��Ŀ����
��1����0.1mol/L��KHSO4��Һ��0.1mol/L��HCl��Һ�е�������Ũ����ͬ����KHSO4�ĵ��뷽��ʽ���Ա�ʾΪ ��
a����0.1mol/L��KHSO4��Һ�м���пƬ��Ԥ�ƿ��Թ۲쵽�������� ����Ӧ�����ӷ���ʽΪ ��
b��ijͬѧ��һ������KHSO4��Һ�еμ�Ba��OH��2��Һ�����Һ�����ԣ���ʱ�ı仯���������ӷ���ʽ��ʾΪ �������μ�Ba��OH��2��Һ����仯���������ӷ���ʽ��ʾΪ ��
��2����п����������þ���ֽ����е�������ɵĻ����10g �����������ᷴӦ�����������ڱ����Ϊ11.2L����������һ�����еĽ��� ��
��3����150����̼�����ȫ�ֽ��Ϊ��̬�����[��NH4��2CO3
2NH3+CO2+H20]������̬������ܶ�����ͬ�����������ܶȵ� ����
a����0.1mol/L��KHSO4��Һ�м���пƬ��Ԥ�ƿ��Թ۲쵽��������
b��ijͬѧ��һ������KHSO4��Һ�еμ�Ba��OH��2��Һ�����Һ�����ԣ���ʱ�ı仯���������ӷ���ʽ��ʾΪ
��2����п����������þ���ֽ����е�������ɵĻ����10g �����������ᷴӦ�����������ڱ����Ϊ11.2L����������һ�����еĽ���
��3����150����̼�����ȫ�ֽ��Ϊ��̬�����[��NH4��2CO3
| ||
���㣺���ӷ���ʽ����д,���뷽��ʽ����д,��ѧ����ʽ���йؼ���
ר�⣺���ӷ�Ӧר��
��������1��0.1mol/L��KHSO4��Һ��0.1mol/L��HCl��Һ�е�������Ũ����ͬ����֪���������ˮ��Һ����ȫ����������ӡ������Ӻ���������ӣ�
a��KHSO4��Һ�м���пƬ����������п����������ԭ��Ӧ����������п���ӣ�
b��KHSO4��Һ�еμ�Ba��OH��2��Һ�����Һ�����ԣ���Ӧ�������ᱵ������ء�ˮ�������μ�Ba��OH��2��Һ�������������������Ӧ�������ᱵ�������������أ�
��2������n=
�������������ʵ��������ת�Ƶ���������ϻ��ϼۼ����ṩ1mol������Ҫ�����������������ƽ��ֵ���жϽ��
��3���ɷ���ʽ��֪�����ɵĻ�������а�����ˮ������������̼��Ħ�������ֱ�Ϊ0.5��0.25��0.25������ƽ����Է�������=Mr1��x1+Mr2��x2+��+Mri��xi����������ƽ����Է�������������Mri�����������Է���������xi���������Ħ�����������������ܶ�֮�ȵ�����Է�������֮�ȼ��㣮
a��KHSO4��Һ�м���пƬ����������п����������ԭ��Ӧ����������п���ӣ�
b��KHSO4��Һ�еμ�Ba��OH��2��Һ�����Һ�����ԣ���Ӧ�������ᱵ������ء�ˮ�������μ�Ba��OH��2��Һ�������������������Ӧ�������ᱵ�������������أ�
��2������n=
| V |
| Vm |
��3���ɷ���ʽ��֪�����ɵĻ�������а�����ˮ������������̼��Ħ�������ֱ�Ϊ0.5��0.25��0.25������ƽ����Է�������=Mr1��x1+Mr2��x2+��+Mri��xi����������ƽ����Է�������������Mri�����������Է���������xi���������Ħ�����������������ܶ�֮�ȵ�����Է�������֮�ȼ��㣮
���
�⣺��1��������0.1mol/L��KHSO4��Һ��0.1mol/L��HCl��Һ�е�������Ũ����ͬ����֪���������ˮ��Һ����ȫ����������ӡ������Ӻ���������ӣ����뷽��ʽΪ��KHSO4�TK++H++SO42-��
a��KHSO4��Һ�м���пƬ����������п����������ԭ��Ӧ����������п���ӣ����ӷ���ʽΪ��Zn+2H+�TZn2++H2��������Ϊ��������ɫ��ζ���ݣ�
b��KHSO4��Һ�еμ�Ba��OH��2��Һ�����Һ�����ԣ���Ӧ�������ᱵ������ء�ˮ�����ӷ���ʽΪ��Ba2++2OH-+2H++SO42-�TBaSO4����+2H2O�������μ�Ba��OH��2��Һ�������������������Ӧ�������ᱵ�������������أ����ӷ���ʽΪ��Ba2++SO42-�TBaSO4����
�ʴ�Ϊ��KHSO4�TK++H++SO42-��
a����ζ���ݣ�Zn+2H+�TZn2++H2����
b��Ba2++2OH-+2H++SO42-�TBaSO4��+2H2O��Ba2++SO42-�TBaSO4������
��2�������ڱ����Ϊ11.2L�������ʵ���=
=0.5mol����ת�Ƶ�����Ϊ0.5mol��2=1mol������������ԭ��Ӧ��ʧ�����غ���ɿ�֪10g����Ӧ�ṩ1mol���ӣ�п����������þ���ᷴӦ�ֱ�õ������ӵĻ��ϼ�Ϊ+2��+2��+3��+2��32.5g����28g��þ12g����9g���ṩ1mol���ӷֱ���Ҫп����������þ����Ϊ��п32.5g����28g��þ12g����9g������ƽ��ֵ˼�룬������е����ֽ�����Ҫ��Ӧ�ֱ�С�ںʹ���10g����С�ڻ����10g��ֻ��������һ��������
�ʴ�Ϊ��������Al����
��3���ɣ�NH4��2CO3
2NH3+CO2+H20��֪�����ɵĻ�������а�����ˮ������������̼��Ħ�������ֱ�Ϊ0.5��0.25��0.25����������
�T17��0.5+18��0.25+44��0.25=24�����Ի�������ܶ�����ͬ�����µ������ܶȵ�
=12����
�ʴ�Ϊ��12��
a��KHSO4��Һ�м���пƬ����������п����������ԭ��Ӧ����������п���ӣ����ӷ���ʽΪ��Zn+2H+�TZn2++H2��������Ϊ��������ɫ��ζ���ݣ�
b��KHSO4��Һ�еμ�Ba��OH��2��Һ�����Һ�����ԣ���Ӧ�������ᱵ������ء�ˮ�����ӷ���ʽΪ��Ba2++2OH-+2H++SO42-�TBaSO4����+2H2O�������μ�Ba��OH��2��Һ�������������������Ӧ�������ᱵ�������������أ����ӷ���ʽΪ��Ba2++SO42-�TBaSO4����
�ʴ�Ϊ��KHSO4�TK++H++SO42-��
a����ζ���ݣ�Zn+2H+�TZn2++H2����
b��Ba2++2OH-+2H++SO42-�TBaSO4��+2H2O��Ba2++SO42-�TBaSO4������
��2�������ڱ����Ϊ11.2L�������ʵ���=
| 11.2L |
| 22.4L/mol |
�ʴ�Ϊ��������Al����
��3���ɣ�NH4��2CO3
| ||
. |
| Mr |
| 24 |
| 12 |
�ʴ�Ϊ��12��
���������⿼�������ӷ���ʽ����ⷽ��ʽ���й����ӷ���ʽ�ļ��㣬��Ŀ�Ѷ��еȣ���ȷ����ʵ�ǿ����������Ӧ��ʵ���ǽ���ؼ���ע������ƽ��Ħ�������ڻ�ѧ�����е�Ӧ�÷�����

��ϰ��ϵ�д�
�����Ŀ
������Ԫ��X��Y��Z��Ԫ�����ڱ��е�λ����ͼ��ʾ������˵��������ǣ�������


| A��X��Y��Z����Ԫ���У�Y�ķǽ�������ǿ |
| B��Y���⻯����ȶ��Ա�Z���⻯��ǿ |
| C��Z����������ϼ�Ϊ+6 |
| D����ѹ��Z�ĵ��ʵ��۵��Y�ĵ� |
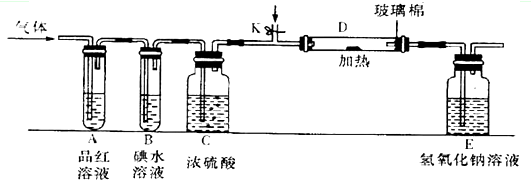
�����йض��������˵���У�������ǣ�������
| A�����������ˮ��Һ��ʹ��ɫʯ����Һ��죬˵�����ܺ�ˮ��Ӧ����H2SO3 |
| B������������Ư��ijЩ���ʣ�˵�������������� |
| C����������������ͨ�����Ը��������Һ�У���Һ��ɫ��˵������������л�ԭ�� |
| D��ʵ���ҿ���Ũ������������������ |
��ͬ��ͬѹ�£�10L����A2��30L����B2ǡ����ȫ��������20L����C��������C�Ļ�ѧʽ����A��B��ʾ��Ϊ��������
| A��AB |
| B��A2B |
| C��AB2 |
| D��AB3 |
���л�ѧ�����ʾ����ȷ���ǣ�������
| A������þ�Ļ�ѧʽ��MgN | ||||
B��������Ϊ18����ԭ�ӣ�
| ||||
| C��Ca��OH��2�ĵ��뷽��ʽ��Ca��OH��2�TCa2++2OH- | ||||
D������������ȼ�գ�2Fe+3Cl2
|