��Ŀ����
14��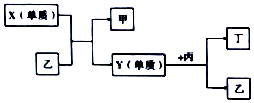 ����W��X��Y��Z���ֶ�����Ԫ�أ�W�ֱ���X��Y��Z������ɼס��ҡ������ֻ�����Ҽס��ҡ�����Ϊ10���ӵķ��ӣ�Y��Z������ɻ����ﶡ���й�Ԫ�صĵ��ʺͼס��ҡ����������ֻ������ת����ϵ��ͼ������˵����ȷ���ǣ�������
����W��X��Y��Z���ֶ�����Ԫ�أ�W�ֱ���X��Y��Z������ɼס��ҡ������ֻ�����Ҽס��ҡ�����Ϊ10���ӵķ��ӣ�Y��Z������ɻ����ﶡ���й�Ԫ�صĵ��ʺͼס��ҡ����������ֻ������ת����ϵ��ͼ������˵����ȷ���ǣ�������| A�� | ԭ�Ӱ뾶�Ĵ�С��W��Y��Z��X | B�� | ������H2���ϵ����׳̶ȣ�X��Y | ||
| C�� | �������Ӧ�������������ӻ����� | D�� | Z������������ˮ����Ϊǿ�� |
���� �ס��ҡ�����Ϊ10���ӵķ��ӣ���X���ҵķ�ӦΪ�û���Ӧ����ӦΪ������ˮ�ķ�Ӧ����֪X����ΪF2��Y����ΪO2����ΪH2O����ΪHF����WΪHԪ�أ�XΪFԪ�أ�YΪOԪ�أ�����NH3��CH4������NO��CO2��ZΪC��NԪ�أ��Դ˽����⣬
��� �⣺�����Ϸ�����֪WΪHԪ�أ�XΪFԪ�أ�YΪOԪ�أ�ZΪC��NԪ�أ�X����ΪF2��Y����ΪO2����ΪHF����ΪH2O������NH3��CH4��
A��ͬ����Ԫ�ش�����ԭ�Ӱ뾶��С����ԭ�Ӱ뾶ӦΪW��X��Y��Z����A����
B���ǽ�����X��Y��Ԫ�صķǽ�����Խǿ��Խ����������Ӧ����B��ȷ��
C�����Ϊ������������ɷ���泥�Ϊ���ӻ������C����
D��Z��ΪC�����Ӧ������������ˮ����Ϊ���ᣬ��D����
��ѡB��
���� ���⿼��������ƶϣ�Ϊ��Ƶ���㣬���շ�������ˮ���û���ӦΪ���Ĺؼ������ط������ƶ������Ŀ��飬ע�ⳣ��10�������ʣ���Ŀ�ѶȽϴ�

��ϰ��ϵ�д�
�����Ŀ
2����֪����ʱCH3COOH��NH3•H2O �ĵ��볣����ȣ�����10mLŨ��Ϊ0.1mol•L-1��CH3COOH��Һ�еμ���ͬŨ�ȵİ�ˮ���ڵμӹ���������˵����ȷ���ǣ�������
| A�� | ˮ�ĵ���̶�ʼ������ | |
| B�� | c��NH4+��/c��NH3•H2O���������ټ�С | |
| C�� | �����백ˮ�����Ϊ10 mLʱ����Һ�е�����Ũ�ȣ�c��NH4+��=c��CH3COO-����c��H+��=c��OH-�� | |
| D�� | �����백ˮ�����Ϊ10 mLʱ����Һ�е�pH=7����ˮ�������c��H+��=1��10-7 |
9�������£���0.10mol/LNaOH��Һ�ζ�20.00mLϡ���ᣨHAc��������˵������ȷ���ǣ�������
| A�� | �������Һ��pH��7ʱ�����в����ܴ���HAc���� | |
| B�� | �������Һ��pH��7ʱ�����Һ��c��Na+����c����Ac-�� | |
| C�� | �жϸõζ����̵��յ㣬���ѡ�������Ϊָʾ�� | |
| D�� | �ﵽ�ζ��յ�ʱ�����Һ��c��Na+����c����Ac-��һ������� |
19������ʵ������д�����ǣ�������
| A�� | �ù��Ϊ10 mL����Ͳ��ȡ6mL��Һ�� | |
| B�� | �������ʱ��Ӧʹ�¶ȼ�ˮ����������ƿ��֧�ܿڴ� | |
| C�� | ��Һ����ʱ����Һ©�����²�Һ����¿ڷų����ϲ�Һ����Ͽڵ��� | |
