��Ŀ����
12����A��B��C��D����ǿ����ʣ�������ˮ�е���ʱ�ɲ����������ӣ�ÿ������ֻ��һ�������Ӻ�һ�������ӣ��һ����ظ�����| ������ | K+��Na+��Ba2+��NH4+ |
| ������ | CH3COO-��Cl-��OH-��SO42- |
��C��Һ��D��Һ����ʱֻ���ɰ�ɫ������B��Һ��C��Һ����ʱֻ�����д̼�����ζ�����壬A��Һ��D��Һ���ʱ����������
��1��A�Ļ�ѧʽΪCH3COONa��
��2�������ӷ���ʽ����B��Һ��pHС��7��ԭ��NH4++H2O?NH3•H2O+H+��
��3��д��C��Һ��D��Һ��Ӧ�����ӷ���ʽ��Ba2++SO42-=BaSO4����
��4����������������ʵ���Ũ�ȵ�B��Һ��C��Һ��ϣ���Ӧ����Һ�и�������Ũ���ɴ�С��˳��Ϊc��OH-����c��Ba2+��=c��Cl-����c��NH4+����c��H+����
���� A��C��Һ��pH������7��ӦΪ�����κͼ���Һ��B��Һ��pHС��7��ӦΪ�����Һ��A��B����Һ��ˮ�ĵ���̶���ͬ������Һˮ��̶���ͬ��D��Һ��ɫ��Ӧ������ɫ�ܲ���������ɫ����Һ�к��м����ӣ�
C��Һ��D��Һ����ʱֻ���ɰ�ɫ������B��Һ��C��Һ����ʱֻ���ɴ̼�����ζ�����壬A��Һ��D��Һ���ʱ������������˵��CΪBa��OH��2��DΪK2SO4����BΪNH4Cl������������AΪCH3COONa��BΪNH4Cl��CΪBa��OH��2��DΪK2SO4���Դ������
��� �⣺��1��������������֪��A�Ļ�ѧʽΪCH3COONa��
�ʴ�Ϊ��CH3COONa��
��2��BΪNH4Cl��笠�����ˮ��ʹ��Һ�����ԣ������ӷ���ʽ��ʾΪNH4++H2O?NH3•H2O+H+��
�ʴ�Ϊ��NH4++H2O?NH3•H2O+H+��
��3��C��Һ��D��Һ��Ӧ�����ӷ���ʽΪBa2++SO42-=BaSO4����
�ʴ�Ϊ��Ba2++SO42-=BaSO4����
��4���Ȼ�狀��������������ʵ�����ȣ���Ϻ���Һ�е�������һˮ�ϰ����Ȼ����������������Ȼ���������������Ũ����ȣ�һˮ�ϰ���Ũ�����Ȼ�������������Ũ�ȵ�2������Һ������������Ũ��������Ӻ�������Ũ����ȣ�һˮ�ϰ�������ֵ��뵼��������Ũ�ȴ���笠�����Ũ�ȣ���Һ�ʼ��ԣ�������Ũ����С��������Һ������Ũ�ȴ�С˳����c��OH-����c��Ba2+��=c��Cl-����c��NH4+����c��H+����
�ʴ�Ϊ��c��OH-����c��Ba2+��=c��Cl-����c��NH4+����c��H+����
���� ���⿼��������ƶϣ�Ϊ��Ƶ���㣬�������ʵ����ʡ������ķ�Ӧ������ˮ��Ϊ���Ĺؼ������ط������ƶ������Ŀ��飬ע���ɫ������������жϣ���Ŀ�ѶȲ���

 �������Ӧ���⼯ѵϵ�д�
�������Ӧ���⼯ѵϵ�д� �ۺ��Բ�ϵ�д�
�ۺ��Բ�ϵ�д�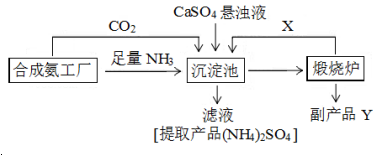
��ش��������⣺
��1�������йأ�NH4��2SO4��Һ��˵����ȷ����
A�����뷽��ʽ����NH4��2SO4?2NH4++SO42-
B��ˮ�����ӷ���ʽ��NH4++H2O?NH3•H2O+H+
C������Ũ�ȹ�ϵ��c��NH4+��+c��H+���Tc��SO42-��+c��OH-��
D����Ũ�ȴ�С��c��NH4+����c��SO42-����c��H+����c��NH3•H2O����c��OH-��
��2�����Ṥҵ�У�V2O5������ʱ������Ӧ2SO2+O2?2SO3��SO2��ת�������¶ȡ�ѹǿ�йأ�������±���Ϣ����Ϲ�ҵ����ʵ�ʣ�ѡ���������ʵ��¶Ⱥ�ѹǿ�ֱ���420�桢1.01��105Pa���÷�Ӧ420��ʱ��ƽ�ⳣ����520��ʱ��ƽ�ⳣ�������������������=������
 | 1.01��105Pa | 5.05��105Pa | 1.01��106Pa |
| 420�� | 0.9961 | 0.9972 | 0.9984 |
| 520�� | 0.9675 | 0.9767 | 0.9852 |
| 620�� | 0.8520 | 0.8897 | 0.9276 |
��4������ƷY�������ƣ��������з�������Ҫ��Ӧ����ʽ��CaSO4+CO2+2NH3+H2O��CaCO3��+��NH4��2SO4��
��5������ɫ��ѧ����Դ�ۺ����õĽǶ�˵���������̵���Ҫ�ŵ��Dz�����CO2ѭ��ʹ�ã����ʳ�����ã�����Ʒ���ã�����Ⱦ�����ʲ�����
| A�� | 75% | B�� | 50% | C�� | 5% | D�� | 12.5% |
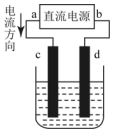
| A�� | c��������ԭ��Ӧ | B�� | ÿ����6.4gCu��ת�Ƶ���0.2mol | ||
| C�� | ������Ӧʽ��Cu2++2e��Cu | D�� | ����Һ�е�����d����c |
| A�� | ���������мɾ��ţ���ë����ס�ڱǣ���˳�緽�����ܷ�Ĺ������ݴ�ת�� | |
| B�� | Ҫ���ٵؽ�������Ȳ۹ް�ȫ����Ƶ�ˮ�أ����м�Һϡ���к� | |
| C�� | Һ�������ٶȺ��������ؾ��ţ����Է����˵����У��ؽ��Ŵ� | |
| D�� | �����ֳ�����ʱ��û�з�����ߣ����������Ũ����Һ������ʪ���� |
| ѡ�� | �ı������ | �����淴Ӧ���ʱ仯 | ��ƽ���ԭƽ��Ƚ� |
| A | ����ѹǿ | �����淴Ӧ���ʶ������� | Z��Ũ�ȼ�С |
| B | ��������Z | �淴Ӧ�������� | X������������� |
| C | ��������Z | v���棩��v������ | Z��Ũ�ȼ�С |
| D | �����¶� | �淴Ӧ�������������� ����Ӧ���������� | Z������������ |
| A�� | A | B�� | B | C�� | C | D�� | D |
| ʵ�� ��� | ��Ӧ�� ��/�� | �μӷ�Ӧ������ | ||||
| Na2S2O3 | H2SO4 | H2O | ||||
| V/mL | c/mol•L-1 | V/mL | c/mol•L-1 | V/mL | ||
| A | 20 | 10 | 0.1 | 10 | 0.1 | 0 |
| B | 20 | 5 | 0.1 | 10 | 0.1 | 5 |
| C | 20 | 10 | 0.1 | 5 | 0.1 | 5 |
| D | 40 | 5 | 0.1 | 10 | 0.1 | 5 |
��2���ڱȽ�ijһ���ض�ʵ�������Ӱ��ʱ�������ų��������صı䶯���ţ�����Ҫ���ƺ���ʵ���йصĸ��Ӧ���������У�
����˵���¶ȶԸ÷�Ӧ����Ӱ��������BD����ʵ����ţ���ͬ����
��A��B��A��C����ϱȽϣ����о�����������ͬ�¶�������Ũ�ȶԸ÷�Ӧ���ʵ�Ӱ�죻
��B��C����ϱȽϣ����о�����������ͬ�¶������£��÷�Ӧ���ʸ���̶���ȡ�������ַ�Ӧ���Ũ��
��3���̲��������˳��ֻ�ɫ�����Ŀ������ȽϷ�Ӧ���ʵĿ������������Ϊ�β�������ˮ��������λʱ������������Ĵ�С���бȽϣ�SO2������ˮ���ⶨ����ȷ��ʵ��װ�ýϸ��ӣ������ƣ�
| A�� | 3.12g | B�� | 4.28g | C�� | 4.68g | D�� | 5.08g |
| A�� | ú�������͵Ļ���� | B�� | ����ϡ����Ļ����Һ | ||
| C�� | ���Ȼ�̼�ͱ� | D�� | �Ҵ���ˮ�Ļ����Һ |