��Ŀ����
���ĺ���������A��B�ܷ�������ת����
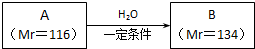
��֪A������C��H��O����Ԫ�ص�������Ϊ12��1��16��ʵ�����A��ʹ��ˮ��ɫ��1 mol A������NaHCO3��Һ��Ӧ�ų�2 mol CO2����ش�
��1��A�������������ŵ������� ��B�ķ���ʽ�� ��
��2��A�������ᷴӦֻ�ܵõ�һ�ֲ����A�Ľṹ��ʽ�� ��
��3��B��Ũ���������¼������Ҵ������������ᷢ��������Ӧ��B������Ҵ���Ӧ�Ļ�ѧ����ʽ�� ��
��4��Bʧȥ1����ˮ�����γ�һ����Ԫ��״������C��C�Ľṹ��ʽ�� ��
��5��д��һ����B����ͬ�����ŵ�B��ͬ���칹��ṹ��ʽ ��
��6��1mol B������Na��NaOH��Һ��NaHCO3��Һ��ַ�Ӧ������Na��NaOH��NaHCO3���ʵ���֮��Ϊ ��

��֪A������C��H��O����Ԫ�ص�������Ϊ12��1��16��ʵ�����A��ʹ��ˮ��ɫ��1 mol A������NaHCO3��Һ��Ӧ�ų�2 mol CO2����ش�
��1��A�������������ŵ�������
��2��A�������ᷴӦֻ�ܵõ�һ�ֲ����A�Ľṹ��ʽ��
��3��B��Ũ���������¼������Ҵ������������ᷢ��������Ӧ��B������Ҵ���Ӧ�Ļ�ѧ����ʽ��
��4��Bʧȥ1����ˮ�����γ�һ����Ԫ��״������C��C�Ľṹ��ʽ��
��5��д��һ����B����ͬ�����ŵ�B��ͬ���칹��ṹ��ʽ
��6��1mol B������Na��NaOH��Һ��NaHCO3��Һ��ַ�Ӧ������Na��NaOH��NaHCO3���ʵ���֮��Ϊ
���㣺�л�����ƶ�,�л���Ľṹ������
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
��������A������C��H��O����Ԫ�ص�������Ϊ12��1��16��A������C��H��O����Ԫ�ص�ԭ�Ӹ�����Ϊ
��
��
=1��1��1��ʵ�����A��ʹ��ˮ��ɫ��1mol A ������NaHCO3 ��Һ��Ӧ�ų�2molCO2����A�к���-C=C-��2��-COOH��A����Է�������Ϊ116��116-90-24=2��������к���2��Hԭ�ӣ���AΪHOOC-CH=CH-COOH��HOOC-C��COOH��=CH2��A�������ᷴӦֻ�ܵõ�һ�ֲ������Aֻ��ΪHOOC-CH=CH-COOH��B����Է�������Ϊ134��134-116=18����A��B����̼̼˫����ˮ�ļӳɷ�Ӧ������B�к���-OH��-COOH����BΪHOOC-CH2CH��OH��-COOH��Ȼ�������ʵĽṹ�����������
| 12 |
| 12 |
| 1 |
| 1 |
| 16 |
| 16 |
���
�⣺��A������C��H��O����Ԫ�ص�������Ϊ12��1��16��A������C��H��O����Ԫ�ص�ԭ�Ӹ�����Ϊ
��
��
=1��1��1��ʵ�����A��ʹ��ˮ��ɫ��1mol A ������NaHCO3 ��Һ��Ӧ�ų�2molCO2����A�к���-C=C-��2��-COOH��A����Է�������Ϊ116��116-90-24=2��������к���2��Hԭ�ӣ���AΪHOOC-CH=CH-COOH��HOOC-C��COOH��=CH2��A�������ᷴӦֻ�ܵõ�һ�ֲ������Aֻ��ΪHOOC-CH=CH-COOH��B����Է�������Ϊ134��134-116=18����A��B����̼̼˫����ˮ�ļӳɷ�Ӧ������B�к���-OH��-COOH����BΪHOOC-CH2CH��OH��-COOH��
��1��A�к�̼̼˫�����Ȼ���BΪHOOC-CH2CH��OH��-COOH�������ʽΪC4H6O5���ʴ�Ϊ��̼̼˫�����Ȼ���C4H6O5��
��2��A�������ᷴӦֻ�ܵõ�һ�ֲ������Aֻ��ΪHOOC-CH=CH-COOH���ʴ�Ϊ��HOOCCH=CHCOOH��
��3��B������Ҵ���Ӧ�Ļ�ѧ����ʽΪ ��
��
�ʴ�Ϊ�� ��
��
��4��B ʧȥ1 ����ˮ�����γ�һ����Ԫ��״������C����C�Ľṹ��ʽΪ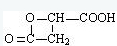 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��5��B�к���-OH��-COOH��BΪHOOC-CH2CH��OH��-COOH���京��ͬ�����ŵ�B ��ͬ���칹��ṹ��ʽΪ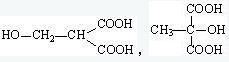 ��
��
�ʴ�Ϊ��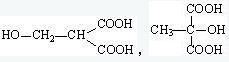 ��������һ�֣���
