��Ŀ����
�������Σ�K2FeO4��Na2FeO4������Դ�������ȷ������Ź㷺����;����������أ�K2FeO4�����м�ǿ�������ԣ���һ��������ˮ���������ɷ���ʪ���Ʊ��������ε�ԭ�����±���ʾ��
��1���ɷ��Ʊ�K2FeO4�Ļ�ѧ����ʽΪFe2O3+3KNO3+4KOH
2K2FeO4+3KNO2+2H2O���÷�Ӧ���������뻹ԭ�������ʵ���֮��Ϊ ��
��2�����������ˮ�м�������ɱ�������ܾ�ˮ����һ�������ˮ�������������FeO42-��ˮ��Ӧ�ķ���ʽ��4FeO42-+10H2O?4Fe��OH��3+8OH-+ ��
��3��K2FeO4�ڴ���ˮ�Ĺ�����������ɱ������Ϊ�� �����ܾ�ˮ��ԭ���Ǣ� ��
��4����ҵ����ʪ���Ʊ�������أ�K2FeO4����������ͼ��ʾ��
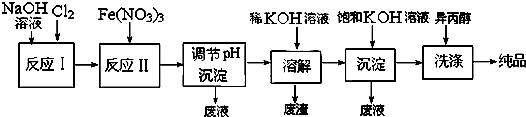
�ٷ�ӦI�Ļ�ѧ����ʽΪ ��
�ڷ�Ӧ������ӷ���ʽΪ ��
�ۼ��뱥��KOH��Һ��Ŀ���� ��
| �ɷ� | Fe2O3��KNO3��KOH��ϼ��ȹ��������Ϻ�ɫK2FeO4��KNO2�Ȳ��� |
| ʪ�� | ǿ���Խ����У�Fe��NO3��3��NaClO��Ӧ�����Ϻ�ɫNa2FeO4��Һ |
| ||
��2�����������ˮ�м�������ɱ�������ܾ�ˮ����һ�������ˮ�������������FeO42-��ˮ��Ӧ�ķ���ʽ��4FeO42-+10H2O?4Fe��OH��3+8OH-+
��3��K2FeO4�ڴ���ˮ�Ĺ�����������ɱ������Ϊ��
��4����ҵ����ʪ���Ʊ�������أ�K2FeO4����������ͼ��ʾ��

