��Ŀ����
19��NiCl2�ǻ����ϳ�������Ҫ����Դ����ҵ���Խ��������ϣ���Fe��Ca��Mg�����ʣ�Ϊԭ������NiCl2���̶�����Ni2O3�Ĺ���������ͼ��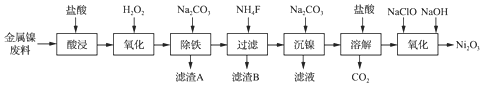
��������ؽ������������������������pH�����ʾ��
| �������� | Fe��OH��3 | Fe��OH��2 | Ni��OH��2 |
| ��ʼ������pH | 1.1 | 6.5 | 7.1 |
| ������ȫ��pH | 3.2 | 9.7 | 9.2 |
��2������H2O2ʱ������Ҫ��Ӧ�����ӷ���ʽΪ2Fe2++H2O2+2H+�T2Fe3++2H2O��
��3����������ʱ��������ҺpH�ķ�ΧΪ3.2��7.1��
��4������B����Ҫ�ɷֵĻ�ѧʽΪMgF2��CaF2��
��5����֪��Ksp��NiCO3��=1.42��10-7��Ϊȷ��������ȫ������Һ��c��Ni2+����1.0��10-6����Ӧ������Һ��c��CO32-����0.142mol•L-1��
��6��������������Ni2O3�����ӷ���ʽΪ2Ni2++ClO-+4OH-�TNi2O3��+Cl-+2H2O��
���� ���������ϣ���Fe��Ca��Mg�����ʣ���������������������Һ����Ҫ����Ni2+��Cl-������������Fe2+��Ca2+��Mg2+�ȣ������������������������Ϊ�����ӣ�����̼������Һ������Һ��pH��ʹ������ȫ������������AΪFe��OH��3�����˺����Һ���ټ������麟���Ca2+��Mg2+������BΪMgF2��CaF2���ٴι��˺�����Һ�м���̼���Ƴ��������ӵ�NiCO3����NiCO3���������ᣬ���Ȼ�����Һ�������м���������ƺ�����������Һ�ɵ�Ni2O3��
��1�������¶ȡ��������Ũ�ȡ���ֽ��衢����Ӵ�����ȿ�����߽��������ʣ�
��2����������������������Ϊ�����ӣ���ԭ����Ϊˮ���ݴ�д�����ӷ���ʽ��
��3������ʱ����������Ҫ������ȫ���������Ӳ��ܳ�����
��4�����˺����Һ���ټ������麟���Ca2+��Mg2+��
��5������Qc��Kspʱ����Һ�����ͼ���c��CO32-���ķ�Χ��
��6��ClԪ����+1�۽��͵�-1�ۣ�����Ӧ����NaCl����Ni��+2�����ߵ�+3�ۣ�����ԭ���غ������ת���غ���ƽ��
��� �⣺���������ϣ���Fe��Ca��Mg�����ʣ���������������������Һ����Ҫ����Ni2+��Cl-������������Fe2+��Ca2+��Mg2+�ȣ������������������������Ϊ�����ӣ�����̼������Һ������Һ��pH��ʹ������ȫ������������AΪFe��OH��3�����˺����Һ���ټ������麟���Ca2+��Mg2+������BΪMgF2��CaF2���ٴι��˺�����Һ�м���̼���Ƴ��������ӵ�NiCO3����NiCO3���������ᣬ���Ȼ�����Һ�������м���������ƺ�����������Һ�ɵ�Ni2O3��
��1��Ϊ����߽��������Ͻ��������ʣ����������������Ũ�ȣ����������гɷ�ĩ���ӳ�����ʱ��ȣ���
�ʴ�Ϊ�����������Ũ�ȣ����������гɷ�ĩ���ӳ�����ʱ��ȣ���
��2����������������������Ϊ�����ӣ���ԭ����Ϊˮ�����ӷ���ʽΪ2Fe2++H2O2+2H+�T2Fe3++2H2O��
�ʴ�Ϊ��2Fe2++H2O2+2H+�T2Fe3++2H2O��
��3������ʱ����������Ҫ������ȫ���������Ӳ��ܲ���������������Һ��PHֵ������3.2��7.1��
�ʴ�Ϊ��3.2��7.1��
��4�����˺����Һ���ټ������麟���Ca2+��Mg2+������BΪMgF2��CaF2��
�ʴ�Ϊ��MgF2��CaF2��
