��Ŀ����
1���±��Dz��ֶ�����Ԫ�ص�ԭ�Ӱ뾶����Ҫ���ϼۣ�| Ԫ�ش��� | W | R | X | Y | Z | Q |
| ԭ�Ӱ뾶/nm | 0.037 | 0.186 | 0.074 | 0.075 | 0.077 | 0.110 |
| ��Ҫ���ϼ� | +1 | +1 | -2 | -3��+5 | +2��+4 | -3��+5 |
��1������Ԫ����ԭ�Ӱ뾶����Ԫ�������ڱ���λ���ǵ�������IA�壮
��2��Y���⻯���Q���⻯��е�ߵ�ԭ����NH3�ķ��Ӽ���������
��3��X����R��1��1��ԭ�Ӹ������γɻ�����ף����д��ڵĻ�ѧ�������Ӽ��ͷǼ��Լ��������Ӽ������ۼ���X����W��ɺ�18e-�Ļ������ң����ҵĵ���ʽΪ
 ��X����Z���һ���ж��ζ����������������и�ԭ������������8e-�ṹ���Һ��зǼ��Լ���1mol���������2mol������ȫ��Ӧ������һ�����壬�ҷ�Ӧǰ���������������䣬����������ʹ����ʯ��ˮ����ǣ���д�������ӵĽṹʽO=C=C=C=O��
��X����Z���һ���ж��ζ����������������и�ԭ������������8e-�ṹ���Һ��зǼ��Լ���1mol���������2mol������ȫ��Ӧ������һ�����壬�ҷ�Ӧǰ���������������䣬����������ʹ����ʯ��ˮ����ǣ���д�������ӵĽṹʽO=C=C=C=O��
���� XԪ��ֻ��-2�ۣ�Ϊ��Ԫ�أ�Y��Q��-3��+5�ۣ�ԭ�Ӱ뾶Q�ϴ�YΪ��Ԫ�ء�QΪ��Ԫ�أ�W��R��+1�ۣ�W��ԭ�Ӱ뾶��С��С����ԭ�ӣ���WΪ��Ԫ�أ�R��ԭ�Ӱ뾶�ܴ�Ϊ��Ԫ�أ�Z��+2��+4�ۣ�ԭ�Ӱ뾶��N��Oԭ������ZΪ̼Ԫ�أ��ɴ˷������
��� �⣺XԪ��ֻ��-2�ۣ�Ϊ��Ԫ�أ�Y��Q��-3��+5�ۣ�ԭ�Ӱ뾶Q�ϴ�YΪ��Ԫ�ء�QΪ��Ԫ�أ�W��R��+1�ۣ�W��ԭ�Ӱ뾶��С��С����ԭ�ӣ���WΪ��Ԫ�أ�R��ԭ�Ӱ뾶�ܴ�Ϊ��Ԫ�أ�Z��+2��+4�ۣ�ԭ�Ӱ뾶��N��Oԭ������ZΪ̼Ԫ�أ�
��1������Ԫ�طֱ�Ϊ��H��Na��O��N��C��P�����Ӳ���Խ��뾶Խ���Ӳ�����ͬʱ�˵����ԽС���뾶Խ�����뾶�������ƣ���λ�ڵ�������IA�壬�ʴ�Ϊ����������IA�壻
��2�������ķе�������⣬������NH3�ķ��Ӽ����������е��쳣�ĸߣ��ʴ�Ϊ��NH3�ķ��Ӽ���������
��3��X����R��1��1��ԭ�Ӹ������γɻ������ǹ������ƣ����������д������Ӽ��ͷǼ��Լ��������Ӽ������ۼ�����X����W��ɺ�18e-�Ļ������ǹ������⣬��������ĵ���ʽΪ�� ��1mol���������2mol������ȫ��Ӧ������һ�����壬�ҷ�Ӧǰ���������������䣬��������3mol�Ķ�����̼���������غ㣬����к���������ԭ�ӣ�����ķ���ʽΪ��C3O2�������и�ԭ������������8e-�ṹ���Һ��зǼ��Լ����ṹʽΪ��O=C=C=C=O��
��1mol���������2mol������ȫ��Ӧ������һ�����壬�ҷ�Ӧǰ���������������䣬��������3mol�Ķ�����̼���������غ㣬����к���������ԭ�ӣ�����ķ���ʽΪ��C3O2�������и�ԭ������������8e-�ṹ���Һ��зǼ��Լ����ṹʽΪ��O=C=C=C=O��
�ʴ�Ϊ�����Ӽ��ͷǼ��Լ��������Ӽ������ۼ��� ��O=C=C=C=O��
��O=C=C=C=O��
���� ���⿼��λ�á��ṹ�����ʵĹ�ϵ��Ӧ�ã�������Ϣ�ƶϳ�Ԫ���ǽ��Ĺؼ�����Ϥ���ʡ�����������ʼ���ѧ���T�ɽ����Ŀ�Ѷ��еȣ�

| A�� | ��CuƬ��������ⱥ��ʳ��ˮ��2Cl-+2H2O$\frac{\underline{\;���\;}}{\;}$H2��+H2��+2OH- | |
| B�� | ������ͭƬ���缫���AgNO3��Һ��4Ag++2H2O$\frac{\underline{\;���\;}}{\;}$Ag+O2+4H+ | |
| C�� | ��ʯī���缫���AlCl3��Һ��2Cl-+2H2O$\frac{\underline{\;���\;}}{\;}$Cl2��+H2��+2OH- | |
| D�� | ��ʯī���缫���CuSO4��Һ��2Cu2++2H2O$\frac{\underline{\;���\;}}{\;}$2Cu+O2��+4H+ |
| A�� | ���ӻ�������һ���������ۼ� | |
| B�� | ���ۻ�������һ���������Ӽ� | |
| C�� | ȫ���ɷǽ���Ԫ���γɵĻ�����һ���ǹ��ۻ����� | |
| D�� | ����Ԫ�غͷǽ���Ԫ��֮���γɵĻ�ѧ��һ�������Ӽ� |
 ȫ��Һ�������һ�ֻ������ʳ�ѭ������Һ̬�ĵ�أ�Ŀǰ����ؼ����Ѿ��������죮��ͼ�Ƿ���ػ�������ԭ��ʾ��ͼ��
ȫ��Һ�������һ�ֻ������ʳ�ѭ������Һ̬�ĵ�أ�Ŀǰ����ؼ����Ѿ��������죮��ͼ�Ƿ���ػ�������ԭ��ʾ��ͼ��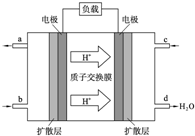 ����Ӧ��ԭ���ԭ�������˶��ֵ�������㲻ͬ����Ҫ����ط�����Խ��Խ��Ҫ�����ã���������ɴ����������ǡ����ԡ�������ȣ����벻����ʽ�����ĵ�أ�ͬʱ�����ĵ����㶪��Ҳ��Ի��������Ⱦ������������ṩ����Ϣ���ش��������⣺
����Ӧ��ԭ���ԭ�������˶��ֵ�������㲻ͬ����Ҫ����ط�����Խ��Խ��Ҫ�����ã���������ɴ����������ǡ����ԡ�������ȣ����벻����ʽ�����ĵ�أ�ͬʱ�����ĵ����㶪��Ҳ��Ի��������Ⱦ������������ṩ����Ϣ���ش��������⣺