��Ŀ����
���û�ѧ��Ӧԭ���о��������ȵ��ʼ��仯����ķ�Ӧ����Ҫ���壮
��1���ϳɰ���ӦN2��g��+3H2��g��?2NH3��g�������ں��¡���ѹ��������ƽ����ϵ��ͨ���������ƽ�� �ƶ�����������ҡ���������ʹ�ô�����������Ӧ�ġ�H ���������С�����ı䡱����
��2��һ����̼�ڸ�������ˮ������Ӧ�ķ���ʽΪ��CO+H2O=CO2+H2����֪��������ȼ�յ��Ȼ�ѧ����ʽ���£�2H2��g��+O2��g���T2H2O��l����H�T-571.6kJ?mol-12CO��g��+O2��g���T2CO2��g����H�T-566kJ?mol-1��֪1molH2O��g��ת��Ϊ1mol H2O��l��ʱ�ų�44.0kJ������д��CO��ˮ�����ڸ��´����·�Ӧ���Ȼ�ѧ����ʽ ��
��3����25���£���Ũ�Ⱦ�Ϊ0.1mol?L-1��MgCl2��CuCl2�����Һ����μ��백ˮ�������� �������ѧʽ�������ɸó��������ӷ���ʽΪ ����֪25��ʱKsp[Mg��OH��2]=1.8��10-11��Ksp[Cu��OH��2]=2.2��10-20��
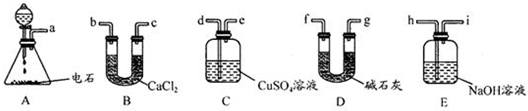
��4����������Ѱ����ʵĴ����͵缫���ϣ���N2��H2Ϊ�缫��Ӧ���HCl-NH4ClΪ�������Һ��������ȼ�յ�أ��ŵ�����У���Һ��笠�����Ũ����������д���õ�ص�������Ӧʽ ��
��5��ij�¶ȣ�t�棩ʱ���0.01mol?L-1��NaOH��Һ��pH=11���ڴ��¶��£���pH=1��H2SO4��ҺVaL��pH=11��NaOH��ҺVbL��ϣ������û��ҺΪ���ԣ���Va��Vb= ��
��6����25���£���a mol?L-1�İ�ˮ��0.01mol?L-1������������ϣ���Ӧƽ��ʱ��Һ��c��NH4+��=c��Cl-��������Һ�� �ԣ���ᡱ������С�����
��7��NH4Cl��һ����Ҫ�Ļ��ʣ�����0.1mol?L-1������ζ�0.1mol?L-1�İ�ˮ���ζ������в����ܳ��ֵĽ���ǣ� ��
a��c��NH4+��=c��Cl-����c��OH-��=c��H+��
b��c��NH4+����c��Cl-����c��OH-����c��H+��
c��c��NH4+����c��Cl-����c��OH-����c��H+��
d��c��NH4+����c��Cl-����c��OH-����c��H+��
��NH4Cl��Һ�д��ڣ�NH4++H2O?NH3?H2O+H+����÷�Ӧ�����µ�ƽ�ⳣ��K= ������֪�������£�NH3?H2O�ĵ���ƽ�ⳣ��Kb=1.7��10-5 mol?L-1��
��1���ϳɰ���ӦN2��g��+3H2��g��?2NH3��g�������ں��¡���ѹ��������ƽ����ϵ��ͨ���������ƽ��
��2��һ����̼�ڸ�������ˮ������Ӧ�ķ���ʽΪ��CO+H2O=CO2+H2����֪��������ȼ�յ��Ȼ�ѧ����ʽ���£�2H2��g��+O2��g���T2H2O��l����H�T-571.6kJ?mol-12CO��g��+O2��g���T2CO2��g����H�T-566kJ?mol-1��֪1molH2O��g��ת��Ϊ1mol H2O��l��ʱ�ų�44.0kJ������д��CO��ˮ�����ڸ��´����·�Ӧ���Ȼ�ѧ����ʽ
