��Ŀ����
������Ӱ�����ǵ������뽡����ij�����������п��ܺ������¿����������ӣ�Na+��NH4+��Mg2+��Al3+��SO42����NO3����Cl����ijͬѧ�ռ��˸õ���������������Ҫ��Ԥ�������������Һ����Ʋ���������µ�ʵ�飺

��֪��3NO3��+8Al +5OH��+2H2O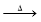 3NH3��+8AlO2��
3NH3��+8AlO2��
�������ϵ�ʵ�����������ͬѧ�ó��Ľ��۴������
A�������п϶�����NH4+��Mg2+��SO42����NO3��
B�������п��ܴ���Na+��Cl��
C������ȷ���������ijɷ���NaNO3��NH4Cl��MgSO4
D��������һ������Al3+
C��D
��������
�����������������Һ�м��������Ba(OH)2��Һ�������ܹ�ʹʪ��ĺ�ɫʯ����ֽ��Ϊ��ɫ������1����������ǰ���������Һ�к���NH4+��ͬʱ����������1����ó����м����ᣬ���������ܽ⣬֤�������к���BaSO4��Mg(OH)2������ԭ��Һ�к���Mg2+��SO42��������Һ1��ͨ��CO2���壬��������2����Һ2������2�м����ᣬ�����ܽⲢ�ų����壬������к���BaCO3�����Dz���ȷ���������Ƿ����Al(OH)3,Ҳ�Ͳ���ȷ������Һ1���Ƿ�Al3+�����к���Ba2+;����Һ2�м���Al���Ӽ���Ȳ�������2���������ܹ�ʹʪ��ĺ�ɫʯ����ֽ��Ϊ��ɫ��֤������Һ�к���NO3-,��ԭ��Һ�к���NO3-������������֪�������п϶�����NH4+��Mg2+��SO42����NO3��������ȷ����������Na+��Al3+��Cl�������ѡ��������C��D��
���㣺�������ӵļ��顢��Һ�ɷֵ�ȷ����֪ʶ��