��Ŀ����
��13�֣�ijǿ������ҺX����Ba2+��Al3+��NH��4��Fe2����Fe3+��CO2��3��SO2��3��SO2��4��Cl����NO��3�е�һ�ֻ��֣�ȡ����Һ��������ʵ�飬ʵ���������£�
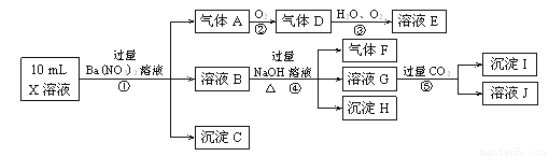
����������Ϣ���ش��������⣺
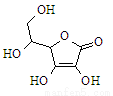
��1����Ӧ�۵Ļ�ѧ����ʽΪ_________________________________________________��
��2������C��H������F�Ļ�ѧʽ�ֱ�Ϊ___________________________________��
��3��д���йط�Ӧ�����ӷ���ʽ��
��������A________________________________________________________��
��_______________________________________________________________��
��4�����������У���ҺX�п϶������е�������________________________________�����ܺ��е�������________________________________��
��1��4NO2+O2+2H2O=4HNO3��
��2��Ba SO4��Fe(OH)3��NH3��
��3��3 Fe2++NO3-+4H+=3 Fe3++NO��+2 H2O��AlO2-+CO2+2H2O=Al(OH)3��+HCO3-��
��4��NO3-��Ba2+��CO32-��SO32-��Cl-��Fe3+��
��������
���������������Һ�в����ܴ�������CO32-��SO32-������������ᱵ��Һ������Һ�к������ᣬ���������ɣ�˵��������������Ļ�ԭ����NO����ԭ��Һ�к����ᷢ��������ԭ��Ӧ��ֻ����Fe2+����ԭ��Һ�к���Fe2+������NO3-�����ó���C�����ᱵ��������ԭ��Һ�д���SO42-��һ������Ba2+,��ҺB�м����������������Һ������������������F�ǰ�����ԭ��Һ�к���NH4+����ҺB�к��������ӣ����Գ���H����������������ͨ�������̼���������������ԭ��Һ�л�Ӧ����Al3+������I�������������������ܺ���Cl-��Fe3+���������Ϸ�����
��1����Ӧ����NO��������Ӧ���ɵĶ�����������������ˮ��Ӧ�������ᣬ��ѧ����ʽΪ4NO2+O2+2H2O=4HNO3��
��2������C��H������F�Ļ�ѧʽ�ֱ�ΪBa SO4��Fe(OH)3��NH3��
��3����������A�����ӷ���ʽΪ3 Fe2++NO3-+4H+=3 Fe3++NO��+2 H2O����������������������Ʒ�Ӧ����ƫ�����ƣ�ƫ��������Һ���������̼��Ӧ������������������̼��������ӣ����Ԣݵ����ӷ���ʽΪAlO2-+CO2+2H2O=Al(OH)3��+HCO3-��
��4��X��һ������NO3-��Ba2+��CO32-��SO32-�����ܺ���Cl-��Fe3+��
���㣺������Һ�����ӵ��жϣ����ӷ���ʽ����д

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д���������������Һ��ÿ�����������Һ����A��B��C˳�����У�
| �� | �� | �� | �� |
A | BaCl2 | Na2CO3 | KCl | K2CO3 |
B | ���� | ���� | ���� | ���� |
C | Na2CO3 | AgNO3 | K2CO3 | BaCl2 |
��������в��������۲������ش�
��CaCl2��Һ�м���A��Һ������ɫ�������������������B��Һ������ʧ����������ɫ��ζ�����壻�ټ���������C��Һ�������ɰ�ɫ������
��1���������������У�ѡ���ˡ�����������Һ��
��2�����ݷ�Ӧ��������д���йط�Ӧ�Ļ�ѧ����ʽ��
�١� ��
�ڡ� ��
�ۡ� ��
��������ɸ�������ɺ����ʽ��з��ࣺ
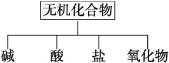
��1����ͼ��ʾ�����ʷ���������ǡ� ��
��2����Na��K��H��O��C��S��N�������ֻ�����Ԫ����ɺ��ʵ����ʣ��ֱ������±��ڢۢĺ��档
���� ��� | �� | �� | �� | ������ |
��ѧʽ | ��HCl ��____ | ��______ ��Ba(OH)2 | ��Na2CO3 ��_____ | ��CO2 ��Na2O2 |
��3��д����ת��Ϊ�ݵĻ�ѧ����ʽ
�� ��
��4��ʵ�����Ʊ��߳��á����������������� ��Ӧ�����������ķ����� �� ��