| D�� | ��ȡ����ʱ��Ӧѡ���л���ȡ��������ȡ�����ܶȱ����ˮ�� |
6������˵����ȷ���ǣ�������
| A�� | �ⵥ�ʵ����������в����ڻ�ѧ���ƻ��Ĺ��� | |
| B�� | NaCl����ˮ����Ϊ���ۼ����ƻ����Ӷ��γ���Na+��Cl- | |
| C�� | ��N2��CO2��SiO2�����У������ڹ��ۼ������Ƕ����ɷ��ӹ��� | |
| D�� | H2S�����У�����ԭ�ӵ�����㶼�ﵽ��8���ӵ��ȶ��ṹ |
3������������ȷ���ǣ�������
| A�� | ������ˮ���ռ������������� | |
| B�� | Ũ����������ɫ����Լ�ƿ�� | |
| C�� | �����Ȼ�������Һʱ�����Ȼ����������ܽ�������ˮ�м��������ἴ�� | |
| D�� | ����ʱӦ������������ͨ������ˮ�ټ��� |
4������˵����ȷ���ǣ�������
| A�� | 1mol������ȫˮ�����������Ƿ�����Ϊ2 NA | |
| B�� | ����������ȼ�ϵ���У�����������22.4L���壨��״��������ת�Ƶ�����Ϊ2NA | |
| C�� | 16.0 gCuO��Cu2S�Ļ���ﺬ��������Ϊ0.2NA | |
| D�� | 2.24L����״����һ�ȼ�������к����ۼ���Ϊ0.4NA |
5�� 1Lij�����Һ�п��ܺ��е����������
1Lij�����Һ�п��ܺ��е����������
��1��������Һ����μ���NaOH��Һ���ʵ����ȣ�������������������ʵ�����n�������NaOH��Һ�������v���Ĺ�ϵ��ͼ��ʾ��
�������Һ��ȷ�����е�������H+��NH4+��Al3+��
�ڿ϶������ڵ�������Mg2+��AlO2-��CO32-��
��д���������ٹ����з��������ӷ���ʽAl��OH��3+OH-�TAlO2-+H2O
��2������⣬����Һ�к��д�����Cl-��Br-��I-������1L�û����Һ��ͨ��һ������Cl2����Һ��Cl-��Br-��I-�����ʵ�����ͨ��Cl2���������״�����Ĺ�ϵ�����ʾ��������ش��������⣺
��ԭ��Һ��Cl-��Br-��I-�����ʵ���Ũ��֮��Ϊ1��2��1��
�ڴ���ʼ��ͨ��Cl2�����Ϊ22.4Lʱ����Һ�з�����Ӧ�������ӷ���ʽΪ4Cl2+2Br-+6I-=8Cl-+Br2+3I2��
 1Lij�����Һ�п��ܺ��е����������
1Lij�����Һ�п��ܺ��е����������| ���ܴ������е������� | H+NH4+Al3+K+ Mg2+ |
| ���ܴ������е������� | Cl-Br-I?AlO2- CO32- |
�������Һ��ȷ�����е�������H+��NH4+��Al3+��
�ڿ϶������ڵ�������Mg2+��AlO2-��CO32-��
��д���������ٹ����з��������ӷ���ʽAl��OH��3+OH-�TAlO2-+H2O
��2������⣬����Һ�к��д�����Cl-��Br-��I-������1L�û����Һ��ͨ��һ������Cl2����Һ��Cl-��Br-��I-�����ʵ�����ͨ��Cl2���������״�����Ĺ�ϵ�����ʾ��������ش��������⣺
| Cl2���������״���� | 11.2L | 22.4L | 28L |
| n��Cl-�� | 2.5mol | 3.5mol | 4.0mol |
| n��Br-�� | 3.0mol | 2.5mol | 2.0mol |
| n��I-�� | a mol | 0 | 0 |
�ڴ���ʼ��ͨ��Cl2�����Ϊ22.4Lʱ����Һ�з�����Ӧ�������ӷ���ʽΪ4Cl2+2Br-+6I-=8Cl-+Br2+3I2��