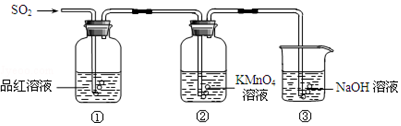
��Ŀ����
10������A����Ҫ���л�����ԭ�ϣ���A�����·�Ӧ���Ʊ�һ���л�������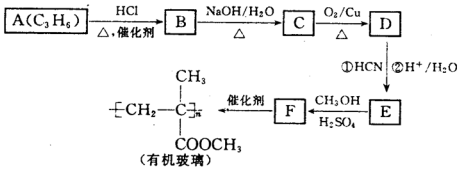
��֪������Ϣ��
�ٺ˴Ź������ױ���Dֻ��һ�ֻ�ѧ�������⣻
���ʻ�������ɷ������·�Ӧ��
 $��_{��H+/H_{2}O}^{��HCN}$
$��_{��H+/H_{2}O}^{��HCN}$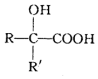 ��ע��R�������������Ҳ������Hԭ�ӣ�
��ע��R�������������Ҳ������Hԭ�ӣ���E�ڼ״�������������£�������������ˮ��Ӧ����F��
�ش��������⣺
��1��A�Ľṹ��ʽΪCH2=CHCH3��A����B�ķ�Ӧ����Ϊ�ӳɷ�Ӧ��
��2��B����C�Ļ�ѧ����ʽΪCH3CHClCH3+NaOH$��_{��}^{H_{2}O}$CH3CH��OH��CH3+NaCl��
��3��D�Ľṹ��ʽΪCH3COCH3�������������6��ԭ�ӹ�ƽ�森
��4��F�Ļ�ѧ����Ϊ2-��-��ϩ�������
��5��F��ͬ���칹������ͬʱ�������������Ĺ���8�֣����������칹�������к˴Ź���������ʾΪ4��壬�ҷ������Ϊ3��2��2��1����CH2=C��CH3��CH2COOH�� ��д������һ�ֵĽṹ��ʽ����
�����뱥��NaHCO3��Һ��Ӧ�������� ����ʹBr2�����Ȼ�̼��Һ��ɫ
��6�������ᣨ
 ����һ������ɽ�����ϣ��ο�������Ϣ������Ҵ��Ʊ�������ĺϳ�·��
����һ������ɽ�����ϣ��ο�������Ϣ������Ҵ��Ʊ�������ĺϳ�·��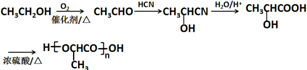 ���ϳ�·������ͼͼʾ�����£�X$��_{��Ӧ����}^{��Ӧ��}$Y$��_{��Ӧ����}^{��Ӧ��}$Z��Ŀ����
���ϳ�·������ͼͼʾ�����£�X$��_{��Ӧ����}^{��Ӧ��}$Y$��_{��Ӧ����}^{��Ӧ��}$Z��Ŀ����
���� F�����Ӿ۷�Ӧ�����л���������F�ṹ��ʽΪCH2=C��CH3��COOCH3��E�ͼ״�����������Ӧ����F��E�ṹ��ʽΪCH2=C��CH3��COOH��
A������������A����ʽ֪��A�ṹ��ʽΪCH2=CHCH3��A��HCl�����ӳɷ�Ӧ����B��B����ȡ����Ӧ����C��C������������Ӧ����D���˴Ź������ױ���Dֻ��һ�ֻ�ѧ�������⣬��D�ṹ��ʽΪCH3COCH3��C�ṹ��ʽΪCH3CH��OH��CH3��B�ṹ��ʽΪCH3CHClCH3��D�����ӳɷ�ӦȻ���ữ�õ�E��
��6�����Ҵ�Ϊ��ʼԭ���Ʊ�������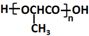 ���Ҵ�������������ȩ����ȩ��HCN�����ӳɷ�Ӧ��Ȼ��ˮ���������ᣬ�����Ӿ۷�Ӧ�����ɾ����ᣮ
���Ҵ�������������ȩ����ȩ��HCN�����ӳɷ�Ӧ��Ȼ��ˮ���������ᣬ�����Ӿ۷�Ӧ�����ɾ����ᣮ
��� �⣺��1��A�Ľṹ��ʽΪCH2=CHCH3��A����B�ķ�Ӧ����Ϊ�ӳɷ�Ӧ��
