��Ŀ����
10����ͼ��ʾ���ü״��������Լ�ǿ�����������Һ������ȼ�ϵ������Դ����A��B װ��ͨ��һ��ʱ�������3.2g�״��μӷ�Ӧ�Ң۵缫���أ����蹤��ʱ��������ʧ������ش��������⣺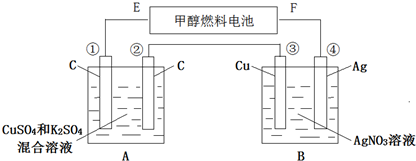
��1��F�缫��������������E�缫�ķ�Ӧʽ��CH3OH+8OH--6e-�TCO32-+6H2O��
��2���ܵ缫�ķ�Ӧʽ��Ag-e-=Ag+��ͨ��һ��ʱ���B��pH���䣨������С�������䡱����
��3����A����ԭ���Һ�����Ϊ500mL��CuSO4��K2SO4Ũ�Ⱦ�Ϊ0.3mol/L����������A���й��ռ�����״���µ�����6.72L���١��۵缫�����������ʵ���֮��1��4��
��4����ʹͨ������Һ�ָ���ԭ״���ɼ���һ��������������ͭ��д���ƣ���
���� ��A��Bװ��ͨ��һ��ʱ�������3.2g�״��μӷ�Ӧ�Ң۵缫���أ���˵���۵缫�������ӵõ���������������ǵ������������Ԣܢ�������������������ԭ�����E�Ǹ�����F��������ԭ��ظ����ϼ״�ʧ���ӷ���������Ӧ������������ʧ���ӷ���������Ӧ�������ϵõ��ӷ�����ԭ��Ӧ��
��1��E�Ǹ�����F�������������ϼ״�ʧ��������̼������ӣ�
��2�������У����������ӵõ������������ʣ�Bװ���������������ӵõ��ӣ�������Agʧ�������������ӣ���Һ��������Ũ�Ȳ��䣻
��3���������������ϵ�ʧ�����غ���㣻
��4��A�е�����ɵ����ʣ�������������������������������Cu��������Һʧȥ��Ԫ�ط�����
��� �⣺��1����A��Bװ��ͨ��һ��ʱ�������1.6g�״��μӷ�Ӧ�Ң۵缫���أ���˵���۵缫�������ӵõ���������������ǵ������������Ԣܢ�������������������ԭ�����E�Ǹ�����F�������������ϼ״�ʧ��������̼������ӣ����缫����ʽΪ��CH3OH+8OH--6e-�TCO32-+6H2O��
�ʴ�Ϊ��������CH3OH+8OH--6e-�TCO32-+6H2O��
��2�������У����������ӵõ������������ʣ��ܵ缫����ʧ�������������ӽ�����Һ���缫��ӦʽΪ��Ag-e-=Ag+��Bװ���������������ӵõ��ӣ�������Agʧ�������������ӣ���Һ��������Ũ�Ȳ��䣬������Һ��pH���䣻
�ʴ�Ϊ��Ag-e-=Ag+�����䣻
��3����3.2g�״���Ӧת�Ƶ��ӵ����ʵ�����x��
CH3OH+8OH--6e-�TCO32-+6H2O ת�Ƶ���
32g 6mol
3.2g x
x=$\frac{3.2g��6mol}{32g}$=0.6mol
��A����ԭ���Һ�����Ϊ500mL��CuSO4��K2SO4Ũ�Ⱦ�Ϊ0.3mol/L����ͭ���ӵ����ʵ���Ϊ0.3mol/L��0.5L=0.15mol����ͭ������ȫ����ʱ��ͭ���ӵõ����ӵ����ʵ���=0.15mol��2=0.3mol��0.6mol�����Ե������Һʱ��������������ͭ��������������������������������ʧ���ӵ����ʵ�������0.6mol��
���������������������ʵ�����y���������������������ʵ�����z��
