJ/mol
�� C(s)��1/2O2(g) ��CO(g) ��H����110.5 kJ/moL
��ˮú����Դ����ȡԭ����C(s)��H2O(g)��H2(g)��CO (g) ��H�� kJ/moL
��2����ͼ��ʾ��ѧ��Ӧ��ϵ�з�Ӧ�����������ͻ�ܵĹ�ϵ�����ϼ�ͼ�ķ���ʽΪ������������ͼ�ķ���ʽΪ����������д��A������B������C������D��ѡ�

A��CO(g)+H2O(g)=CO2(g)+H2(g) B��H2(g)+I2(g)=2HI(g)
C��H+(aq)+OH-(aq)=H2O D��CaCO3(s)=CaO(s)+CO2(g)
 ��3����ͼ��ʾ���������о�ΪCu(NO3)2��Һ���ס������ص缫���϶���������ʯī������Ӧһ��ʱ�����ش��������⣺
��3����ͼ��ʾ���������о�ΪCu(NO3)2��Һ���ס������ص缫���϶���������ʯī������Ӧһ��ʱ�����ش��������⣺
���к�ɫ�����������Ǽ׳��е�______�����ҳ��е�_______����
���ҳ��������ĵ缫��Ӧʽ��___________________________��
��4����֪25��ʱAgCl���ܶȻ�Ϊ1.77��10-10������AgCl�ı�����Һ��c(Ag+)Ϊ ������֪![]() =1.33��
=1.33��
��1����131.3 ��1�֣� ��2�� C��1�֣� B ��1�֣�
��3����ʯī��1�֣� ������1�֣� ����4OH����4e����2H2O��O2����2�֣�
��4��1.33��10��5![]() ��1�֣�
��1�֣�

 ��ʱѵ���������������ϵ�д�
��ʱѵ���������������ϵ�д� ��Դ����������ͷ�չ����Ҫ֧�����о�����Ч�ؿ�������Դ����Դ��ȱ�Ľ��������Ҫ���������壮
��Դ����������ͷ�չ����Ҫ֧�����о�����Ч�ؿ�������Դ����Դ��ȱ�Ľ��������Ҫ���������壮
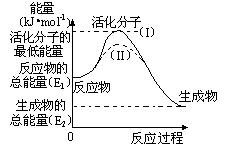

 ����ʽ��H2(g)��1/2O2(g) �� H2O(g) ��H����
����ʽ��H2(g)��1/2O2(g) �� H2O(g) ��H���� 241.8 kJ/mol���÷�Ӧ�Ļ��Ϊ167.2 kJ/mol�������淴Ӧ�Ļ��Ϊ kJ/mol��
241.8 kJ/mol���÷�Ӧ�Ļ��Ϊ167.2 kJ/mol�������淴Ӧ�Ļ��Ϊ kJ/mol��