�ʴ�Ϊ��CH2=CHCH3���ӳɷ�Ӧ��
��2��C�ṹ��ʽΪCH3CH��OH��CH3��B�ṹ��ʽΪCH3CHClCH3��B����C�Ļ�ѧ����ʽΪCH3CHClCH3+NaOH$��_{��}^{H_{2}O}$CH3CH��OH��CH3+NaCl��
�ʴ�Ϊ��CH3CHClCH3+NaOH$��_{��}^{H_{2}O}$CH3CH��OH��CH3+NaCl��
��3��D�Ľṹ��ʽΪCH3COCH3���ʻ�Ϊƽ��ṹ�������������6��ԭ�ӹ�ƽ�棬
�ʴ�Ϊ��CH3COCH3��6��
��4��F�ṹ��ʽΪCH2=C��CH3��COOCH3����ѧ����Ϊ2-��-��ϩ�������
�ʴ�Ϊ��2-��-��ϩ�������
��5��F�ṹ��ʽΪCH2=C��CH3��COOCH3��F��ͬ���칹������ͬʱ��������������
�����뱥��NaHCO3��Һ��Ӧ�������壬˵�������Ȼ���
����ʹBr2�����Ȼ�̼��Һ��ɫ˵������̼̼˫����
-COOH��C=C-C-C��������4�֣�
-COOH��C-C=C-C������2�֣�
-COOH��C-C��CH3��=C������2�֣�
���������Ľṹ��8�֣�
���к˴Ź���������ʾΪ4��壬�ҷ������Ϊ3�s2�s2�s1����CH2=C��CH3��CH2COOH��
�ʴ�Ϊ��8��CH2=C��CH3��CH2COOH��
��6�����Ҵ�Ϊ��ʼԭ���Ʊ�������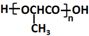 ���Ҵ�������������ȩ����ȩ��HCN�����ӳɷ�Ӧ��Ȼ��ˮ���������ᣬ�����Ӿ۷�Ӧ�����ɾ����ᣬ����Ϊ
���Ҵ�������������ȩ����ȩ��HCN�����ӳɷ�Ӧ��Ȼ��ˮ���������ᣬ�����Ӿ۷�Ӧ�����ɾ����ᣬ����Ϊ ��
��
�ʴ�Ϊ�� ��
��
���� ���⿼���л���ĺϳɣ�Ϊ��Ƶ���㣬���պϳ������й����ŵı仯���л���ӦΪ���Ĺؼ������ط�����Ӧ�������Ŀ��飬ע���л������ʵ�Ӧ�ã���Ŀ�ѶȲ���

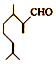 ����һ����Ҫ�����ϣ�������������������ɶ�������ױ���
����һ����Ҫ�����ϣ�������������������ɶ�������ױ��� �� �����й���Ҷȩ���������ױ���������ȷ���ǣ�������
�� �����й���Ҷȩ���������ױ���������ȷ���ǣ�������| A�� | ����Ϊͬ���칹�� | |
| B�� | ���߾�������ˮ�����ӳɷ�Ӧ | |
| C�� | ��Ҷȩ��һ��ͬ���칹������Ƿ��㴼 | |
| D�� | ��������ױ��ķ���ʽΪC10H14 |
| A�� | �ߴ��ȵĹ赥�ʹ㷺�����������ά | |
| B�� | ����β���е�������IJ�����Ҫ�����������к��е�Ԫ�� | |
| C�� | ��պ��Ũ��ˮ���ް��������������Ĺܵ��Ƿ�©�� | |
| D�� | �����������Ի�ѧҩƷ���У��Ҵ�������������Һ�����Խ������������ﵽ������Ŀ�� |
| A�� | Fe | B�� | CH4 | C�� | H2SO4 | D�� | NaNO3 |
| A�� | ��ľȡ�� | B�� | ��ˮ�ɱ� | C�� | ��ɳ���� | D�� | ����ĥ���� |