��Ŀ����
 ��������Ԫ�صĵ��ʼ��������ڹ�ũҵ������Ӧ�ù㷺��
��������Ԫ�صĵ��ʼ��������ڹ�ũҵ������Ӧ�ù㷺����1����������Ԫ���У���̬ԭ�ӵ������ֻ��1�����ӵ�Ԫ�ع���
��2����˿պȡ�Ȼ����ھƾ��������գ��������ש��ɫ����ɫԭ����
A���Ȼ������Ȼӷ�
B���Ȼ������ȷֽ�
C���������е���ԾǨ
D���������е���ԾǨ
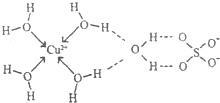
��3����ˮ����ͭ�ʰ�ɫ����ˮ���γɵ���������ɫ����ѧ��ͨ��X���߲ⶨ�������Ľṹ����ṹ������ͼ��ʾ��
�ٵ��������г����ۼ�����λ�������ķ��»����⣬�����ڵ�������������
�ڵ�������CuSO4?5H2O����д��[Cu��H2O��4]SO4?H2O����������ԭ�ӣ����ӣ���
��ͭ�����γɵ�������λ���ӵ����ӷ���ʽΪ
���㣺�����ijɼ����,ԭ�Ӻ�������Ų�
ר�⣺ԭ�������ṹר��,��ѧ���뾧��ṹ
��������1�����ݵ����Ų����ɷ�����
��2����Ԫ�ص���ɫ��ӦΪש��ɫ��������ɫ��Ӧʱ�����������������ӵ��ӷ���ԾǨ��
��3������ͼ��֪H��O֮����������ͭ�����������֮��������Ӽ���
�ڸ���ˮ��ͭ����[Cu��H2O��4]2+������
��ͭ������ˮ����ͨ����λ���γ�ˮ��ͭ����[Cu��H2O��4]2+��
��2����Ԫ�ص���ɫ��ӦΪש��ɫ��������ɫ��Ӧʱ�����������������ӵ��ӷ���ԾǨ��
��3������ͼ��֪H��O֮����������ͭ�����������֮��������Ӽ���
�ڸ���ˮ��ͭ����[Cu��H2O��4]2+������
��ͭ������ˮ����ͨ����λ���γ�ˮ��ͭ����[Cu��H2O��4]2+��
���
�⣺��1����������Ԫ���У���̬ԭ�ӵ������ֻ��1�����ӵ�Ԫ�أ���4s1����ԭ��4s�ܼ�δ����������֮һ�ǰ����ܼ�˳���������Ľ����1s22s22p63s23p64s1����Ϊ19KԪ�أ�
���֮���ǰ��պ��ع�����������Ľ����1s22s22p63s23p63d54s1��1s22s22p63s23p63d104s1����Ϊ24Cr��29Cu��
�ʴ�Ϊ��3��
��2����Ԫ�ؽ�����ɫ��Ӧʱ�����������������ӵ��ӷ���ԾǨ�������������Ϊש��ɫ���ʴ�Ϊ��C��
��3������ͼ��֪H��O֮����������ͭ�����������֮��������Ӽ����ʴ�Ϊ������� ���Ӽ���
��ͭ������ˮ����ͨ����λ���γ�ˮ��ͭ����[Cu��H2O��4]2+����������ԭ�ӣ����ӣ���Cu2+��������H2O����λ��Ϊ4��Oԭ�Ӻ��йµ��Ӷ�����ԭ�ӣ�
�ʴ�Ϊ��Cu2+��H2O��O��4��
��ͭ������ˮ����ͨ����λ���γ�ˮ��ͭ����[Cu��H2O��4]2+�����ӷ���ʽΪCu2++4H2O=[Cu��H2O��4]2+���ʴ�Ϊ��Cu2++4H2O=[Cu��H2O��4]2+��
���֮���ǰ��պ��ع�����������Ľ����1s22s22p63s23p63d54s1��1s22s22p63s23p63d104s1����Ϊ24Cr��29Cu��
�ʴ�Ϊ��3��
��2����Ԫ�ؽ�����ɫ��Ӧʱ�����������������ӵ��ӷ���ԾǨ�������������Ϊש��ɫ���ʴ�Ϊ��C��
��3������ͼ��֪H��O֮����������ͭ�����������֮��������Ӽ����ʴ�Ϊ������� ���Ӽ���
��ͭ������ˮ����ͨ����λ���γ�ˮ��ͭ����[Cu��H2O��4]2+����������ԭ�ӣ����ӣ���Cu2+��������H2O����λ��Ϊ4��Oԭ�Ӻ��йµ��Ӷ�����ԭ�ӣ�
�ʴ�Ϊ��Cu2+��H2O��O��4��
��ͭ������ˮ����ͨ����λ���γ�ˮ��ͭ����[Cu��H2O��4]2+�����ӷ���ʽΪCu2++4H2O=[Cu��H2O��4]2+���ʴ�Ϊ��Cu2++4H2O=[Cu��H2O��4]2+��
���������⿼���������Ų�ʽ����ѧ����ԭ�ӵĽṹ�����ʹ�ϵ�Լ�ѧ����ͼ��������Ŀ�Ѷ��еȣ�ּ�ڿ���ѧ����֪ʶ�����������ã�

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
��һ���¶�����a L�ܱ������м���1mol X�����2mol Y���壬�������·�ӦX��g��+Y��g��?2Z��g�����˷�Ӧ�ﵽƽ��ı�־�ǣ�������
| A��������ѹǿ����ʱ��仯 |
| B�������ڸ����ʵ�Ũ�Ȳ���ʱ��仯 |
| C��������X��Y��Z��Ũ��֮��Ϊ1��1��2 |
| D����λʱ��������0.1 mol Xͬʱ����0.2 mol Z |
���ڷ��ӻ���������ԭ�ӵ��ӻ�������ͺͿռ乹�ͣ������ж���ȷ���ǣ�������
| A��PH3��sp2ƽ�������� |
| B��SO32-��sp2 ƽ�������� |
| C��NO3-��sp3 ������ |
| D��SO2��sp2V�� |
����������ȷ���ǣ�������
| A��c��H+������1��10-7 mol/L����Һһ����������Һ |
| B���κ�Ũ�ȵ���Һ��������pH����ʾ�����Ե�ǿ�� |
| C�������Ǵ�ˮ�����ԡ����Ի�����ϡ��Һ�������£���c��H+��?c��OH-��=1��10-14 |
| D��0.2 mol/L CH3COOH��Һ�е�c��H+����0.1 mol/L CH3COOH��Һ�е�c��H+����2�� |
�������ʵ���Ҫ�ɷ����ڹ����ε��ǣ�������
| A����оƬ | B���մ� |
| C������ | D��ʯ��ʯ |
ç��������ںϳ�ҩ���ƣ���ṹ��ʽ��ͼ��ʾ�����й���ç�����˵����ȷ�ģ�������
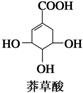

| A����ʹFeCl3��Һ��ɫ |
| B�������к���2�ֹ����� |
| C���ɷ����ӳɺ�ȡ����Ӧ |
| D������������ԭ�ӿ��Թ�ƽ�� |
���з���ʽֻ��ʾһ�ִ�������ǣ�������
| A��C2H6O |
| B��C3H8 |
| C��C4H10 |
| D��C |