��0.15mol��2+2y=4z=0.6mol��y=0.15mol��z=0.15mol������A���й��ռ�������������ʵ����ǣ�0.15+0.15��mol=0.3mol�����������=0.3mol��22.4L/mol=6.72L������ͭ���ӵõ�����������ͭ��ͭ���ӵ����ʵ���Ϊ0.3mol/L��0.5L=0.15mol�����������ӵõ������������ʣ����ݵ����غ㣬ת��0.6mol����������������0.6mol�������缫�����������ʵ���֮��Ϊ1��4��
�ʴ�Ϊ��6.72�� 1��4��
��4��A�е�����ɵ����ʣ�������������������������������Cu��������ʹͨ������Һ�ָ���ԭ״���ɼ���һ��������������ͭ��
�ʴ�Ϊ��������ͭ��
���� ���⿼����ԭ��غ͵���ԭ����ע�⣨4�����У������ϲ�������ͭ���������������������ϵõ�����������ԭ�����ת�Ƶ��������м��㣬�Ӷ��ó���������������Ϊ�״��㣬�����ڿ���ѧ���ķ��������ͼ�����������Ŀ�Ѷ��еȣ�

��1����һ�ݼ����������ᣬ���κ����������
��2���ڶ��ݼ�������NaOH��Һ�������衢���ˡ�ϴ�ӡ����ա����õ�����ɫ���壻
��3�������ݵμ�0.1mol��L-1����KMnO4��Һ��KMnO4��Һ����ɫ��ʧ
��4�����ýྻ�IJ�˿պȡ�û����Һ����dzɫ���������գ����ֻ�����ֻ�ɫ��
��������ʵ�飬����˵����ȷ���ǣ�������
| A�� | ԭ�����Һ��ֻ����Na+��Fe3+��SO42-�������ܴ���K+��CO32- | |
| B�� | ��ʵ�飨1�����ƶ�ԭ�����Һ���Ƿ���SO42- | |
| C�� | ��ʵ�飨2�����ƶ�ԭ�����Һ���Ƿ���Fe3+ | |
| D�� | ��ʵ�飨3�����ƶ�ԭ�����Һ�д���Fe2+ |
| A�� | HCl | B�� | NaOH | C�� | Na2SO4 | D�� | NaCl |
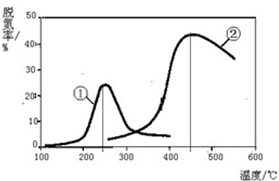 ���붬��������ʼ��ů�����������������أ�����PM2.5��PM10������������������������������Ⱦ���У���Ϊ�������ǻ�����β���еĵ��������ȼú������������
���붬��������ʼ��ů�����������������أ�����PM2.5��PM10������������������������������Ⱦ���У���Ϊ�������ǻ�����β���еĵ��������ȼú����������������֪��ӦN2O4��g��?2NO2��g����H�����¶����ߣ�����������ɫ�����һ����N2O4��������������һ��ʱ����о�ѹ����������������л���������������ʣ�������ɫԽdz������Խ����ʱ��仯���������˵����˵�����ʲ��ٷ����ı����ad��
a��������ɫ���ٸı� b����H���ٸı�
c��v����N2O4��=2v����NO2�� d��N2O4��ת���ʲ��ٸı�
����NH3����ԭNOx�������������������Ⱦ������ͼ������NH3����ԭ����������һ��������ͨ�����ֲ�ͬ�����������ݳ������е������ﺬ�����Ӷ�ȷ�������ѵ��ʣ�ע���ѵ��ʼ���������ת���ʣ�����Ӧԭ��Ϊ��
NO��g��+NO2��g��+2NH3��g��?2N2��g��+3H2O��g����
��1���÷�Ӧ�ġ�S��0�����������=����������
��2������˵����ȷ����C��
A���ڢ��ִ����ȵڢ��ִ����ѵ��ʸ�
B����ͬ�����£��ı�ѹǿ���ѵ���û��Ӱ��
C�������١��ڷֱ��ʺ���250���450�������ѵ�
����CH4����ԭNOxҲ�������������������Ⱦ��
��3����֪��CH4��g���ı�ȼ����Ϊ-890kJ/mol������1mol H2O��l����Ҫ����44kJ������
CH4��g��+4NO��g���T2N2��g��+CO2��g��+2H2O��g����H=-1114kJ/mol
2NO��g��+O2��g���T2NO2��g����H=-114kJ/mol
д��CH4����ԭNO2��g������N2��H2O��g�����Ȼ�ѧ����ʽ��CH4��g��+2NO2��g��=N2��g��+CO2��g��+2H2O��g����H=-844kJ/mol��
��4�����¶�ΪT1���T2��ʱ���ֱ�0.5mol CH4��1.2mol NO2�������Ϊ1L���ܱ������У����NO2�����ʵ�����ʱ��仯�������±���
ʱ��/min �¶�/�� | 0 | 10 | 20 | 40 | 50 |
| T1 | 1.2 | 0.9 | 0.7 | 0.4 | 0.4 |
| T2 | 1.2 | 0.8 | 0.56 | �� | 0.5 |
��T1��T2��������������¿�ͬ�����ж������������¶ȣ�NO2�����ʵ�������ƽ�������ƶ�������ӦΪ���ȷ�Ӧ��
��T1��ʱ����ӦCH4��g��+2NO2��g��?N2��g��+CO2��g��+2H2O��g��ƽ�ⳣ��K=6.4��
���¶�ΪT2��ʱ����ƽ������������м���0.5mol CH4��1.2mol NO2������ƽ��ʱCH4��ת���ʽ���С�����������С�����䡱����
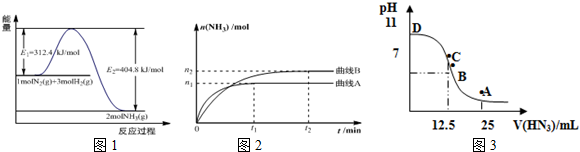
��1��ͼ1�Ǻϳɰ���Ӧ�������뷴Ӧ�������ͼ��δʹ�ô�������ͼ2�Ǻϳɰ���Ӧ��2L�����С���ͬͶ������¡���������������ʱ��ijһ��Ӧ�����ĸı�Է�Ӧ��Ӱ��ͼ��
������˵����ȷ����BDE��
A��ʹ�ô�����ʹE1����ֵ����
B����H=-92.4kJ•mol-1
C��Ϊ�����ת���ʣ���ҵ�����з�Ӧ���¶�Խ��Խ��
D��������A�����£���Ӧ�ӿ�ʼ��ƽ�⣬����N2��ƽ������Ϊ$\frac{{n}_{1}}{4{t}_{1}}$mol•L-1•min-1
E���÷�Ӧ��ƽ�ⳣ��KA��KB
F��ͼ2�Dz�ͬѹǿ�·�Ӧ��ϵ�а������ʵ����뷴Ӧʱ���ϵͼ����PA��PB
��������˵���÷�Ӧ�ﵽƽ��״̬����D
A��������N2��H2��NH3��Ũ��֮��Ϊ1��3��2
B��v����N2��=v����H2��
C�����������ܶȱ��ֲ���
D��������ѹǿ���ֲ���
��2���ϳɰ������Ĵ����������˹��̵�����Ҫ;�����ϳɰ���Ӧ��ƽ�ⳣ��Kֵ���¶ȵĹ�ϵ���£�
| �¶�/�� | 200 | 300 | 400 |
| K | 1.0 | 0.86 | 0.5 |
��3����������25mL0.1mol/LNH3•H2O ��Һ�У���μ��� 0.2mol/L��HN3��Һ����Һ��pH�仯������ͼ3��ʾ��
A��B��C��D �ĸ����У�ˮ�ĵ���̶�������C��A��ʱ��Һ�и�����Ũ����С�����˳��Ϊc��OH-����c��H+����c��NH4+����c��N3-����
��4����������Ѱ����ʵĴ����͵缫���ϣ���N2��H2Ϊ�缫��Ӧ���HCl-NH4ClΪ�������Һ��������ȼ�ϵ�أ���д���õ�ص�������ӦʽN2+8H++6e-�T2NH4+��
| A�� | CO2 | B�� | CaO | C�� | Na2O2 | D�� | Al2O3 |
| A�� | ʳ�κ�ʳ�� | B�� | ʳ�κʹ��� | C�� | �����ʳ�� | D�� | ȫ�� |