��Ŀ����
���ڿ����о��õ���Ƭ5.0gͶ��ʢ��500mL 0.5mol��L-1������Һ���ձ��У�����Ƭ�����ᷴӦ���������������뷴Ӧʱ��������ҵ�������������ʾ���ش��������⣺
1.������0��a�β�����������ԭ��ߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣ�
�йص����ӷ���ʽΪ�ߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣ�
2.������a��b�β������������ʽ�����ԭ��ߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣ�
3.������b��c�Σ������������������ӽϿ����Ҫԭ��ߣߣߣߣߣߣߣߣߣߣߣ�
4.������c�Ժ����������������½�����Ҫԭ��ߣߣߣߣߣߣߣߣߣߣߣߡ�
1.��Ƭ������������������������������������Ӧ��
Al2O3 + 6H+ = 2Al3++ 3H2O��
2.��ʼʱ����������Ƭֻ�в������ã���Һ�¶ȵͣ�
3.��Ӧ�ų�������ʹ��Һ���¶����߶��ӿ췴Ӧ���ʣ�
4.���ŷ�Ӧ�Ľ��У�������ҺŨ���½���
����:
1.�����ڻ��õĽ���������汻����������һ�����ܵ�������������Ͷ�뵽�����У�����������������������Ӧ��Ȼ���ٺ͵�������Ӧ������������ӦʽΪAl2O3 + 6H+= 2Al3+ + 3H2O��
2.���ڿ�ʼ��Ӧʱ��Һ���¶ȱȽϵͣ���Ӧ���ʾ�����
3.��Ϊ�������ᷴӦ���ڷ��ȷ�Ӧ�����ŷ�Ӧ�Ľ��У���Һ���¶������ߣ����Է�Ӧ�����ӿ졣
4.����Ӧ���е�һ���̶�ʱ�����Ũ�Ƚ��ͣ�����ʱŨ�ȵ�Ӱ�쳬�����¶ȶԷ�Ӧ���ʵ�Ӱ�죬���Է�Ӧ����Ҫ���͡�

 ��У����ϵ�д�
��У����ϵ�д� ���ڿ����о��õ���Ƭ5.0gͶ��ʢ��500mL0��5mol?L-1������Һ���ձ��и���Ƭ�����ᷴӦ���������������뷴Ӧʱ�������ͼ��ʾ��������������ʾ���ش��������⣺
���ڿ����о��õ���Ƭ5.0gͶ��ʢ��500mL0��5mol?L-1������Һ���ձ��и���Ƭ�����ᷴӦ���������������뷴Ӧʱ�������ͼ��ʾ��������������ʾ���ش��������⣺ ��2011?ӥ̶��ģ�����ڿ����о��õ���Ƭ5.0gͶ��ʢ�� 500mL 0.5mol?L-1������Һ���ձ��У�����Ƭ�����ᷴӦ��������������v�뷴Ӧʱ��t������ͼ��������������ʾ���������۴�����ǣ�������
��2011?ӥ̶��ģ�����ڿ����о��õ���Ƭ5.0gͶ��ʢ�� 500mL 0.5mol?L-1������Һ���ձ��У�����Ƭ�����ᷴӦ��������������v�뷴Ӧʱ��t������ͼ��������������ʾ���������۴�����ǣ�������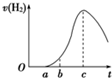 ���ڿ����о��õ���Ƭ5.0gͶ��ʢ��500mL 0.5mol?L-1������Һ���ձ��У�����Ƭ�����ᷴӦ���������������뷴Ӧʱ��Ĺ�ϵ������ͼ��������ʾ��
���ڿ����о��õ���Ƭ5.0gͶ��ʢ��500mL 0.5mol?L-1������Һ���ձ��У�����Ƭ�����ᷴӦ���������������뷴Ӧʱ��Ĺ�ϵ������ͼ��������ʾ��