��5��Ksp��NiCO3��=1.42��10-7����������ȫ����ʱ����Һ��c��Ni2+����1.0��10-6mol/L��Ӧ������Һ��c��CO32-����$\frac{{K}_{sp}��NiC{O}_{3}��}{C��N{i}^{2+}��}$��$\frac{1.42��1{0}^{-7}}{1.0��1{0}^{-6}}$=0.142mol/L��
�ʴ�Ϊ��0.142��
��6��ClԪ����+1�۽��͵�-1�ۣ�����Ӧ����NaCl����Ni��+2�����ߵ�+3�ۣ�����Ni2O3����Ӧ���ӷ���ʽΪ��2Ni2++ClO-+4OH-=Ni2O3��+Cl-+2H2O��
�ʴ�Ϊ��2Ni2++ClO-+4OH-=Ni2O3��+Cl-+2H2O��
���� ���⿼�����ʷ�����ᴿ�ķ����ͻ��������ۺ�Ӧ�ã��漰��Ӧ����Ӱ�����ء��������ơ�İ������ʽ��д����Ϣ��ȡ�����ȣ��Ǹ߿��������ͣ��Ƕ�ѧ���ۺ������Ŀ��飬��Ŀ�Ѷ��еȣ�

 �����Ƹ���ʦ����ϵ�д�
�����Ƹ���ʦ����ϵ�д� ��ͨ����ͬ����ϰ��ϵ�д�
��ͨ����ͬ����ϰ��ϵ�д� ����С����ͬ������ϵ�д�
����С����ͬ������ϵ�д�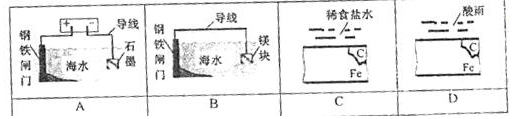
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ����������NaOH��Һ������÷�Һ©����Һ�����²���Һ | |
| B�� | ������ˮ���������屽�ӣ��ٹ��˳�ȥ | |
| C�� | ��ˮ�����ȳ��� 70�棨��������ˮ���ܽ�Ⱥܴ�Ȼ���÷�Һ©����Һ�������²���Һ | |
| D�� | ����������ױ��е�110�棬���ӷе�182�棩�����������ױ� |
$\frac{1}{2}$H2+NiO��OH��$?_{�ŵ�}^{���}$Ni��OH��2�����ݴ˷�Ӧ�жϣ�������������ȷ���ǣ�������
| A�� | ��طŵ�ʱ����ظ�����Χ��Һ��c��OH-��Ϊ������ | |
| B�� | ��طŵ�ʱ��H2�Ǹ��� | |
| C�� | ��ط�ʱʱ����Ԫ�ر����� | |
| D�� | ��طŵ�ʱ����Ԫ�ر����� |
| A�� | ��0.1mol•L-1 Na2SO3��Һ��Һ�У�c��Na+���T2c��SO32-��+c��HSO3-��+c��H2SO3�� | |
| B�� | ��0.1 mol•L-1Na2CO3��Һ�У�c��OH-��-c��H+���Tc��HCO3-��+2c��H2CO3�� | |
| C�� | ��0.2 mol•L-1NaHCO3��Һ�м�������0.1 mol•L-1NaOH��Һ��c��CO32-����c��HCO3-����c��OH-����c��H+�� | |
| D�� | �����£�CH3COONa��CaCl2�����Һ�У�c��Na+��+c��Ca2+���Tc��CH3COOH��+c��CH3COO-��+2 c��Cl-�� |
| W | X | Y |
| Z |
��1��XԪ�������ڱ��е�λ�ã��������ڢ�A�壮
��2��Z��ԭ�ӽṹʾ��ͼ��
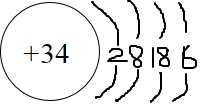 ��
����3��W������������������ռ���Һ����Ӧ�Ļ�ѧ����ʽΪ��SiO2+2NaOH=Na2SiO3+H2O��
��4��̽Ѱͬһ����Ԫ�����ʵ�һЩ��ͬ���ɣ���ѧϰ��ѧ����Ҫ����֮һ�����±����г���H2ZO3���ֲ�ͬ��ѧ���ʵ��Ʋ⣬������д����Ӧ�Ļ�ѧ����ʽ��
| ��� | �����Ʋ� | ��ѧ����ʽ |
| ʾ�� | ������ | H2ZO3+4HI�TZ��+2I2+1H2O |
| �� | ���� | H2SeO3+2NaOH=Na2SeO3+2H2O |
| �� | ��ԭ�� | H2SeO3+Cl2+H2O=H2SeO4+2HCl |
| A�� | ��3mLϡ������Һ������Zn��Ӧ��������ϡ��ʱ����5mLŨ���ᣬ��Ѹ�ٲ����϶����ݣ�����������������ó����ۣ�����Ũ��������H2�ķ�Ӧ���ʼӿ� | |
| B�� | FeCl3+3KSCN?Fe��SCN��3��Ѫ��ɫ��+3KCl������Һ�м���KCl����ɿ��ٿ���Ѫ��ɫ | |
| C�� | Ba��OH��2•8H2O��NH4Cl�ķ�Ӧ�Ƿ��ȷ�Ӧ | |
| D�� | ��ѧ��Ӧ�е������仯��ͨ������Ϊ�����ı仯 |