��Ŀ����
����˵���У���ȷ���У�������
��1������������ˮ������������̼�ᱵ�������ڱ�����
��2�����������̼�⻯�����ǹ��ɹ⻯ѧ��������Ҫ����
��3���������������������Ϊ��е㼫�ͣ�������Һ��
��4������ᴿ��Ӧ�ã��ٽ��˰뵼��Ԫ���뼯��оƬ�ķ�չ������˵��������Ϣ���������Ĵ�����
��5������������淋��Ʊ�ʵ���У�Ϊ�˵õ���������茶��壬ӦС�����������ֱ���д�����������ʱֹͣ����
��6��ͨ��ʱ����Һ���������ӷֱ��������ƶ��������н�����ijһ���ƶ�
��7�����ۺ�����þ��ĩ��ϣ������ܷ������ȷ�Ӧ
��8���ߴ��ȵĶ�������㷺�����������ά�����ά����ǿ��ᡰ��·��
��9������ϡ���ᡢ̼������Һ����������Һ���ʵ������֤�ȡ�̼��������Ԫ�صķǽ�����
��10��ͬ����Ԫ�صļ������ӻ�ԭ��Խǿ��ˮ��̶�Խ��
��11��Al2O3�ڹ�ҵ�������������²��ϣ�Ҳ���ڵ�ⷨ������
��12��������ͭ�ᾧˮ�����ⶨʵ���У�����Ҫ����ƽ������������
��13����ˮ����ͨ�����ȵ����ۣ���ĩ��죬˵�����ڸ��������¿���ˮ������ѧ��Ӧ
��14����ȡ������Һ���Թ��У��ټ�������������Һ������ʪ�ĺ�ɫʯ����ֽ�����Թܿڣ���ɫʯ����ֽ��������֤����Һ�в���笠����ӣ�
��15����֪�����ᣨH3PO2����һԪ��ǿ�ᣬ��NaH2PO2�����Σ�
��1������������ˮ������������̼�ᱵ�������ڱ�����
��2�����������̼�⻯�����ǹ��ɹ⻯ѧ��������Ҫ����
��3���������������������Ϊ��е㼫�ͣ�������Һ��
��4������ᴿ��Ӧ�ã��ٽ��˰뵼��Ԫ���뼯��оƬ�ķ�չ������˵��������Ϣ���������Ĵ�����
��5������������淋��Ʊ�ʵ���У�Ϊ�˵õ���������茶��壬ӦС�����������ֱ���д�����������ʱֹͣ����
��6��ͨ��ʱ����Һ���������ӷֱ��������ƶ��������н�����ijһ���ƶ�
��7�����ۺ�����þ��ĩ��ϣ������ܷ������ȷ�Ӧ
��8���ߴ��ȵĶ�������㷺�����������ά�����ά����ǿ��ᡰ��·��
��9������ϡ���ᡢ̼������Һ����������Һ���ʵ������֤�ȡ�̼��������Ԫ�صķǽ�����
��10��ͬ����Ԫ�صļ������ӻ�ԭ��Խǿ��ˮ��̶�Խ��
��11��Al2O3�ڹ�ҵ�������������²��ϣ�Ҳ���ڵ�ⷨ������
��12��������ͭ�ᾧˮ�����ⶨʵ���У�����Ҫ����ƽ������������
��13����ˮ����ͨ�����ȵ����ۣ���ĩ��죬˵�����ڸ��������¿���ˮ������ѧ��Ӧ
��14����ȡ������Һ���Թ��У��ټ�������������Һ������ʪ�ĺ�ɫʯ����ֽ�����Թܿڣ���ɫʯ����ֽ��������֤����Һ�в���笠����ӣ�
��15����֪�����ᣨH3PO2����һԪ��ǿ�ᣬ��NaH2PO2�����Σ�
| A��8�� | B��6�� | C��5�� | D��7�� |
���㣺����ˮ���Ӧ��,�ᡢ��Ρ�������ĸ�������ϵ,��ɢϵ����������Һ�ĸ����ϵ,�ǽ�����Ԫ�����ڱ��е�λ�ü������ʵݱ�Ĺ���,������������,���������������Ⱦ������,��Ͷ�������,þ��������Ҫ������,���Ļ�ѧ����,ҩ�����Ҫ�ɷֺ���Ч,����������ʹ�÷���
ר�⣺�����������������,Ԫ�ؼ��仯����
��������1������ֻ��ˮ����������������BaSO4��
��2���⻯ѧ����������ŷ�̼�⻯����͵����������йأ�
��3�����������������
��4�����ʹ������õİ뵼����ϣ���������оƬ��
��5���������Ӳ��ȶ����ױ�������
��6��ͨ��ʱ���ʵ�������������ӷֱ����������������Ƿǵ����ʱ�������κε缫��
��7�����ۺ�����þ����Ӧ��
��8�����ά����Ҫ�ɷ��Ƕ��������Լ�����������������������Һ��Ӧ�����ɹ����ƺ�ˮ��
��9���ǽ���Ԫ�ص���ۺ����������ǿ����֤���ǽ����Ե�ǿ����
��10��ͬ����Ԫ�صļ������ӻ�ԭ��Խǿ��ˮ��̶�ԽС
��11�����ݽ����Ļ�����ȷ��������ұ��������
��12����ˮ����ͭ�ᾧˮ�ⶨʵ��������١��ijơ���
��13������ˮ����������Ӧ���ɺ�ɫ��������������
��14����ȡ������Һ���Թ��У��ټ�������������Һ������ʪ�ĺ�ɫʯ����ֽ�����Թܿڣ���ɫʯ����ֽ��������֤����Һ�в���笠����ӣ�
��15����֪�����ᣨH3PO2����һԪ��ǿ�ᣬ��NaH2PO2�����Σ�
��2���⻯ѧ����������ŷ�̼�⻯����͵����������йأ�
��3�����������������
��4�����ʹ������õİ뵼����ϣ���������оƬ��
��5���������Ӳ��ȶ����ױ�������
��6��ͨ��ʱ���ʵ�������������ӷֱ����������������Ƿǵ����ʱ�������κε缫��
��7�����ۺ�����þ����Ӧ��
��8�����ά����Ҫ�ɷ��Ƕ��������Լ�����������������������Һ��Ӧ�����ɹ����ƺ�ˮ��
��9���ǽ���Ԫ�ص���ۺ����������ǿ����֤���ǽ����Ե�ǿ����
��10��ͬ����Ԫ�صļ������ӻ�ԭ��Խǿ��ˮ��̶�ԽС
��11�����ݽ����Ļ�����ȷ��������ұ��������
��12����ˮ����ͭ�ᾧˮ�ⶨʵ��������١��ijơ���
��13������ˮ����������Ӧ���ɺ�ɫ��������������
��14����ȡ������Һ���Թ��У��ټ�������������Һ������ʪ�ĺ�ɫʯ����ֽ�����Թܿڣ���ɫʯ����ֽ��������֤����Һ�в���笠����ӣ�
��15����֪�����ᣨH3PO2����һԪ��ǿ�ᣬ��NaH2PO2�����Σ�
���
�⣺��1������ֻ��ˮ��������������̼�ᱵ������θ�����ᣬ�ʲ��������ͣ�������BaSO4���ʴ���
��2���⻯ѧ����������ŷ�̼�⻯����͵����������йأ�����ȷ��
��3��������Һ��������ʱ�����մ������ȣ��ʳ����������������ȷ��
��4�����ʹ������õİ뵼����ϣ���������оƬ������ȷ��
��5���������Ӽ��ȹ������ױ����������Եò�����������茶��壬�ʴ���
��6��ͨ��ʱ����Һ�е����������ǵ���ʣ���������������ӷֱ��������ƶ����������Ƿǵ���ʲ�����缫�������еķ�ɢ��������ijһ���ƶ����ʴ���
��7�����۲��ܽ�þ�û���������������þ�ڸ��������²���Ӧ���ʴ���
��8�����������������ƹ��ά������������������������Һ��Ӧ�����ɹ����ƺ�ˮ�����Թ��ά��ǿ��ᡰ��·��������ȷ��
��9���������Ԫ�ص���ۺ����ᣬ���Բ�����ϡ���ᡢ̼������Һ����������Һ���ʵ������֤Ԫ�صķǽ�����Cl��C��Si���ʴ���
��10��ͬ����Ԫ�صļ������ӻ�ԭ��Խǿ��ˮ��̶�ԽС����F-ˮ��̶ȴ���Cl-���ʴ���
��11�����������۵�ܸߣ�����Al2O3�ڹ�ҵ�������������²��ϣ����ǻ��ý��������ý�����ұ���õ�ⷨ�������õ�ⷨұ��������������ȷ��
��12������ͭ����ᾧˮ�ⶨʵ���У������Ĵγ������ʴ���
��13������ˮ������Ӧ���ɺ�ɫ�������������������DZ�죬�ʴ���
��14��笠����ӵļ��鷽���ǣ�������������Թ��У�������������������Һ���ھƾ��ƻ������ȣ������Թܿ��ϵ�ʪ��ĺ�ɫʯ����ֽ����ɫ��֤���������к���笠����ӣ���������ȣ������а����ݳ�����������ֽ�������ʴ���
��15��H3PO2��һԪ��ǿ��˵��ֻ�е�һ��H���Ե��룬H2PO2-���ܵ��룬��NaH2PO2�Ǵ���������Σ�����Һ�в��ܵ���������ӣ�����ȷ��
����ȷ���У���2������3������4������8������11������15��������6����ȷ����ѡB��
��2���⻯ѧ����������ŷ�̼�⻯����͵����������йأ�����ȷ��
��3��������Һ��������ʱ�����մ������ȣ��ʳ����������������ȷ��
��4�����ʹ������õİ뵼����ϣ���������оƬ������ȷ��
��5���������Ӽ��ȹ������ױ����������Եò�����������茶��壬�ʴ���
��6��ͨ��ʱ����Һ�е����������ǵ���ʣ���������������ӷֱ��������ƶ����������Ƿǵ���ʲ�����缫�������еķ�ɢ��������ijһ���ƶ����ʴ���
��7�����۲��ܽ�þ�û���������������þ�ڸ��������²���Ӧ���ʴ���
��8�����������������ƹ��ά������������������������Һ��Ӧ�����ɹ����ƺ�ˮ�����Թ��ά��ǿ��ᡰ��·��������ȷ��
��9���������Ԫ�ص���ۺ����ᣬ���Բ�����ϡ���ᡢ̼������Һ����������Һ���ʵ������֤Ԫ�صķǽ�����Cl��C��Si���ʴ���
��10��ͬ����Ԫ�صļ������ӻ�ԭ��Խǿ��ˮ��̶�ԽС����F-ˮ��̶ȴ���Cl-���ʴ���
��11�����������۵�ܸߣ�����Al2O3�ڹ�ҵ�������������²��ϣ����ǻ��ý��������ý�����ұ���õ�ⷨ�������õ�ⷨұ��������������ȷ��
��12������ͭ����ᾧˮ�ⶨʵ���У������Ĵγ������ʴ���
��13������ˮ������Ӧ���ɺ�ɫ�������������������DZ�죬�ʴ���
��14��笠����ӵļ��鷽���ǣ�������������Թ��У�������������������Һ���ھƾ��ƻ������ȣ������Թܿ��ϵ�ʪ��ĺ�ɫʯ����ֽ����ɫ��֤���������к���笠����ӣ���������ȣ������а����ݳ�����������ֽ�������ʴ���
��15��H3PO2��һԪ��ǿ��˵��ֻ�е�һ��H���Ե��룬H2PO2-���ܵ��룬��NaH2PO2�Ǵ���������Σ�����Һ�в��ܵ���������ӣ�����ȷ��
����ȷ���У���2������3������4������8������11������15��������6����ȷ����ѡB��
���������⿼��������ˮ���Ӧ�ã��ѶȲ�����ס����ˮ������ǽ���ؼ���

��ϰ��ϵ�д�
�����Ŀ
���������һ�����Ϳɳ���أ��������ҺΪKOH���ŵ�ʱ���ܷ�ӦʽΪ3Zn+2K2FeO4+8H2O?3Zn��OH��2+2Fe��OH��3+4KOH������������ȷ���ǣ�������
| A���ŵ�ʱ������ӦΪ��3Zn-6e-+6OH-�T3Zn��OH��2 |
| B���ŵ�ʱOH-�������ƶ� |
| C�����ʱÿת��3mol���ӣ�������1molFe��OH��3����ԭ |
| D�����ʱ������ӦʽΪ��3Zn��OH��2+6e-�T3Zn+6OH- |
 ��֪���ٷ�Ӧ2C+O2=2CO �������仯��ͼ��ʾ���ں�5.6g KOH��ϡ��Һ��0.5L 0.1mol/L��H2SO4��Һ��Ӧ�ų�5.73kJ������������˵����ȷ���ǣ�������
��֪���ٷ�Ӧ2C+O2=2CO �������仯��ͼ��ʾ���ں�5.6g KOH��ϡ��Һ��0.5L 0.1mol/L��H2SO4��Һ��Ӧ�ų�5.73kJ������������˵����ȷ���ǣ�������| A��2mol C��s��������O2��g����Ӧ����CO2��g�����ų�����������221 kJ |
| B��12g C��s����һ����O2��g����Ӧ����14 g CO��g�����ų�������Ϊ110.5 kJ |
| C���÷�Ӧ���Ȼ�ѧ����ʽ�ǣ�2C��s��+O2��g��=2CO��g����H=-110.5 kJ/mol |
| D����ʾ�к��ȵ��Ȼ�ѧ����ʽ2KOH ��aq��+H2SO4 ��aq��=K2SO4��aq��+2H2O ��l����H=-114.6 kJ/mol |
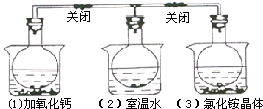 ��ͼ��ʾ��������ƿ�зֱ����NO2���壬���ֱ������ʢ���������ʵ��ձ��У��ձ�����ˮ�����ڣ�1���м���CaO���ڣ�2���в��������κ����ʣ��ڣ�3���м���NH4Cl���壬���֣�1���к���ɫ�����3���к���ɫ��dz������������ȷ���ǣ�������
��ͼ��ʾ��������ƿ�зֱ����NO2���壬���ֱ������ʢ���������ʵ��ձ��У��ձ�����ˮ�����ڣ�1���м���CaO���ڣ�2���в��������κ����ʣ��ڣ�3���м���NH4Cl���壬���֣�1���к���ɫ�����3���к���ɫ��dz������������ȷ���ǣ�������| A����ƿ��3���������ѹǿ���� |
| B��NH4Cl����ˮʱ�ų����� |
| C����ƿ��1����ƽ��ʱ��������ƽ����Է����������� |
| D��2NO2?N2O4�Ƿ��ȷ�Ӧ |
Ҫ��ȡ��ˮ�еĵ⣬�����Լ����ܳɹ����ǣ�������
| A��CCl4 |
| B���� |
| C������ |
| D���ƾ� |
�ں��¡������£�����Ӧ��������ѹǿ����ʱ��仯ʱ�����п��淴Ӧһ���ﵽƽ����ǣ�������
| A��A��g��+B��g��?C��g�� |
| B��A��g��+2B��g��?3C��g�� |
| C��A��g��+B��g��C��g��?D��g�� |
| D�����϶��ﵽƽ�� |
����һ��Ӧ�ù㷺�Ľ����������й�����˵����ȷ���ǣ�������
| A���������ᷴӦ�����Ȼ��������� |
| B������������Ũ�����жۻ� |
| C������������ȼ��������Ϊ���������� |
| D����������״���Ǻ�ɫ���� |