��������һ�֣���
��6��BΪHOOC-CH2CH��OH��-COOH��-OH��-COOH����Na��Ӧ��ֻ��-COOH��NaOH��NaHCO3��Ӧ����1molB����Na��NaOH��Һ��NaHCO3��Һ��ַ�Ӧ������Na��NaOH��NaHCO3���ʵ���֮��Ϊ3��2��2���ʴ�Ϊ��3��2��2��
| 12 |
| 12 |
| 1 |
| 1 |
| 16 |
| 16 |
��1��A�к�̼̼˫�����Ȼ���BΪHOOC-CH2CH��OH��-COOH�������ʽΪC4H6O5���ʴ�Ϊ��̼̼˫�����Ȼ���C4H6O5��
��2��A�������ᷴӦֻ�ܵõ�һ�ֲ������Aֻ��ΪHOOC-CH=CH-COOH���ʴ�Ϊ��HOOCCH=CHCOOH��
��3��B������Ҵ���Ӧ�Ļ�ѧ����ʽΪ
 ��
���ʴ�Ϊ��
 ��
����4��B ʧȥ1 ����ˮ�����γ�һ����Ԫ��״������C����C�Ľṹ��ʽΪ
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����5��B�к���-OH��-COOH��BΪHOOC-CH2CH��OH��-COOH���京��ͬ�����ŵ�B ��ͬ���칹��ṹ��ʽΪ
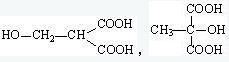 ��
���ʴ�Ϊ��
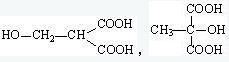 ��������һ�֣���
��������һ�֣�����6��BΪHOOC-CH2CH��OH��-COOH��-OH��-COOH����Na��Ӧ��ֻ��-COOH��NaOH��NaHCO3��Ӧ����1molB����Na��NaOH��Һ��NaHCO3��Һ��ַ�Ӧ������Na��NaOH��NaHCO3���ʵ���֮��Ϊ3��2��2���ʴ�Ϊ��3��2��2��
���������⿼���л�����ƶϣ�Ϊ��Ƶ���㣬��ȷA��B�Ľṹ��A�����ʡ�A��B�����ӳ��ǽ�����ͻ�ƿڣ���ϣ�2��ȷ��A�Ľṹ�ǽ����Ĺؼ����ط������ƶ��������л���ṹ�����ʵĿ��飬��Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ǧ�������������ĩ�����Ծ�����ϵ�д�
ǧ�������������ĩ�����Ծ�����ϵ�д�
�����Ŀ
һ���¶��£����������ԼΪ1.0L�ĺ����ܱ������з�����Ӧ��2CH3OH��g���TCH3OCH3��g��+H2O��g��
����˵����ȷ���ǣ�������
����˵����ȷ���ǣ�������
| ���� ��� | �¶ȣ��棩 | ��ʼ���ʵ�����mol�� | ƽ�����ʵ�����mol�� | |
| CH3OH��g�� | CH3OCH3��g�� | H2O��g�� | ||
| I | 387 | 0.20 | 0.080 | 0.080 |
| �� | 387 | 0.40 | ||
| �� | 207 | 0.20 | 0.090 | 0.090 |
| A���÷�Ӧ������ӦΪ���ȷ�Ӧ |
| B���ﵽƽ��ʱ������I�е�CH3OH����������������е�С |
| C������I�з�Ӧ�ﵽƽ������ʱ����������еij� |
| D������ʼʱ������I�г���CH3OH 0.1mol��CH3OCH30.15mol��H2O 0.10mol����Ӧ��������Ӧ������� |
����������Fe2O3��ɵĻ����m gͶ�뵽������ϡH2SO4�У��ڱ�״�����ռ���V L H2��������Һ��KSCN��Һ���Ժ�ɫ����������Fe2O3������Ϊ��������
A��
| ||
B��
| ||
C��
| ||
D��
|
���з������ݺͽ��۶���ȷ���ǣ�������
| A��HClO��Ũ���ᡢHNO3�����������ԣ������������� |
| B��H2O��HCOOH��Cu2��OH��2CO3��������Ԫ�أ����������� |
| C��HF��CH3COOH��CH3CH2OH��������ˮ�����ǵ���� |
| D��HCOOH��H2CO3��H2SO4�����о�����������ԭ�ӣ����Ƕ�Ԫ�� |
�ڶ�����Ԫ���У�ԭ�������ֻ��1����2�����ӵ�Ԫ���ǣ�������
| A������Ԫ�� |
| B��ϡ������ |
| C���ǽ���Ԫ�� |
| D����ȷ��Ϊ��һ��Ԫ�� |
�����ڴ������Է�ˮ�������ǣ�������
| A�������� | B������ |
| C����ʯ�� | D����ҵ�ƾ� |
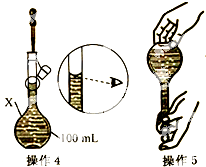 ijͬѧ������100mL 0.10mol/L CuSO4��Һ�����²���1��5�������ƹ��̼�ʾ��ͼ��
ijͬѧ������100mL 0.10mol/L CuSO4��Һ�����²���1��5�������ƹ��̼�ʾ��ͼ��