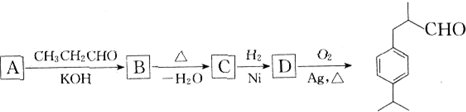
��Ŀ����
����ʵ�鷽����ȷ���ǣ�������
| A������һ�����ʵ���Ũ�ȵ���Һʱ������ˮ��������ƿ�Ŀ̶��ߣ��ý�ͷ�ιܽ�����Һ���������� |
| B���ýྻ�IJ�˿պȡ������Һ�ھƾ��ƻ��������գ�����ʻ�ɫ������Һ��һ��������K+ |
| C����CCl4��ȡ��ˮ���Һʱ���ȴ�Һ©���������ų��²�Һ�壬�ٹرջ�������©���ڵ����ϲ�Һ�� |
| D������ij��Һ���Ƿ���Fe3+ʱ�����ȼ�����������ˮ���ٵμ����軯����Һ������Һ��Ϊ��ɫ����˵����Һ��һ������Fe3+ |
���㣺��ѧʵ�鷽��������
ר�⣺ʵ��������
������A����ˮ��������ƿ�Ŀ̶��ߣ�ʵ��ʧ�ܣ�
B������ʻ�ɫ��һ���������ӣ��۲�K����ɫӦ����ɫ���ܲ�����
C����Һʱ���������²�Һ���ϣ�
D�����ȼ�����������ˮ���������ӿ�����Ϊ�����ӣ�
B������ʻ�ɫ��һ���������ӣ��۲�K����ɫӦ����ɫ���ܲ�����
C����Һʱ���������²�Һ���ϣ�
D�����ȼ�����������ˮ���������ӿ�����Ϊ�����ӣ�
���
�⣺A����ˮ��������ƿ�Ŀ̶��ߣ�ʵ��ʧ�ܣ�Ӧ�������ƣ���A����
B������ʻ�ɫ��һ���������ӣ��۲�K����ɫӦ����ɫ���ܲ���������Һ�п��ܺ���K+����B����
C����Һʱ���������²�Һ���ϣ����Һʱ���ȴ�Һ©���������ų��²�Һ�壬�ٹرջ�������©���ڵ����ϲ�Һ�壬��C��ȷ��
D�����ȼ�����������ˮ���������ӿ�����Ϊ�����ӣ������ij��Һ���Ƿ���Fe3+ʱ���μ����軯����Һ������Һ��Ϊ��ɫ����˵����Һ��һ������Fe3+����D����
��ѡC��
B������ʻ�ɫ��һ���������ӣ��۲�K����ɫӦ����ɫ���ܲ���������Һ�п��ܺ���K+����B����
C����Һʱ���������²�Һ���ϣ����Һʱ���ȴ�Һ©���������ų��²�Һ�壬�ٹرջ�������©���ڵ����ϲ�Һ�壬��C��ȷ��
D�����ȼ�����������ˮ���������ӿ�����Ϊ�����ӣ������ij��Һ���Ƿ���Fe3+ʱ���μ����軯����Һ������Һ��Ϊ��ɫ����˵����Һ��һ������Fe3+����D����
��ѡC��
���������⿼�黯ѧʵ�鷽�������ۣ�Ϊ��Ƶ���㣬�漰��Һ�����ơ���ɫ��Ӧ����Һ���������Ӽ��飬���շ�Ӧԭ�������ʵ�����Ϊ���Ĺؼ���ע�ⷽ���IJ����ԡ������Է�������Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ��ʦ�㲦��ϵ�д�
��ʦ�㲦��ϵ�д� Ӣ�żƻ���ĩ����ϵ�д�
Ӣ�żƻ���ĩ����ϵ�д�
�����Ŀ
���в����У�����ȷ���ǣ�������
| A������մ������Ӧ�����þƾ�ϴ�� |
| B��ȼ�ŵľƾ��ƴ����Ӧ��ˮ���� |
| C��ʵ�����ҽ���ʯ�ͷ�������ϩʵ��ʱ�������ڷ�Ӧ���м����������Ƭ |
| D����ȼ���顢��ϩ�ȿ�ȼ������ǰ�����ȼ����䴿�� |
����Ũ�Ⱦ�Ϊ0.10mo1?L-1�Ģ�Na2CO3��Һ�͢�NaHCO3��Һ������˵������ȷ���ǣ�������
| A��������c��Na+����c��CO32-����c��HCO3-����c��OH-����c��H+�� |
| B��������c��Na+��=c��HCO3-��+c��CO32-��+c��H2CO3�� |
| C���ٺ͢ڻ����Һ�У�c��Na+��+c��H+��=c��HCO3-��+2c��CO32-��+c��OH-�� |
| D���ٻ���ж��У�c��HCO3-��+c��CO32-��+c��H2CO3��=0.10 mol?L-1 |
��ˮ��Һ���ܴ��������һ���ǣ�������
| A��Ba2+��Na+��SO32-��Cl- |
| B��MnO4-��SO42-��K+��H+ |
| C��Al3+��Mg2+��NO3-��HCO3- |
| D��Fe3+��Na+��SCN-��Cl- |
���и������ӣ���ǿ������Һ�п��Դ���������ǣ�������
| A��K+��Ca2+��HCO3-��Cl- |
| B��Ba2+��Na+��AlO2-��NO3- |
| C��NH4+��Na+��NO3-��CO32- |
| D��Mg2+��Na+��Cl-��SO42- |
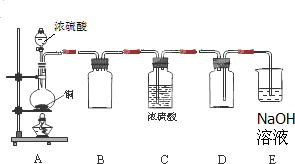 Na2SO3��SO2����ѧ���������ʣ�
Na2SO3��SO2����ѧ���������ʣ� ����ʳƷ����ױƷ�ȹ�ҵ�������������Ӽ�����ϳ�����Ϊ�����ֲ��P��Ӧ��������ȥ������ʽ�μ����Ծ���12�⣩��
����ʳƷ����ױƷ�ȹ�ҵ�������������Ӽ�����ϳ�����Ϊ�����ֲ��P��Ӧ��������ȥ������ʽ�μ����Ծ���12�⣩��