��3����25���£���Ũ�Ⱦ�Ϊ0.1mol?L-1��MgCl2��CuCl2�����Һ����μ��백ˮ��������
��4����������Ѱ����ʵĴ����͵缫���ϣ���N2��H2Ϊ�缫��Ӧ���HCl-NH4ClΪ�������Һ��������ȼ�յ�أ��ŵ�����У���Һ��笠�����Ũ����������д���õ�ص�������Ӧʽ
��5��ij�¶ȣ�t�棩ʱ���0.01mol?L-1��NaOH��Һ��pH=11���ڴ��¶��£���pH=1��H2SO4��ҺVaL��pH=11��NaOH��ҺVbL��ϣ������û��ҺΪ���ԣ���Va��Vb=
��6����25���£���a mol?L-1�İ�ˮ��0.01mol?L-1������������ϣ���Ӧƽ��ʱ��Һ��c��NH4+��=c��Cl-��������Һ��
��7��NH4Cl��һ����Ҫ�Ļ��ʣ�����0.1mol?L-1������ζ�0.1mol?L-1�İ�ˮ���ζ������в����ܳ��ֵĽ���ǣ�
a��c��NH4+��=c��Cl-����c��OH-��=c��H+��
b��c��NH4+����c��Cl-����c��OH-����c��H+��
c��c��NH4+����c��Cl-����c��OH-����c��H+��
d��c��NH4+����c��Cl-����c��OH-����c��H+��
��NH4Cl��Һ�д��ڣ�NH4++H2O?NH3?H2O+H+����÷�Ӧ�����µ�ƽ�ⳣ��K=
���㣺��ѧƽ���Ӱ������,�ø�˹���ɽ����йط�Ӧ�ȵļ���,pH�ļ���,���ܵ���ʵ��ܽ�ƽ�⼰����ת���ı���
ר�⣺��ѧ��Ӧ�е������仯,��ѧƽ��ר��,����ƽ������Һ��pHר��
��������1����ѹ��������ƽ����ϵ��ͨ��������������ƽ�����淽���ƶ��������Է�Ӧ�Ȳ�Ӱ�죻
��2�����ݸ�˹���ɼ��㷴Ӧ���ʱ䣬�����Ȼ�ѧ����ʽ����д���ش�
��3�������ܶȻ�����ȷ���ȳ��������ʣ��ܶȻ�����ԽС������Խ�ȳ�����ͭ���ӺͰ�ˮ��Ӧ����������ͭ��笠����ӣ�
��4������ȼ�ϵ�صĹ���ԭ��֪ʶ����д�缫��Ӧ��
��5����������к͵�ʵ���������Ӻ�������֮��ķ�Ӧ������ش�
��6����Һ�����ԣ�����Һ��������Ũ�ȵ�������������Ũ�ȣ����ݵ���غ�ȷ����Һ��笠�����Ũ�Ⱥ�������Ũ�ȵĹ�ϵ��
��7���������ζ��У�������Һ�е��������Ȼ�李��Ȼ�狀��Ȼ��⡢�Ȼ�狀�һˮ�ϰ�������Һһ�����Ե��ԣ���������ѭ����غ����������
��2�����ݸ�˹���ɼ��㷴Ӧ���ʱ䣬�����Ȼ�ѧ����ʽ����д���ش�
��3�������ܶȻ�����ȷ���ȳ��������ʣ��ܶȻ�����ԽС������Խ�ȳ�����ͭ���ӺͰ�ˮ��Ӧ����������ͭ��笠����ӣ�
��4������ȼ�ϵ�صĹ���ԭ��֪ʶ����д�缫��Ӧ��
��5����������к͵�ʵ���������Ӻ�������֮��ķ�Ӧ������ش�
