��Ŀ����
11��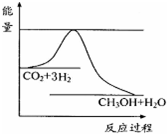 ������ʵ��д�����з�Ӧ���Ȼ�ѧ����ʽ
������ʵ��д�����з�Ӧ���Ȼ�ѧ����ʽ��1��Ŀǰ��ҵ����һ�ַ�������CO2�������״���һ�������·�����Ӧ��CO2��g��+3H2��g��?CH3OH��g��+H2O��g������ͼ��ʾ�÷�Ӧ���й�������������λΪkJ•mol-1���ı仯�������Ϊ1L�ĺ����ܱ������У�����1mol CO2��3mol H2�����д�ʩ����ʹc ��CH3OH���������cd��
a�������¶�
b������He��g����ʹ��ϵѹǿ����
c����H2O��g������ϵ�з������
d���ٳ���1mol CO2��3mol H2
��2����25�桢101kPa�£�1g�״�ȼ������CO2��Һ̬ˮʱ����22.68kJ�����ʾ�״�ȼ���ȵ��Ȼ�ѧ����ʽΪCH3OH��l��+$\frac{3}{2}$O2��g���TCO2��g��+2H2O��l����H=-725.8kJ•mol-1��
��3�����к��Ȳⶨʵ���У����β��������������Һ�ķ��������³鶯��������ͭ˿����������ԭ���ǽ����ĵ����Ժܺã��ᵼ��������ɢʧ��д��ϡ�����ϡ����������Һ��Ӧ��ʾ�к��ȵ��Ȼ�ѧ����ʽ���к���Ϊ-57.3kJ/mol��$\frac{1}{2}$H2SO4��aq��+NaOH��aq��=$\frac{1}{2}$Na2SO4��aq��+H2O��l����H=-57.3kJ/mol��
��4���¿���Ϊ�����������ȼ�ϣ���������N2O4��Ӧ����N2��ˮ������
��֪����N2��g��+2O2��g��=N2O4 ��1����H1=-19.5kJ•mol-1
��N2H4 ��1��+O2��g��=N2��g��+2H2O��g����H2=-534.2kJ•mol-1
д���º�N2O4 ��Ӧ���Ȼ�ѧ����ʽ2N2H4��1��+N2O4��1���T3N2��g��+4H2O��g����H=-1048.9kJ/mol��
���� ��1������ͼ��֪���÷�Ӧ������Ӧ�Ƿ��ȷ�Ӧ����ʹc��CH3OH������˵��ƽ��������Ӧ�����ƶ���
��2��ȼ������1mol��ȼ����ȫȼ�������ȶ�������ʱ�ų�����������25�桢101kPa�£�1g�״���CH3OH��ȼ������CO2��Һ̬ˮʱ����22.68kJ��32g�״�ȼ������CO2��Һ̬ˮʱ����22.68kJ��32=725.76kJ��1mol�״�����Ϊ32�ˣ�������ȫȼ��1mol�״����ɶ�����̼��Һ̬ˮ����725.76KJ������ȼ���ȵĸ���������ɽ��
��3�����β��������������Һ�ķ��������½��裬ʹ��Һ��Ͼ��ȣ����ݽ����ĵ����Ժܺã��ᵼ��������ɢʧ����������кͷ�Ӧ���������Ȼ�ѧ����ʽ����дԭ��õ���
��4����֪��N2��g��+2O2��g���TN2O4��g������H=+8.7kJ/mol��
��N2H4��g��+O2��g���TN2��g��+2H2O��g������H=-534.7kJ/mol��
���ø�˹���ɽ��ڡ�2-�ٿɵ�2N2H4��g��+N2O4��g���T3N2��g��+4H2O��g�������Դ˼��㷴Ӧ�ȣ�
��� �⣺��1��a�������¶ȣ�ƽ�������ƶ���c��CH3OH����С����a����
b������He��g����ʹ��ϵѹǿ������������Ϊ���������������Ũ�Ȳ��䣬ƽ�ⲻ�ƶ���c��CH3OH�����䣬��b����
c����H2O��g������ϵ�з��������ƽ�������ƶ���c��CH3OH������c��ȷ��
d���ٳ���1mol CO2��3molH2��ƽ�������ƶ���c��CH3OH������d��ȷ��
�ʴ�Ϊ��cd��
��2��1mol�״���ȫȼ�����ɶ�����̼��Һ̬ˮ����725.8KJ��ȼ�����Ȼ�ѧ����ʽΪ��CH3OH��l��+$\frac{3}{2}$O2��g���TCO2��g��+2H2O��l����H=-725.8 kJ•mol-1��
�ʴ�Ϊ��CH3OH��l��+$\frac{3}{2}$O2��g���TCO2��g��+2H2O��l����H=-725.8 kJ•mol-1��
��3�����к��Ȳⶨʵ���У����β��������������Һ�ķ��������³鶯��ʹ��Һ��Ͼ��ȣ��û���ͭ˿��������滷�β���������������ĵ����Ժܺã��ᵼ��������ɢʧ��ʹ��õġ�H��ֵƫС��ϡ�����NaOHϡ��Һ��Ӧ����1molˮ����57.3KJ���÷�Ӧ���Ȼ�ѧ����ʽΪ��$\frac{1}{2}$H2SO4��aq��+NaOH��aq��=$\frac{1}{2}$Na2SO4��aq��+H2O��l����H=-57.3 kJ/mol��
�ʴ�Ϊ�����³鶯�������ĵ����Ժܺã��ᵼ��������ɢʧ��$\frac{1}{2}$H2SO4��aq��+NaOH��aq��=$\frac{1}{2}$Na2SO4��aq��+H2O��l����H=-57.3 kJ/mol��
��4����N2��g��+2O2��g��=N2O4 ��1����H1=-19.5kJ•mol-1
��N2H4 ��1��+O2��g��=N2��g��+2H2O��g����H2=-534.2kJ•mol-1
���ݸ�˹���ɼ���ڡ�2-�ٵõ��º�N2O4 ��Ӧ���Ȼ�ѧ����ʽ2N2H4 ��1��+N2O4��1���T3N2��g��+4H2O��g����H=-1048.9 kJ/mol��
�ʴ�Ϊ��2N2H4 ��1��+N2O4��1���T3N2��g��+4H2O��g����H=-1048.9 kJ/mol��
���� ������Ҫ�����˻�ѧƽ��Ӱ�����ء��Ȼ�ѧ����ʽ����д����㡢�к���ʵ������Լ��к��ȵĸ���ѶȲ���ע��ʵ��ԭ�������գ�

| A�� | pH=12����Һ�У�K+��Na+��AlO2-��CO32- | |
| B�� | ��0.1mol•L-1��NaHCO3 ��Һ�У�K+��Al3+��I-��Cl- | |
| C�� | ��Al��Ӧ�ܷų�H2����Һ�У�Fe2+��K+��NO3-��SO42- | |
| D�� | ʹ��̪���ɫ����Һ�У�Na+��Al3+��SO42-��Cl- |
X����CH4+10OH--8e-=CO32-+7H2O
Y����O2+4e-+2H2O=4OH-
���й��ڴ�ȼ�ϵ�ص��й�˵���д�����ǣ�������
| A�� | ͨ������ĵ缫��X��Ϊ��صĸ�����ͨ�������ĵ缫��Y��Ϊ������� | |
| B�� | �ŵ�һ��ʱ��������Һ��KOH�����ʵ������ı� | |
| C�� | �ڱ�״���£�ͨ��5.6L��������ȫ��Ӧ����1.0mol�ĵ��ӷ���ת�� | |
| D�� | �ŵ�ʱ��ͨ�������һ��������pH���� |
| A�� | ��ˮ�е�������NH3•H2O | |
| B�� | NH3�м��ԣ�������ʹʪ��ĺ�ɫʯ����ֽ����ɫ | |
| C�� | ���е���ζ�������ˮ���������ֽ� | |
| D�� | �����£�Ũ���ᡢŨ������������������Ӧ�����Կ������Ƶ���������������� |