ΧβΡΩΡΎ»ί
(08ΈΪΖΜ÷ Φλ)”ΟNA±μ ΨΑΔΖπΦ”Β¬¬ό≥Θ ΐΒΡ÷ΒΘ§œ¬Ν–ΥΒΖ®’ΐ»ΖΒΡ «Θ® Θ©
AΘ°46gNO2ΚΆN2O4ΜλΚœΤχΧε÷–Κ§”–ΒΡΖ÷Ή”Ήή ΐΈΣNA
BΘ°”ΟΚ§4molHC1ΒΡ≈®―ΈΥαΗζΉψΝΩΕΰ―θΜ·ΟΧΖ¥”ΠΘ§…ζ≥…ΒΡ¬»Τχ‘Ύ±ξΉΦΉ¥Ωωœ¬ΒΡΧεΜΐΈΣ22.4L
CΘ°0.1molΫπ τΡΤ”κ―θΤχ‘Ύ“ΜΕ®ΧθΦΰœ¬ΖΔ…ζΖ¥”ΠΘ§…ζ≥…―θΜ·ΡΤΒΡΜλΚœΈο ±Θ§ ß»ΞΒΡΒγΉ”
ΐΈΣ0.1NA
DΘ°±ξΉΦΉ¥Ωωœ¬Θ§11.2L““¥ΦΆξ»Ϊ»Φ…’ ±≤ζ…ζΒΡΕΰ―θΜ·ΧΦΖ÷Ή” ΐΈΣNA
¥πΑΗΘΚC

ΝΖœΑ≤αœΒΝ–¥πΑΗ
 ΧλΧλœρ…œ“Μ±ΨΚΟΨμœΒΝ–¥πΑΗ
ΧλΧλœρ…œ“Μ±ΨΚΟΨμœΒΝ–¥πΑΗ –Γ―ß…ζ10Ζ÷÷””Π”ΟΧβœΒΝ–¥πΑΗ
–Γ―ß…ζ10Ζ÷÷””Π”ΟΧβœΒΝ–¥πΑΗ
œύΙΊΧβΡΩ
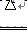 NH3Γϋ+H2O
NH3Γϋ+H2O