��6����Һ�����ԣ�����Һ��������Ũ�ȵ�������������Ũ�ȣ����ݵ���غ�ȷ����Һ��笠�����Ũ�Ⱥ�������Ũ�ȵĹ�ϵ��
��7���������ζ��У�������Һ�е��������Ȼ�李��Ȼ�狀��Ȼ��⡢�Ȼ�狀�һˮ�ϰ�������Һһ�����Ե��ԣ���������ѭ����غ����������
���
�⣺��1����ѹ��������ƽ����ϵ��ͨ��������������ƽ�����淽���ƶ������������ֻ�ı䷴Ӧ�Ļ�ܣ�����Ӧ�Ȳ��䣬�ʴ�Ϊ�������ı䣻
��2����֪����2H2��g��+O2��g���T2H2O��l����H=-571.6kJ?mol-1
��2CO��g��+O2��g���T2CO2��g����H=-566kJ?mol-1��
��H2O��g��=H2O��l����H=-44.0kJ/mol����ӦCO��g��+H2O��g��=CO2��g��+H2��g��������
����+��+�ڵõ�������CO��g��+H2O��g��=CO2��g��+H2��g����H=-41.2kJ/mol���ʴ�Ϊ��CO��g��+H2O��g��=CO2��g��+H2��g����H=-41.2kJ/mol��
��3���ܶȻ�����ԽС������Խ�ȳ�����������ͭ���ܶȻ�С��������þ���ܶȻ�������������ͭ�ȳ�����ͭ���ӺͰ�ˮ��Ӧ����������ͭ������笠����ӣ����ӷ���ʽΪCu2++2NH3?H2O?Cu��OH��2��+2NH4+���ʴ�Ϊ��Cu��OH��2��Cu2++2NH3?H2O?Cu��OH��2��+2NH4+��
��4����N2��H2Ϊ�缫��Ӧ���HCl-NH4ClΪ�������Һ��������ȼ�յ���У��������Ƿ����õ��ӵĻ�ԭ��Ӧ�������Ի����£��缫��ӦʽΪ��N2+8H++6e-=2NH4+���ʴ�Ϊ��N2+8H++6e-=2NH4+��
��5������к͵�ʵ���������Ӻ�������֮��ķ�Ӧ���¶ȣ�t�棩ʱ���0.01mol?L-1��NaOH��Һ��pH=11��Kw=10-13����pH=1��H2SO4��ҺVaL��pH=11��NaOH��ҺVbL��ϣ������û��ҺΪ���ԣ���0.1Va=0.01Vb�����Va��Vb=1��10���ʴ�Ϊ��1��10��
��6����Һ�д��ڵ���غ㣬����c��Cl-��+c��OH-��=c��NH4+��+c��H+������Һ��ʾ���ˣ�����c��OH-��=c��H+������Һ��ʾ���ԣ��ʴ�Ϊ���У�
��7�����ζ�����Һ�е�����Ϊ�Ȼ�狀�һˮ�ϰ�����һ����Һ�Լ��ԣ���c��OH-����c��H+������Һ��������ʵĵ��룾�ε�ˮ�⣬��c��NH4+����c��Cl-��������ϵ���غ㣬��c�ǿ��ܳ��ֵĽ�������ζ�����Һ�е�����Ϊ�Ȼ�泥���笠�����ˮ������Һ�����ԣ���c��H+����c��OH-������ˮ��ij̶Ⱥ�������c��Cl-����c��NH4+�����ҷ��ϵ���غ㣬��bd�����ܳ��֣����ζ�����Һ�е�����Ϊ�Ȼ�狀�һˮ�ϰ�������Һ��������ʵĵ���̶����ε�ˮ��̶���ͬʱ����ҺΪ���ԣ���c��OH-��=c��H+�����ɵ���غ��֪c��NH4+��=c��Cl-������a�ǿ��ܳ��ֵĽ����NH3?H2O�ĵ���ƽ�ⳣ��Kb=
