��Ŀ����
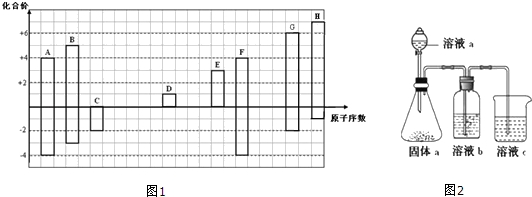
6�����������������Ȼ�պ����У���������ˮ������������Ϣ����������һ����������ϳ������������·����ͼ��ʾ��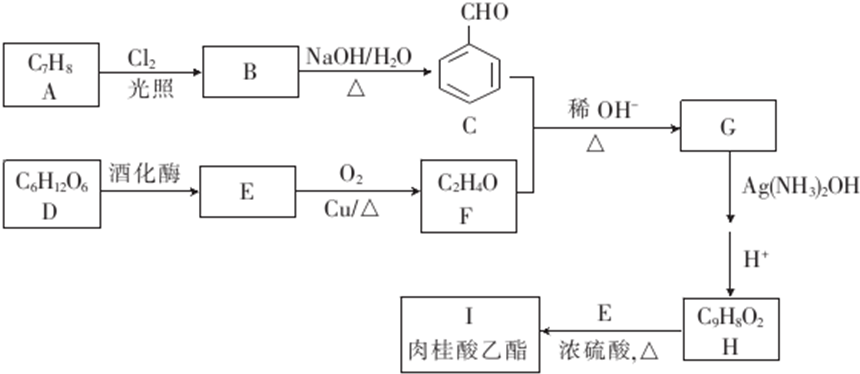
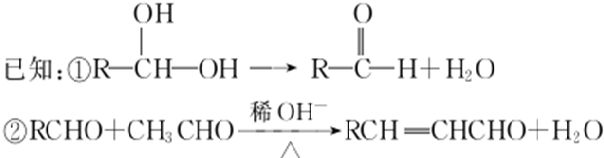
�ش��������⣺
��1��A�������Ǽױ���C�к��еĹ����ŵ�������ȩ����
��2��B�Ľṹ��ʽ��
 ��E��F�ķ�Ӧ������������Ӧ��
��E��F�ķ�Ӧ������������Ӧ����3��G�����������18��ԭ�ӹ�ƽ�森
��4��H+E��I�Ļ�ѧ����ʽΪ
 ��
����5����H��Ϊͬ���칹�壬��ͬʱ���������������л���Ľṹ�������������칹����16�֣�
���ܷ���������Ӧ ��������������Һ������ɫ��Ӧ�۳������ⲻ�����������к˴Ź�������5��壬�ҷ����֮��Ϊ1��2��2��2��1�Ľṹ��ʽΪ
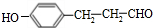 ��
����6����������������ĺϳ�·�ߣ����һ����EΪ��ʼԭ���Ʊ�1-��ϩ�ĺϳ�·�ߣ�CH3CH2OH$��_{Cu��}^{O_{2}}$CH3CHO$��_{��}^{ϡ��}$CH3CH=CHCHO$��_{����}^{����}$CH3CH2CH2CH2OH$��_{��}^{ŨH_{2}SO_{4}}$CH3CH2CH=CH2��
���� A�������ڹ�������������������ȡ����Ӧ����B��B����±������ˮ�ⷴӦ������ȥ1����ˮ�õ� ����AΪ
����AΪ ��BΪ
��BΪ ������D��F�ķ���ʽ��֪��DΪ�������ھƻ�ø����������EΪCH3CH2OH��E������������Ӧ����FΪCH3CHO��
������D��F�ķ���ʽ��֪��DΪ�������ھƻ�ø����������EΪCH3CH2OH��E������������Ӧ����FΪCH3CHO�� ��CH3CHO������Ϣ���еļӳɷ�ӦȻ����ȥ1����ˮ�õ�GΪ
��CH3CHO������Ϣ���еļӳɷ�ӦȻ����ȥ1����ˮ�õ�GΪ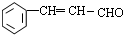 ��
��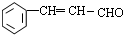 ��������Һ����������Ӧ���ữ�õ������HΪ
��������Һ����������Ӧ���ữ�õ������HΪ ��H���Ҵ�����������Ӧ����IΪ
��H���Ҵ�����������Ӧ����IΪ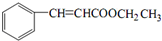 ����CH3CH2OHΪ��ʼԭ���Ʊ�1-��ϩ�����Խ��Ҵ���������ȩ��������ȩ������Ϣ���еķ�Ӧ����CH3CH=CHCHO��CH3CH=CHCHO���������ӳɵ�CH3CH2CH2CH2OH��������ȥ��Ӧ�ɵ�CH3CH2CH=CH2���ݴ˷������
����CH3CH2OHΪ��ʼԭ���Ʊ�1-��ϩ�����Խ��Ҵ���������ȩ��������ȩ������Ϣ���еķ�Ӧ����CH3CH=CHCHO��CH3CH=CHCHO���������ӳɵ�CH3CH2CH2CH2OH��������ȥ��Ӧ�ɵ�CH3CH2CH=CH2���ݴ˷������
��� �⣺A�������ڹ�������������������ȡ����Ӧ����B��B����±������ˮ�ⷴӦ������ȥ1����ˮ�õ� ����AΪ
����AΪ ��BΪ
��BΪ ������D��F�ķ���ʽ��֪��DΪ�������ھƻ�ø����������EΪCH3CH2OH��E������������Ӧ����FΪCH3CHO��
������D��F�ķ���ʽ��֪��DΪ�������ھƻ�ø����������EΪCH3CH2OH��E������������Ӧ����FΪCH3CHO�� ��CH3CHO������Ϣ���еļӳɷ�ӦȻ����ȥ1����ˮ�õ�GΪ
��CH3CHO������Ϣ���еļӳɷ�ӦȻ����ȥ1����ˮ�õ�GΪ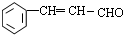 ��
��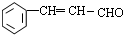 ��������Һ����������Ӧ���ữ�õ������HΪ
��������Һ����������Ӧ���ữ�õ������HΪ ��H���Ҵ�����������Ӧ����IΪ
��H���Ҵ�����������Ӧ����IΪ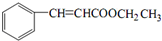 ��
��
��1��AΪ ��A�������� �ױ�������C�Ľṹ��ʽ��֪��C�к��еĹ����ŵ������� ȩ����
��A�������� �ױ�������C�Ľṹ��ʽ��֪��C�к��еĹ����ŵ������� ȩ����
�ʴ�Ϊ���ױ���ȩ����
��2��BΪ ��E��F�ķ�Ӧ������������Ӧ��
��E��F�ķ�Ӧ������������Ӧ��
�ʴ�Ϊ�� ��������Ӧ��
��������Ӧ��
��3��GΪ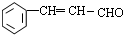 ������������ԭ�ӡ�̼̼˫��������ԭ�Ӽ�̼��˫��������ԭ�Ӷ����Թ��棬��������ת��������G�����������18��ԭ�ӹ�ƽ�棬
������������ԭ�ӡ�̼̼˫��������ԭ�Ӽ�̼��˫��������ԭ�Ӷ����Թ��棬��������ת��������G�����������18��ԭ�ӹ�ƽ�棬
�ʴ�Ϊ��18��
��4��H+E��I�Ļ�ѧ����ʽΪ ��
��
�ʴ�Ϊ�� ��
��
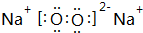
��5��HΪ �������������ܷ���������Ӧ��˵����ȩ������������������Һ������ɫ��Ӧ��˵���з��ǻ����۳������ⲻ���������������������H��ͬ���칹��ṹΪ����������-OH��-CH=CHCHO����-OH��-C��CHO��=CH2�������Ľṹ�����ڼ�����֣�Ҳ����������-OH��-CH=CH2��-CHO�������ţ������Ľṹ��10�֣����Թ���16�֣����к˴Ź�������5��壬�ҷ����֮��Ϊ1��2��2��2��1�Ľṹ��ʽΪ
�������������ܷ���������Ӧ��˵����ȩ������������������Һ������ɫ��Ӧ��˵���з��ǻ����۳������ⲻ���������������������H��ͬ���칹��ṹΪ����������-OH��-CH=CHCHO����-OH��-C��CHO��=CH2�������Ľṹ�����ڼ�����֣�Ҳ����������-OH��-CH=CH2��-CHO�������ţ������Ľṹ��10�֣����Թ���16�֣����к˴Ź�������5��壬�ҷ����֮��Ϊ1��2��2��2��1�Ľṹ��ʽΪ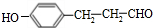 ��
��
�ʴ�Ϊ��16��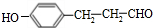 ��
��
��6��EΪCH3CH2OH����EΪ��ʼԭ���Ʊ�1-��ϩ�����Խ��Ҵ���������ȩ��������ȩ������Ϣ���еķ�Ӧ����CH3CH=CHCHO��CH3CH=CHCHO���������ӳɵ�CH3CH2CH2CH2OH��������ȥ��Ӧ�ɵ�CH3CH2CH=CH2���ϳ�·��ΪCH3CH2OH$��_{Cu��}^{O_{2}}$CH3CHO$��_{��}^{ϡ��}$CH3CH=CHCHO$��_{����}^{����}$CH3CH2CH2CH2OH$��_{��}^{ŨH_{2}SO_{4}}$CH3CH2CH=CH2��
�ʴ�Ϊ��CH3CH2OH$��_{Cu��}^{O_{2}}$CH3CHO$��_{��}^{ϡ��}$CH3CH=CHCHO$��_{����}^{����}$CH3CH2CH2CH2OH$��_{��}^{ŨH_{2}SO_{4}}$CH3CH2CH=CH2��
���� ���⿼���л����ƶϣ�Ϊ��Ƶ���㣬���ؿ���ѧ�������ƶ�������ע����ݷ�Ӧ�������л���ṹ�����ƶϣ���Ҫѧ���������չ����ŵ�������ת�����Ѷ��еȣ�

 ���Ž�������С״Ԫϵ�д�
���Ž�������С״Ԫϵ�д�| A�� | KCl | B�� | NaHSO4 | C�� | NaHCO3 | D�� | AlCl3 |
| A�� | ��������KOH���� | B�� | ������������ | ||
| C�� | �����������ƹ��� | D�� | ����������ˮ |

| A�� | ��������ķ���ʽΪC26H22Cl6O8 | |
| B�� | 1mol�����������������ϡ��Һ��Ӧʱ��������±�ز�ˮ�⣩���������6mol NaOH | |
| C�� | �����������������ˮ����Եõ������л��� | |
| D�� | 1mol���������������ȫ��Ӧ����Ҫ����10mol |
| A�� | ��������������������Ʒ�Ļ���ԭ�� | |
| B�� | ˮ���������������ϼ��ͷ���� | |
| C�� | ������ؿ�����ҽҩ�������� | |
| D�� | ̼���ƿ�����ϴ�Ӽ� |
| A�� | ��״���£�1L C2H5OH��ȫȼ�պ����ɵ�CO2���Ӹ���ԼΪNA/11.2 | |
| B�� | 18g 50%������ˮ��Һ��ԭ������ԼΪ1.6 NA | |
| C�� | 0�桢101KPa�£�4.48LNH3�й��õ��Ӷ���Ϊ0.6NA | |
| D�� | ��lmol Cl2ͨ��ˮ�У�HClO��Cl-��ClO- ������֮��Ϊ2NA |
| A�� | 1 mol��Ϊ2 g | B�� | H2O��Ħ������Ϊ18 g | ||
| C�� | 44 g CO2�����Ϊ22.4 L | D�� | 9.8 g H2SO4��0.1NA��H2SO4���� |
| A�� | Na2CO3 | B�� | NaOH | C�� | Na2SiO3 | D�� | CaCl2 |
 ��
�� ��
��