�ٷ�ӦI�Ļ�ѧ����ʽΪ
�ڷ�Ӧ������ӷ���ʽΪ
�ۼ��뱥��KOH��Һ��Ŀ����
���㣺���ʷ�����ᴿ�ķ����ͻ��������ۺ�Ӧ��,�Ʊ�ʵ�鷽�������
ר�⣺ʵ�������
��������1����ӦFe2O3+3KNO3+4KOH
2K2FeO4+3KNO2+2H2O�У�FeԪ�ػ��ϼ����ߣ����ϼ���+3�����ߵ�+6�ۣ���������NԪ�ػ��ϼ���+5�۽��͵�+2�ۣ���Ϸ���ʽ�жϣ�
��2��FeO42-��ˮ��Ӧ����������Fe��OH��3��֪��FeԪ�ػ��ϼ۽��ͣ�Ӧ����������ԭ��Ӧ��ͬʱ����������
��3��K2FeO4����ǿ�����ԣ�������ɱ��������������Fe��OH��3�����������ԣ�
��4����Ӧ��2NaOH+Cl2�TNaCl+NaClO+H2O����Ӧ��3ClO-+10OH-+2Fe3+=2FeO42-+3Cl-+5H2O�����뱥��KOH��Һ��������K+��Ũ�ȣ�ʹƽ�������ƶ����Դ˽����⣮
| ||
��2��FeO42-��ˮ��Ӧ����������Fe��OH��3��֪��FeԪ�ػ��ϼ۽��ͣ�Ӧ����������ԭ��Ӧ��ͬʱ����������
��3��K2FeO4����ǿ�����ԣ�������ɱ��������������Fe��OH��3�����������ԣ�
��4����Ӧ��2NaOH+Cl2�TNaCl+NaClO+H2O����Ӧ��3ClO-+10OH-+2Fe3+=2FeO42-+3Cl-+5H2O�����뱥��KOH��Һ��������K+��Ũ�ȣ�ʹƽ�������ƶ����Դ˽����⣮
���
�⣺��1����ӦFe2O3+3KNO3+4KOH
2K2FeO4+3KNO2+2H2O�У�FeԪ�ػ��ϼ����ߣ����ϼ���+3�����ߵ�+6�ۣ���������NԪ�ػ��ϼ���+5�۽��͵�+2�ۣ��������뻹ԭ�������ʵ���֮��Ϊ3��1��
�ʴ�Ϊ��3��1��
��2��FeO42-��ˮ��Ӧ����������Fe��OH��3��֪��FeԪ�ػ��ϼ۽��ͣ�Ӧ����������ԭ��Ӧ��ͬʱ������������Ӧ�ķ���ʽΪ4FeO42-+10H2O?4Fe��OH��3+8OH-+3O2��
�ʴ�Ϊ��3O2��
��3��K2FeO4����ǿ�����ԣ�������ɱ��������������Fe��OH��3�����������ԣ��ɳ�ȥˮ�����������ʣ�
�ʴ�Ϊ���ٸ��������ǿ�����ԣ��ڸ��������ˮ��Ӧ���ɵ�Fe��OH��3�����������ԣ�
��4���ٷ�Ӧ��Ϊ���ֽⷴӦ������ʽΪ��2NaOH+Cl2�TNaCl+NaClO+H2O���ʴ�Ϊ��Cl2+2NaOH�TNaCl+NaClO+H2O��
�ڷ�Ӧ��Ϊ������ԭ��Ӧ��+3������������+6������+1�۵��ȱ�����Ϊ-1�ۣ�����ʽ��3ClO-+10OH-+2Fe3+=2FeO42-+3Cl-+5H2O��
�ʴ�Ϊ��3ClO-+10OH-+2Fe3+�T2FeO42-+3Cl-+5H2O��
��Na2FeO4+2KOH?K2FeO4+2NaOH�����뱥��KOH��Һ��������K+��Ũ�ȣ�ʹƽ�������ƶ����������壬�ʴ�Ϊ������K+��Ũ�ȣ��ٽ�K2FeO4����������
| ||
�ʴ�Ϊ��3��1��
��2��FeO42-��ˮ��Ӧ����������Fe��OH��3��֪��FeԪ�ػ��ϼ۽��ͣ�Ӧ����������ԭ��Ӧ��ͬʱ������������Ӧ�ķ���ʽΪ4FeO42-+10H2O?4Fe��OH��3+8OH-+3O2��
�ʴ�Ϊ��3O2��
��3��K2FeO4����ǿ�����ԣ�������ɱ��������������Fe��OH��3�����������ԣ��ɳ�ȥˮ�����������ʣ�
�ʴ�Ϊ���ٸ��������ǿ�����ԣ��ڸ��������ˮ��Ӧ���ɵ�Fe��OH��3�����������ԣ�
��4���ٷ�Ӧ��Ϊ���ֽⷴӦ������ʽΪ��2NaOH+Cl2�TNaCl+NaClO+H2O���ʴ�Ϊ��Cl2+2NaOH�TNaCl+NaClO+H2O��
�ڷ�Ӧ��Ϊ������ԭ��Ӧ��+3������������+6������+1�۵��ȱ�����Ϊ-1�ۣ�����ʽ��3ClO-+10OH-+2Fe3+=2FeO42-+3Cl-+5H2O��
�ʴ�Ϊ��3ClO-+10OH-+2Fe3+�T2FeO42-+3Cl-+5H2O��
��Na2FeO4+2KOH?K2FeO4+2NaOH�����뱥��KOH��Һ��������K+��Ũ�ȣ�ʹƽ�������ƶ����������壬�ʴ�Ϊ������K+��Ũ�ȣ��ٽ�K2FeO4����������
���������⿼��ʵ�鷽����������ۣ�Ϊ�߿������һ�����ͣ�ÿ��߿��ؿ������ȵ����͡�Ҳ�Ǹ߿��ѵ�֮һ��������Ҫ������������ԭ��Ӧ�����ӷ�Ӧ����ʽ����д����ѧƽ����ƶ�������ľ�ˮԭ�����ص������û��ϼ۵����ۺͻ�ѧ�������ж��������ͻ�ԭ���ı������Ѷ��еȣ�

��ϰ��ϵ�д�
 53������ϵ�д�
53������ϵ�д�
�����Ŀ

 ij�о���ѧϰС���õ��CuSO4��Һ�ķ����ⶨͭ�����ԭ��������װ����ͼ����ʵ���в��ij�缫����������Ϊm g����֪��Ԫ�����ԭ������Ϊ16����ش�
ij�о���ѧϰС���õ��CuSO4��Һ�ķ����ⶨͭ�����ԭ��������װ����ͼ����ʵ���в��ij�缫����������Ϊm g����֪��Ԫ�����ԭ������Ϊ16����ش