=1.7��10-5 mol?L-1��NH4++H2O?NH3?H2O+H+����÷�Ӧ�����µ�ƽ�ⳣ��K=
=
=
=5.88��10-8���ʴ�Ϊ��bd��5.88��10-8��
��2����֪����2H2��g��+O2��g���T2H2O��l����H=-571.6kJ?mol-1
��2CO��g��+O2��g���T2CO2��g����H=-566kJ?mol-1��
��H2O��g��=H2O��l����H=-44.0kJ/mol����ӦCO��g��+H2O��g��=CO2��g��+H2��g��������
| 1 |
| 2 |
��3���ܶȻ�����ԽС������Խ�ȳ�����������ͭ���ܶȻ�С��������þ���ܶȻ�������������ͭ�ȳ�����ͭ���ӺͰ�ˮ��Ӧ����������ͭ������笠����ӣ����ӷ���ʽΪCu2++2NH3?H2O?Cu��OH��2��+2NH4+���ʴ�Ϊ��Cu��OH��2��Cu2++2NH3?H2O?Cu��OH��2��+2NH4+��
��4����N2��H2Ϊ�缫��Ӧ���HCl-NH4ClΪ�������Һ��������ȼ�յ���У��������Ƿ����õ��ӵĻ�ԭ��Ӧ�������Ի����£��缫��ӦʽΪ��N2+8H++6e-=2NH4+���ʴ�Ϊ��N2+8H++6e-=2NH4+��
��5������к͵�ʵ���������Ӻ�������֮��ķ�Ӧ���¶ȣ�t�棩ʱ���0.01mol?L-1��NaOH��Һ��pH=11��Kw=10-13����pH=1��H2SO4��ҺVaL��pH=11��NaOH��ҺVbL��ϣ������û��ҺΪ���ԣ���0.1Va=0.01Vb�����Va��Vb=1��10���ʴ�Ϊ��1��10��
��6����Һ�д��ڵ���غ㣬����c��Cl-��+c��OH-��=c��NH4+��+c��H+������Һ��ʾ���ˣ�����c��OH-��=c��H+������Һ��ʾ���ԣ��ʴ�Ϊ���У�
��7�����ζ�����Һ�е�����Ϊ�Ȼ�狀�һˮ�ϰ�����һ����Һ�Լ��ԣ���c��OH-����c��H+������Һ��������ʵĵ��룾�ε�ˮ�⣬��c��NH4+����c��Cl-��������ϵ���غ㣬��c�ǿ��ܳ��ֵĽ�������ζ�����Һ�е�����Ϊ�Ȼ�泥���笠�����ˮ������Һ�����ԣ���c��H+����c��OH-������ˮ��ij̶Ⱥ�������c��Cl-����c��NH4+�����ҷ��ϵ���غ㣬��bd�����ܳ��֣����ζ�����Һ�е�����Ϊ�Ȼ�狀�һˮ�ϰ�������Һ��������ʵĵ���̶����ε�ˮ��̶���ͬʱ����ҺΪ���ԣ���c��OH-��=c��H+�����ɵ���غ��֪c��NH4+��=c��Cl-������a�ǿ��ܳ��ֵĽ����NH3?H2O�ĵ���ƽ�ⳣ��Kb=
| c(NH4+)?c(OH-) |
| c(NH3?H2O) |
| c(NH3?H2O)?c(H+) |
| c(NH4+) |
| Kw |
| Kb |
| 1��10-14 |
| 1.7��10-5 |
�����������漰�绯ѧ���Ȼ�ѧ����ѧƽ��ļ���ȷ���Ӧ�õ�֪ʶ��ע��֪ʶ�Ĺ��ɺ������ǽ���Ĺؼ����ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
 ˮ�ĵ���ƽ��������ͼ��ʾ��
ˮ�ĵ���ƽ��������ͼ��ʾ��